Complexos de Química de Coordenação
Visão Geral
Fonte: Laboratório do Dr. Neal Abrams — SUNY College of Environmental Science and Forestry
Metais de transição são encontrados em todos os lugares, desde suplementos vitamínicos até banhos de eletroplaca. Metais de transição também compõem os pigmentos em muitas tintas e compõem todos os minerais. Tipicamente, metais de transição são encontrados na forma cationic, uma vez que facilmente oxidam, ou perdem elétrons, e são cercados por doadores de elétrons chamados ligantes. Estes ligantes não formam ligações iônicas ou covalentes com o centro metálico, em vez disso, assumem um terceiro tipo de ligação conhecida como coordenada-covalente. A ligação coordenada-covalente entre um ligante e um metal é dinâmica, o que significa que os ligantes estão continuamente trocando e coordenando ao redor do centro metálico. As identidades do metal e do ligante ditam quais ligantes se ligarão preferencialmente sobre outro. Além disso, propriedades coloridas e magnéticas também se devem aos tipos de complexos que se formam. Os compostos de coordenação que formam são analisados utilizando uma variedade de instrumentos e ferramentas. Este experimento explora por que tantos complexos são possíveis e usa um método espectroquímico (cor e química) para ajudar a identificar o tipo de complexo de coordenação que se forma.
Procedimento
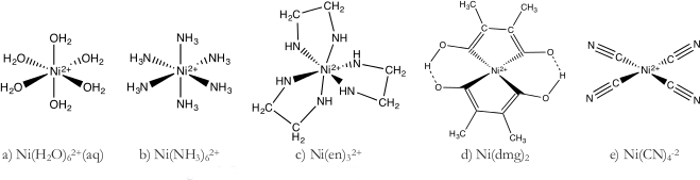
1. Complexos e Cores de Níquel
- Ni(H2O)62+ complexo(Figura 1a)
- Prepare uma solução de 1 M de Ni(H2O)62+ dissolvendo o NiSO4 no volume adequado de água.
- Diluir ainda mais a solução Ni(H2O)62+,adicionando 70 mL da solução de 1 M a 1.000 mL de água desionizada.
- Divida o Ni(H2O)62+ entre sete béquers de 400 mL.
- A soluç
Aplicação e Resumo
De pigmentos a pessoas, metais transitórios são encontrados em campos de química, biologia, geologia e engenharia. Entender o comportamento dos metais de transição em diferentes estados químicos pode ser tão simples quanto monitorar a cor ou o comportamento magnético. Quase todas as 3d (4ª linha) o metal de transição é vital para a função fisiológica e, em todos os casos, esses metais são obrigados por ligantes a formar complexos de coordenação. Por exemplo, o ferro é vital para o transporte...
Tags
Pular para...
Vídeos desta coleção:

Now Playing
Complexos de Química de Coordenação
General Chemistry
91.8K Visualizações

Vidraria de laboratório comuns e seus usos
General Chemistry
659.1K Visualizações

Soluções e Concentrações
General Chemistry
275.4K Visualizações

Determinando a densidade de um sólido e um líquido
General Chemistry
557.0K Visualizações

Determinação de composição percentual em massa em uma solução aquosa
General Chemistry
383.9K Visualizações

Determinação da Fórmula Empírica
General Chemistry
183.8K Visualizações

Determinação das Regras de Solubilidade de Compostos Iônicos
General Chemistry
141.6K Visualizações

Usando um medidor de pH
General Chemistry
347.0K Visualizações

Introdução à Titulação
General Chemistry
425.8K Visualizações

Lei dos gases ideais
General Chemistry
79.4K Visualizações

Determinação espectrofotométrica de uma constante de equilíbrio
General Chemistry
158.9K Visualizações

Princípio de Le Châtelier
General Chemistry
265.8K Visualizações

Depressão do ponto de congelamento para determinar um composto desconhecido
General Chemistry
160.8K Visualizações

Determinação das Leis de Velocidade e da Ordem de Reação
General Chemistry
196.4K Visualizações

Uso de Calorimetria de Varredura Diferencial para Medir Mudanças na Entalpia
General Chemistry
44.8K Visualizações
Copyright © 2025 MyJoVE Corporation. Todos os direitos reservados