磁気活性化細胞選別(MACS):胸腺Tリンパ球の単離
概要
ソース: ムニエシルヴァン1,2,3, パーチェットティボー1,2,3, ソフィー・ノヴォールト 4, レイチェル・ゴルブ1,2,3
リンパポイシスのための1ユニット、免疫学科、パスツール研究所、パリ、フランス
2 INSERM U1223, パリ, フランス
3ユニバーシテ パリ ディデロ, ソルボンヌ パリ シテ, セルレ パストゥール, パリ, フランス
4フローサイトメトリープラットから, サイトメトリーとバイオマーカー UtechS, 翻訳科学センター, パスツール研究所, パリ, フランス
病原体に対する防御は、免疫系による監視に依存する。このシステムは複雑で、多くのセルタイプで構成され、それぞれが特定の機能を持ちます。この複雑な組成物は、病原体および傷害の大きな多様性に対する免疫応答を可能にする。適応免疫は、特定の病原体に対する特定の応答を可能にします。このタイプの免疫を担う細胞の大半はリンパ球(B細胞およびT細胞)である。通常、B細胞は細胞外感染(細菌感染など)に反応し、T細胞は細胞内感染(ウイルス感染など)に反応する。リンパ球集団における細胞の異なるタイプは、それらが発現する細胞表面タンパク質の組み合わせおよび/または分泌されたサイトカインのパネルによって特徴付けることができる。
磁気ソートは、1つまたは複数の細胞表面タンパク質の磁気特性および発現を使用して標的細胞集団の濃縮を可能にする(1、2)。この手法は、3 つの手順で構成されます。まず、細胞は、1つまたは複数のモノクローナル特異的抗体と結合された磁気ビーズでインキュベートされる。これらの抗体に結合する表面タンパク質を発現する細胞は、磁気ビーズに付着する。次に、標的細胞集団を磁石で捕捉する。最後に、標的細胞は磁石からeledされます。最後に、2つの選別産物が得られ、1つは標識されていない細胞を含み、もう1つは磁気ビーズと結合した標的細胞を含む。カラムは、磁気ソートの効率を向上させるために使用することができます。列では、非磁性要素は、列を通るセルのパスを長くします。したがって、細胞の流れが遅くなり、磁石による細胞捕捉が容易になります。
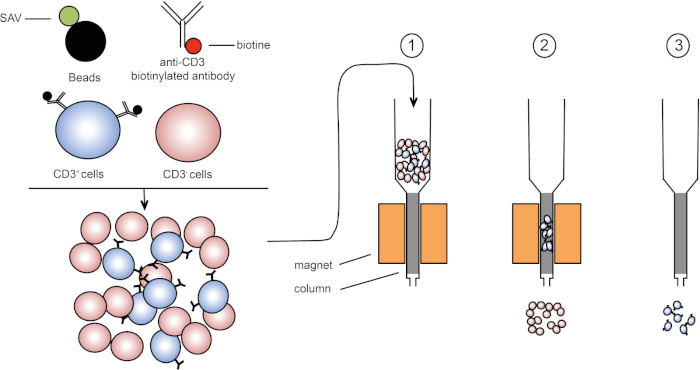
図1:磁気分離の概略図。胸腺白血病は、抗CD3ビオチン化抗体で染色される。洗浄後、ストレプトアビジン(SAV)結合ビーズは、抗CD3抗体上のビオチンを特異的に固定する。(1) セルは列内で転送されます。(2) 磁石は標識されていない細胞を保持せず、CD3陽性細胞はカラムに残ります。最後に、カラムを磁石から分離し、(3)CD3陽性細胞を培地中でelqする。この図のより大きなバージョンを表示するには、ここをクリックしてください。
磁気選別には2種類あります(3)。正の選別では、目的の細胞を磁気ビーズで捕捉します。負の選別では、適切な抗体を運ぶ磁気ビーズで捕捉することにより不要な細胞を除去する。このMACS技術は、標的細胞の良好な濃縮を可能にし、臓器内の1-20%から60-98%に回収された細胞の割合を改善します。ソート後、細胞の純度を確認し、異なる方法(例えばフローサイトメトリー)でソートする必要があります。MACS技術は、細胞培養や細胞周期解析などの他の実験の標的集団を豊かにするのに理想的です。
このラボでは、胸腺白血病を単離し、その後、磁気細胞選別技術を用いて、胸腺CD3陽性細胞をミックスから濃縮する方法を示す。
手順
1. 準備
- 始め始めの前に、実験室の手袋と適切な防護服を着用してください。
- すべての解剖ツールを洗剤で洗い、次に70%のエタノールで洗い、清潔なペーパータオルで乾かします。
- 2%の胎児子牛血清(FCS)を含むハンクのバランス塩溶液(HBSS)の200 mLを調調します。
2. 解剖
- スピーヌ位置の解剖プレートに安楽死マウスをピン留めします。
- はさみと鉗子を使用して、胸腔にアクセスするために縦腹腔切り目を行います。
- 心臓の上に位置する胸腺へのアクセスを得るために心臓を削除します。次に、2つの白い葉で構成され、心臓の上の胸腔に位置する胸腺を識別します。
- 鉗子を使用して慎重に胸腺を取り外し、HBSS 2%FCSの5 mLでペトリ皿の上に置きます。
3. 免疫細胞分離
結果
申請書と概要
参考文献
- Owen, C. S. and Sykes, N. L. Magnetic labeling and cell sorting. Journal of Immunological Methods. 73 (1), 41-48 (1984).
- Miltenyi, S., Müller, W., Weichel, W. and Radbruch, A. High gradient magnetic cell separation with MACS. Cytometry. 11 (2), 231-238 (1990).
- Plouffe, B. D., Murthy, S. K. and Lewis, L. H. Fundamentals and application of magnetic particles in cell isolation and enrichment: a review. Reports on Progress in Physics. 78 (1), (2014).
タグ
スキップ先...
このコレクションのビデオ:

Now Playing
磁気活性化細胞選別(MACS):胸腺Tリンパ球の単離
Immunology
22.9K 閲覧数

フローサイトメトリーと蛍光活性化細胞選別(FACS):脾臓Bリンパ球の単離
Immunology
93.0K 閲覧数

ELISAアッセイ:間接、サンドイッチ、競争力
Immunology
238.5K 閲覧数

ELISPOTアッセイ:IFN-γ分泌脾細胞の検出
Immunology
28.5K 閲覧数

免疫組織化学と免疫細胞化学:光顕微鏡による組織イメージング
Immunology
78.9K 閲覧数

抗体生成:ハイブリドーマを用いたモノクローナル抗体の産生
Immunology
43.6K 閲覧数

免疫蛍光顕微鏡:パラフィン埋め込組織切片の免疫蛍光染色
Immunology
53.9K 閲覧数

共焦点蛍光顕微鏡:マウス線維芽細胞におけるタンパク質の局在を決定する技術
Immunology
43.2K 閲覧数

免疫沈殿ベースの技術:アガロースビーズを用いた内因性タンパク質の精製
Immunology
87.7K 閲覧数

細胞周期分析:CFSE染色と流れサイトメトリーを用いた刺激後のCD4およびCD8 T細胞増殖の評価
Immunology
24.3K 閲覧数

養子細胞移植:宿主マウスへのドナーマウス脾細胞導入とFACSによる成功評価
Immunology
22.3K 閲覧数

細胞死に対するアッセイ:細胞毒性能のクロム放出アッセイ
Immunology
151.4K 閲覧数
Copyright © 2023 MyJoVE Corporation. All rights reserved