A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
O-Ring Aortic Banding Versus Traditional Transverse Aortic Constriction for Modeling Pressure Overload-Induced Cardiac Hypertrophy
In This Article
Summary
The present protocol describes a new technique of aortic banding in mice to induce pressure overload cardiac hypertrophy. For banding, a rubber ring with a fixed inner diameter is used. This new technique promises less variance and more reproducible data for future experiments.
Abstract
Aortic banding in mice is one of the most commonly used experimental models for cardiac pressure overload-induced cardiac hypertrophy and the induction of heart failure. The previously used technique is based on a threaded suture around the aortic arch tied over a blunted 27 G needle to create stenosis. This method depends on the surgeon manually tightening the thread and, thus, leads to high variance in the diameter size. A newly refined method described by Melleby et al. promises less variance and more reproducibility after surgery. The new technique, o-ring- aortic banding (ORAB), uses a non-slip rubber ring instead of a suture with a thread, resulting in reduced variation in pressure overload and reproducible phenotypes of cardiac hypertrophy. During surgery, the o-ring is placed between the brachiocephalic and left carotid arteries. Successful constriction is confirmed by echocardiography. After 1 day, correct placement of the ring results in an increased flow velocity in the transverse aorta over the o-ring-induced stenosis. After 2 weeks, impaired cardiac function is proven by decreased ejection fraction and increased wall thickness. Importantly, besides less variance in the diameter size, ORAB is associated with lower intra- and post-operative mortality rates compared with transverse aortic constriction (TAC). Thus, ORAB represents a superior method to the commonly used TAC surgery, resulting in more reproducible results and a possible reduction in the number of animals needed.
Introduction
While physiological cardiac hypertrophy can be observed during development, exercise, and pregnancy, pathological cardiac hypertrophy responds to hemodynamic stress conditions like arterial hypertension, valvular heart disease, or gene mutations. Initially, the heart undergoes a remodeling characterized by increased cardiomyocyte size and thickening of the ventricular walls to maintain cardiac function1,2. On the other hand, pathological cardiac remodeling is associated with an increased risk for arrhythmia, sudden death, and high mortality. Finally, with time, it results in ventricular dilation, a strong decrease in contractile function, and eventual progression to heart failure (HF), which is associated with high morbidity, mortality, and societal costs3. Therefore, there is an urgent need to understand the molecular background in order to develop new therapeutic strategies4.
Aortic banding is a model that mimics pressure overload-induced left ventricular (LV) hypertrophy and heart failure in mice5. With this method, it is possible to examine the pathomechanisms of pressure overload-induced cardiac remodeling in vivo. The first aortic banding procedure in mice was reported by Rockman et al.6. Pressure overload is induced by a thread suture-based ligation around the aorta (between the brachiocephalic and left common carotid artery). To create a 0.4 mm diameter stenosis, a suture is placed around a 27 G needle and the aorta. After ligation, the needle is removed6,7.
Even though the needle diameter is fixed, the tightness of the thread is highly dependent on the surgeon and, therefore, affects the induced phenotype of cardiac hypertrophy. In addition, in the thread/suture-based method, there is a variable degree of stenosis diameter after surgery, associated with a high variance in mortality8,9. Moreover, training this method is challenging, especially regarding finding the right level and consistency in tightening the thread. Finally, at the beginning of training, high intra- and post-operative mortality due to disruption of the aorta or other tissue injury occurs, as well as high variation in the extent of stenosis in the surviving animals.
Recently, an optimized procedure of aortic banding was described by Melleby et al.10. They presented the ORAB (o-ring aortic banding) method with less variance in stenosis and highly reproducible levels of pressure overload by using a non-slip rubber o-ring with a fixed inner diameter of 0.71 mm, 0.66 mm, and 0.61 mm. In short, the o-ring is cut open, placed around the ascending arch, and closed again by threads. Other scientists using these o-rings reported less variability in the induced cardiac hypertrophy9. They also observed intra- and post-operative mortality, as well as better reproducibility and less variance in the induced hypertrophic phenotype9,11. The present article describes the procedure of this unique strategy in a step-by-step protocol. The expertise shared in this report will help other scientists to improve their techniques in this area.
To induce cardiac hypertrophy resulting in heart failure after 6 weeks, 12-week-old C57BL/6N male mice are recommended for surgery. A comparison 2 weeks after aortic banding between the mouse substrains C57BL/6N and C57BL/6J showed severe cardiac dysfunction and associated increased mortality in C57BL/6N mice. Therefore, these are better suited for models of heart failure12. Twelve-week-old male and female mice have an optimal size for exposure of the aorta and placement of the o-ring with special instruments.
Protocol
The animal experiments were carried out under the principles of the regional committee (Ministerium für Energiewende, Landwirtschaft, Umwelt, Natur und Digitalisierung des Landes Schleswig-Holstein, permission number: V242-21249/2020 [38-4/20]). The mice used for the present study were obtained from a commercial source (see Table of Materials). The animals were kept under standard conditions with a 12 h light, 12 h night cycle; water and food were offered ad libitum.
1. Animal care
- House the mice in specialized cages with bedding, nesting material, a hiding place, and proper access to drinking water and food.
- Keep the animals under continuous specialized veterinary control and treatment.
NOTE: For mice ordered by external suppliers, please assure 7 days of acclimation before starting the procedure.
2. Preparation of the o-ring
NOTE: An o-ring with a fixed diameter of 0.4 mm is recommended to induce cardiac hypertrophy after 2 weeks. The extent and severity of the cardiac phenotype induced depend on the size of the o-ring diameter.
- First, perform one cut of the o-ring (see Table of Materials) under the microscope using scissors or a scalpel to enable the placement around the aorta (Figure 1A, B).
- Pierce each ring side close to the cut with a needle connected with an 8-0 non-absorbable suture and pull the thread. Cut and leave 2-3 cm on one side and 2 cm on the other side to fasten the o-ring around the aorta in the final step (Figure 1C, D).
- Before surgery, take the ligation aid (species instrument, see Table of Materials) and pull the end of the thread (which is kept longer) of one ring side through the hole of the constriction (Figure 1E, F). Put the ligation aid with the attached o-ring aside for placement in the following step (step 6).
- For the disinfection of the ring with the threads, place the ring in an alcohol solution for half an hour. Afterward, lay it on cellulose to dry. Keep the dried ring in a closed tub or case until use. During the surgery, after pulling the thread through the ligation aid, place the ring on a clean surface until use.
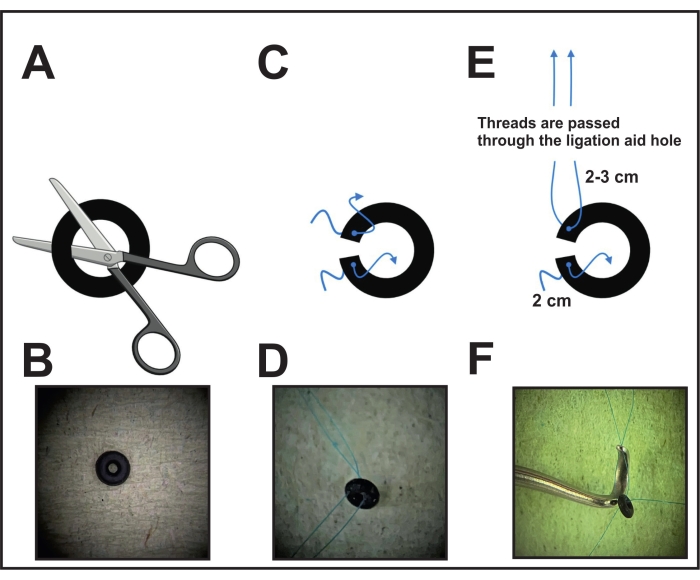
Figure 1: Performing the o-ring preparation for ligation. (A) An o-ring with a fixed diameter is cut with scissors or a scalpel on one side. (B) Image of an o-ring. (C) Each o-ring side is pierced with an 8-0 prolene thread. (D) O-ring pierced with two threads. (E) The threads of one ring side of the o-ring are pulled through the hole of the ligation aid. (F) Final position before placement: the threads of one side are placed through the hole of the ligation aid, while the threads of the other side are kept loose. Please click here to view a larger version of this figure.
3. Premedication of the mice and preparation of the operating field
- To receive sufficient analgesia during the surgery, inject the analgesic buprenorphine (0.1 mg/kg, see Table of Materials) intraperitoneally 20 min before proceeding to the surgery.
NOTE: For the present study, the pain medication was used following the recommendations of the Gesellschaft für Versuchstierkunde/Society of Laboratory Animal Science (GV-SOLAS). - After premedication, anesthetize the mouse in an induction chamber with 2%-4% isoflurane mixed with 0.5-1.0 L/min of 100% O2.
- Shave the fur on the left thorax side of the sedated mouse. After shaving, put the mouse back into the isoflurane-filled chamber and wait for sufficient sedation before intubating the animal.
NOTE: The right time point of the sedation shows slow breathing but avoids snap breathing. Depending on the isoflurane gas setting, it takes 2-3 min to achieve the right level of sedation. - Turn the heating pad on before the surgery to maintain the animal's body temperature (37 °C). Connect the heating pad to a rectal probe (see Table of Materials) to maintain the mouse's body temperature automatically.
4. Intubation of the mice
- Prepare the required instruments (Figure 2A). Disinfect the laryngoscope before the surgery by placing it in alcohol for about 1-3 minutes and then keeping it for drying overnight.
- Stretch a rubber band around the heating pad to fix the mouse with the front teeth on the plate. Place the sedated mouse on the heating pad in a supine position.
- Place the rubber band over the animal's front teeth to extend the neck on the plate.
- Focus a light source on the throat for good visibility of the opening of the trachea for the endotracheal intubation (Figure 2B).
- Open the mouth gently with one hand positioning an intubation aid (handmade laryngoscope, see Table of Materials) (Figure 2A[3]).
- With the other hand and small forceps, gently move the tongue to clear the opening of the trachea.
- Use this hand also to introduce the endotracheal tube into the trachea. On the other hand, still hold the intubation aid. For intubation, use a 22 G cannula (see Table of Materials) (Figure 2A[1]).
- Connect the endotracheal tube position via a three-way stopcock to a ventilator (see Table of Materials) for mice to confirm the correct endotracheal tube position (endotracheal).
- Monitor the right ventilation according to the manufacturer's instructions (tidal volume of 200 µL and respiration rate between 100-150 breaths/min) (Figure 2C).
- Confirm sufficient anesthetic depth by a toe pinch reflex check (no reflex response).
- Turn the anesthesia setting to 2% isoflurane mixed with 0.5-1.0 L/min 100% O2.
- Apply ophthalmic ointment on the eyes to avoid dryness during the surgery.
- Using a cotton swab, disinfect the surgery area 3 times with a commercially available disinfectant solution (see Table of Materials).
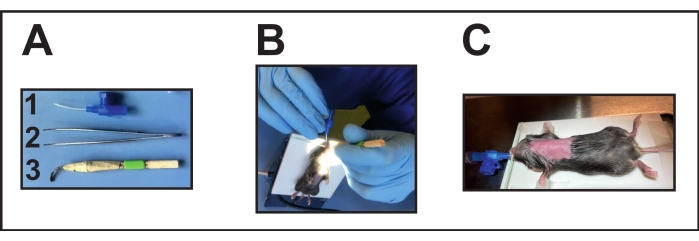
Figure 2: Intubation of the mouse. (A) Intubation instruments: (1) A 22 G i.v. cannula is used as an endotracheal tube (without mandarin); (2) Forceps; (3) Handmade laryngoscope (deformed/flattened cannula glued with wooden sticks and tape). (B) Performing intubation on the positioned heating pad. (C) Intubated mouse connected to a ventilator. Please click here to view a larger version of this figure.
5. Surgery and preparation for ring positioning
NOTE: Use sterile materials and instruments to avoid infections.
- Use scissors to make a 0.5-1 cm long skin incision in the middle of a line between the xyphoid process and left axilla. Use forceps to separate the muscle layer from the underlying ribs and place two retractors (5 mm length, see Table of Materials) into the incision to expose the rib cage.
- To start the left thoracotomy, perform a small incision (~1-2 mm) in the intercostal muscles between the second and third rib using micro spring scissors. Open the thoracic cavity and spread the incision with 45° angled forceps.
- Place three chest retractors (1.0-2.5 mm length) into the incision for opening the thoracic cavity to improve visualization.
- To expose the aortic arch, try to lift up and gently separate the thymus and fat tissue from the arch with fine tip 45° angled forceps.
6. Ligation of the transverse aorta with the o-ring
- Expose the aortic arch with 45° angled forceps in one hand. Position, with the other hand, the o-ring connected with the ligation aid via the threads of one side (step 2).
- Pass the threads using the ligation aid under the aortic arch from the caudal side to the cranial side of the transverse aorta between the brachiocephalic and left common carotid arteries (Figure 3A).
- Take both threads between the ligation aid and the aortic arch with the forceps carefully. Retract and remove the ligation aid and gently position the o-ring around the arch by pulling the threads on each side (Figure 3B).
- After successful positioning, fix the o-ring with the threads and a surgical knot. Make an additional one to avoid opening the knot on each side (Figure 3C).
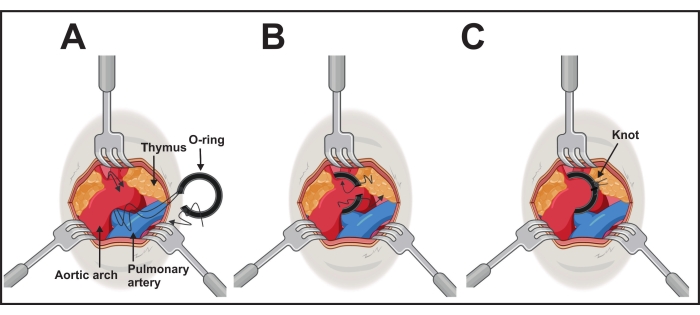
Figure 3: Performing the o-ring implantation. (A) The aortic arch is exposed by three retractors of 1.0-2.5 mm. Both long threads of one ring side are passed under the aorta. (B) The o-ring will be placed by gently pushing the ring and pulling the threads. (C) The is o-ring is in the right position, and one cranial thread is knotted with caudal thread on each side. Please click here to view a larger version of this figure.
7. Suture and post-operative recovery
- Remove the three chest retractors (2.5 mm length) from the incision.
- If needed, eliminate the residual air from the thorax by filling it with a warm 0.9% isotonic salt solution.
- To expose the thorax incision for suture, take two retractors (5 mm length) again to hold the skin on the side.
- Close the thorax with two or three 6-0 non-absorbable sutures (see Table of Materials) and pinch off the outflow of the ventilator for 2 s to reinflate the lungs.
- Remove the two retractors and close the skin with three to five 4-0- absorbable sutures.
- Turn off the isoflurane and monitor. When the animal starts self-breathing, the whiskers are moving, and the toe pinch reflexes can be triggered, extubate the mouse. Lay the mouse on the left side under the heat lamp in its care unit and observe it until it is completely awake.
- Do not leave an animal unattended until it has regained sufficient consciousness.
NOTE: An animal that has undergone surgery should get its own care unit (cage) for better recovery. - Perform pain management with tramadol (1 mg/mL) in drinking water for 7 days and buprenorphine (0.1 mg/kg, 3x daily) by intraperitoneal injection for 3 days after surgery if needed.
NOTE: Follow local animal ethics committee recommendations for postoperative analgesia. - Check medication by weighing the water bottles daily and watch the animal's behavior.
8. Confirmation of successful constriction and right position of the ring
- One day after surgery, verify the stenosis using ultrasound by measuring the maximal flow velocity over the stenosis.
- For measurements, use echocardiography with an ultrasound system and a transducer probe with a frequency of 30 MHz (see Table of Materials).
- As described above, maintain anesthesia using a mask at 1.5%-2% isoflurane with 0.5-1.0 L/min of 100% O2.
- Place the anesthetized animal on the heating pad in a supine position. Connect the heating pad with a rectal probe to maintain the body temperature at 37 °C ± 1 °C, and monitor the heart rate with an ECG using four mouse paw sensors (see Table of Materials).
- For better visualization, use depilation creme.
- A successfully performed ORAB results in an increased flow velocity over the stenosis as measured by ultrasound (~2,400 mm/s) (Figure 4C). For this measurement, position the transducer head parasternally on the right thorax side to localize the aortic arch by two-dimensional (2D) imaging ("B-mode").
- Use the color Doppler to visualize the blood flow in the aorta and measure with the pulsed wave Doppler blood flow velocity over the stenosis.
NOTE: Sham-operated mice (control surgery without a constriction) show a blood flow velocity of ~600-900 mm/s. In addition, a successful ORAB also results in an increased velocity flow ratio between the right carotid (~150 mm/s) (RC, Figure 4A) and the left carotid (~300 mm/s) LC, Figure 4B) in the mouse.
- Use the color Doppler to visualize the blood flow in the aorta and measure with the pulsed wave Doppler blood flow velocity over the stenosis.
- Visualize the right and left arteria carotis interna by two-dimensional (2D) imaging (B-mode). Position the transducer head horizontal on the left and right sides of the neck at a 45° angle and use the pulsed wave Doppler to determine the blood flow velocity.
NOTE: In sham-operated mice, the velocity flow in both arteries is similar.
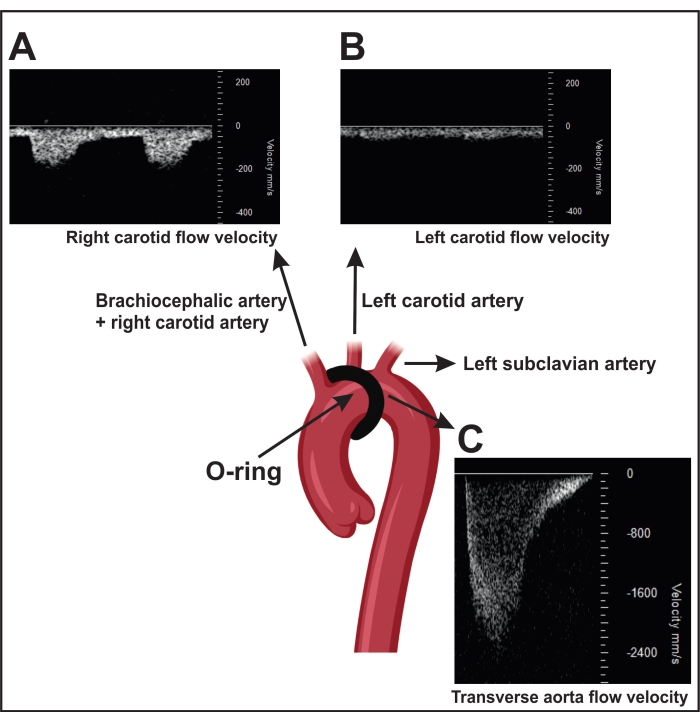
Figure 4: Confirmation of transverse aorta ligation using pulse wave Doppler velocity measurement in the carotid arteries. (A) Representative pulsed wave Doppler velocity signals of the right carotid artery. (B) The stenosis results in a higher flow velocity in the right carotid artery than in the left. (C) The stenosis induced by constriction results in a flow velocity in the descending aorta of more than 2,400 mm/s. Sham mice show a flow velocity of 600-900 mm/s. Please click here to view a larger version of this figure.
Results
Generally, aortic banding mimics human aortic stenosis and induces cardiac hypertrophy in mice. A successful procedure is characterized by heart tissue remodeling reflected by cardiac hypertrophy and reduced heart function5,6.
Directly 1 day after the operation, the effect of o-ring constriction of the transverse aorta can be determined in vivo by echocardiography13. Successful induction of aortic steno...
Discussion
Thread/suture-based aortic banding has been used for many years to induce pressure-overload cardiac hypertrophy in mice. It is an established method to investigate the pathomechanisms of cardiac remodeling and disease progression in vivo. The limitations are the relatively high variance in the degree of stenosis and, consequently, the remodeling. The recently introduced ORAB technique first described by Melleby et al.10 optimizes the conventional method by using a rubber o-ring.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Bundesministerium für Bildung und Forschung (BMBF) to L.L., N.F., and O.J.M. (IVOLADMT-HF; FKZ 01KC2006A).
Materials
| Name | Company | Catalog Number | Comments |
| 1 x long Fixators, 6 cm | 18200-01 | ||
| 2 x Blunt Retractors, 5 mm wide | 18200-11 | ||
| 2 x Short Fixators, 3 cm | 18200-02 | ||
| 2 x fine tip 45° angled forceps | FST (fine sience tools) | 11160-10 | |
| 3 x Blunt Retractors, 2.5 mm wide | 18200-10 | ||
| 3 x Retractor Wires | 18200-05 | ||
| 4-0 absorbable suture (Vicryl) | ETHICON | SABBKLT0 | Used to suture skin |
| 6-0 suture (Prolene), needle size:13 mm | ETHICON | JDP879 | Used to suture chest muscle |
| 8-0 suture (Prolene), needle size: 6.5 mm | ETHICON | RHBECH | Used to fast the Ring |
| Anesthetizing Box, Small | Havard apparatus | 50-0108 | |
| C57BL/6N mice | Charles River | ||
| Fluosorber Activated Charcoal Filter Canister | Havard apparatus | 34-0415 | Used to induce and maintain anesthesia |
| Handmade laryngsopcope | Intubationshelp | ||
| Harvard Apparatus Anesthetic Vaporizer | Havard apparatus | Used to induce and maintain anesthesia | |
| Heating pad + rectal probe (LSI Letica Scientific Instruments:Temperature control unit HB 101/2 ) | Panlab/ Havard apparatus | Used to control and maintain body temperature | |
| i.v. cannula blue 22-gauge (Vasofix Braunüle 0.9 x 25 mm) | B/Braunsharing Expertise | 4268091B | intubation |
| isofluran | Baxter | Anesthesia | |
| Kodan (betadine solution) | Schülke | 20003960-A | Desinfection |
| ligation aid | FST (fine sience tools) | 18062-12 | Used to perform liagtion with O-ring |
| Microscope Lighting: Schott VisiLED Set MC1500/S80-55 (+ controller) | SCHOTT | Ligth | |
| Microscope camera (Leica IC80 HD) | Leica | Used for visualiation operating field | |
| MiniVac Complete Anesthesia Systems for small rodents | Havard apparatus | 75-0233 | Used to induce and maintain anesthesia |
| Mouse Ventilator MiniVent Type 845 | Havard Apparatus | 73-0044 | Used to ventilation during surgery |
| Needle holder | FST (fine sience tools) | TE-10804 | |
| O-ring, non-slip rubber (0.0018 mm x 0.020 mm) | Apple Rubber Products | Liagtion of the aortic arch | |
| Scissors | FST (fine sience tools) | 14040-09 | Used to cut the skin and threads |
| Small Animal Retraction System (Kit for Animals up to 200 g) | FST (fine sience tools) | 18200-20 | |
| Small Base Plate, 20 x 30 cm | 18200-03 | ||
| Table intgerated with heating pad + rectal probe + ECG and transducer tripod | FujiFilm Visual Sonics Imaging System | Echocardiography | |
| Temgesic (Buprenorphin) | Indivior UK Limited | 997.00.00 | Pain pre-medication |
| three-way stop cock (blue) | |||
| Tramal (Tamadol) | Grünental | Pain post-medication | |
| transducer probe MS400 (Visual Sonics) | FujiFilm Visual Sonics Imaging System | Echocardiography | |
| Ultrasound system with cardioligy package | FujiFilm Visual Sonics Imaging System | Echocardiography | |
| Vannas Spring Scissors - 2.5 mm Cutting Edge | FST (fine sience tools) | 15000-08 | Used to cut intercostal chest muscle |
| vet ointment | Bepanten | Used to prevent eyes from drying out |
References
- Frey, N., Olson, E. N. Cardiac hypertrophy: The good, the bad, and the ugly. Annual Review of Physiology. 65, 45-79 (2003).
- Bui, A. L., Horwich, T. B., Fonarow, G. C. Epidemiology and risk profile of heart failure. Nature Reviews Cardiology. 8 (1), 30-41 (2011).
- Bolli, R. New initiatives to improve the rigor and reproducibility of articles published in Circulation Research. Circulation Research. 121 (5), 472-479 (2017).
- Dunlay, S. M., Weston, S. A., Jacobsen, S. J., Roger, V. L. Risk factors for heart failure: A population-based case-control study. The American Journal of Medicine. 122 (11), 1023-1028 (2009).
- Riehle, C., Bauersachs, J. Small animal models of heart failure. Circulation Research. 115 (13), 1838-1849 (2019).
- Rockman, H. A., et al. Segregation of atrial-specific and inducible expression of an atrial natriuretic factor transgene in an in vivo murine model of cardiac hypertrophy. Proceedings of the National Academy of Sciences of the United States of America. 88 (18), 8277-8281 (1991).
- Hu, P., et al. Minimally invasive aortic banding in mice: effects of altered cardiomyocyte insulin signaling during pressure overload. American Journal of Physiology-Heart and Circulatory Physiology. 285 (3), 1261-1269 (2003).
- Mohammed, S. F., et al. Variable phenotype in murine transverse aortic constriction. Cardiovascular Pathology. 21 (3), 188-198 (2012).
- Nakao, Y., et al. O-ring-induced transverse aortic constriction (OTAC) is a new simple method to develop cardiac hypertrophy and heart failure in mice. Scientific Reports. 12, 85 (2022).
- Melleby, A. O., et al. A novel method for high precision aortic constriction that allows for generation of specific cardiac phenotypes in mice. Cardiovascular Research. 114 (12), 1680-1690 (2018).
- Lindsey, M. L., Kassiri, Z., Virag, J. A. I., de Castro Bras, L. E., Scherrer-Crosbie, M. Guidelines for measuring cardiac physiology in mice. American Journal of Physiology-Heart and Circulatory Physiology. 314 (4), 733-752 (2018).
- Garcia-Menendez, L., Karamanlidis, G., Kolwicz, S., Tian, R. Substrain specific response to cardiac pressure overload in C57BL/6 mice. American Journal of Physiology-Heart and Circulatory Physiology. 305 (3), 397-402 (2013).
- deAlmeida, A. C., van Oort, R. J., Wehrens, X. H. Transverse aortic constriction in mice. Journal of Visualized Experiments. (38), e1729 (2010).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved