3.7 : The Equilibrium Binding Constant and Binding Strength
The equilibrium binding constant (Kb) quantifies the strength of a protein-ligand interaction. Kb can be calculated as follows when the reaction is at equilibrium:
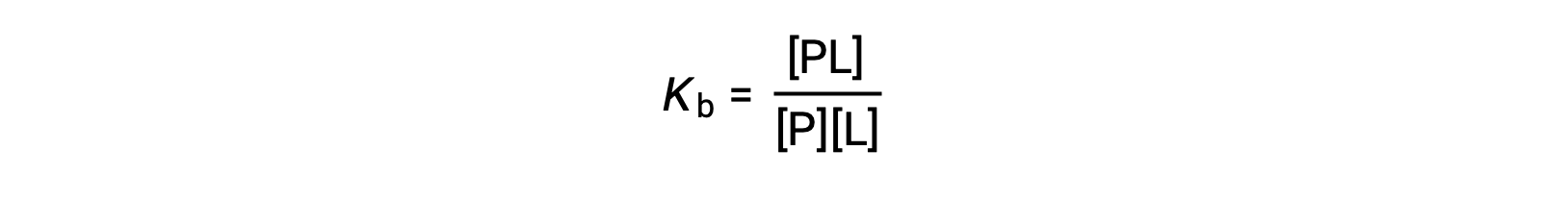
where P and L are the unbound protein and ligand, respectively, and PL is the protein-ligand complex.
As the amount of bound ligand is also related to the rate of ligand binding, experiments can also determine Kb by examining the rates of protein-ligand association (kon) and dissociation (koff) using the following ratio:
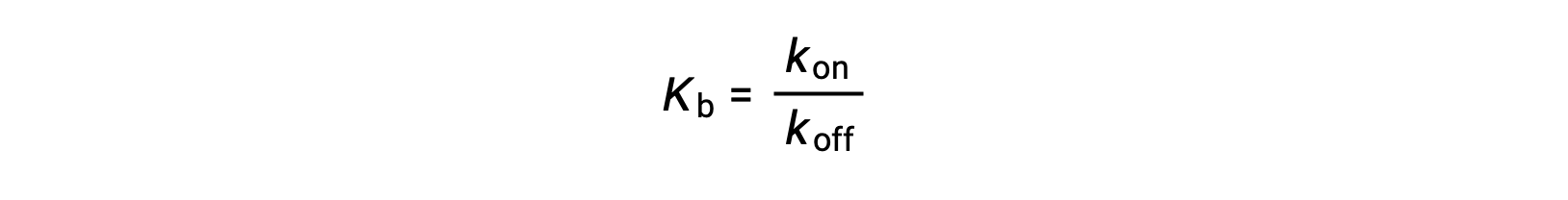
Thus, two categories of binding assays are used to determine the equilibrium binding constant – those that measure equilibrium concentrations and those that measure a reaction’s kinetics. In case, the reaction must be at equilibrium at the time of measurement.
The method of determining the equilibrium concentrations depends on the desired sensitivity and ease of signal detection. For these reasons, spectroscopic assays are most widely used. In these experiments, the reaction produces an absorbance change of a reactant or a product at a given wavelength, detectable by a UV-Vis spectrophotometer. Alternatively, the reactant or product can be tagged with a fluorescent probe or may contain an intrinsic fluorophore. Then, the reaction progress can be measured from the change in fluorescence. These assays are performed by varying one reactant’s concentrations while the rest of the experiment is held constant. The results can then be graphed and analyzed with various curve fitting methods.
Interactions between proteins and ligands are also studied using a variety of biochemical and spectroscopic techniques. Structural analysis, using X-ray crystallography and NMR spectroscopy, aids in predicting protein-ligand interactions through molecular simulations. Theoretical and computational approaches, such as protein-ligand docking studies, are used extensively to characterize the position and interactions of small molecule ligands, including drug candidates. Computer-aided drug design is a fast and low-cost alternative to accelerate the pace of conventional trial and error drug testing.
Dal capitolo 3:

Now Playing
3.7 : The Equilibrium Binding Constant and Binding Strength
Energy and Catalysis
12.7K Visualizzazioni

3.1 : Il Primo F.L. della Termodinamica
Energy and Catalysis
5.4K Visualizzazioni

3.2 : Il secondo principio della termodinamica
Energy and Catalysis
5.0K Visualizzazioni

3.3 : Entalpia all'interno della cellula
Energy and Catalysis
5.7K Visualizzazioni

3.4 : Entropia all'interno della cellula
Energy and Catalysis
10.3K Visualizzazioni

3.5 : Un'introduzione all'energia libera
Energy and Catalysis
8.1K Visualizzazioni

3.6 : Reazioni endergoniche ed esoergoniche in cellula
Energy and Catalysis
14.6K Visualizzazioni

3.8 : Energia libera ed equilibrio
Energy and Catalysis
6.0K Visualizzazioni

3.9 : Non equilibrio nella cellula
Energy and Catalysis
4.1K Visualizzazioni

3.10 : Ossidazione e riduzione di molecole organiche
Energy and Catalysis
6.1K Visualizzazioni

3.11 : Introduzione agli enzimi
Energy and Catalysis
16.9K Visualizzazioni

3.12 : Enzimi ed energia di attivazione
Energy and Catalysis
11.5K Visualizzazioni

3.13 : Introduzione alla cinetica enzimatica
Energy and Catalysis
19.5K Visualizzazioni

3.14 : Numero di fatturato ed efficienza catalitica
Energy and Catalysis
9.8K Visualizzazioni

3.15 : Enzimi cataliticamente perfetti
Energy and Catalysis
3.8K Visualizzazioni
See More