Cristalização de Ácido Salicílico via Modificação Química
Visão Geral
Fonte: Kerry M. Dooley e Michael G. Benton, Departamento de Engenharia Química, Louisiana State University, Baton Rouge, LA
O processamento de bioquímicos envolve operações unitárias como cristalização, ultracentrifugação, filtração de membrana e cromatografia preparatória, todas em comum a necessidade de separar grandes de pequenas moléculas, ou sólidas do líquido. Destes, a cristalização é a mais importante do ponto de vista da tonelagem. Por essa razão, é comumente empregado nas indústrias farmacêutica, química e de processamento de alimentos. Exemplos bioquímicos importantes incluem separações quirais,1 purificação de antibióticos,2 separação de aminoácidos de precursores,3 e muitos outros farmacêuticos,4-5 aditivos alimentares,6-7 e purificações agroquímicas. 8 O controle da morfologia cristalina e da distribuição de tamanho é fundamental para a economia do processo, pois esses fatores afetam os custos das operações de processamento a jusante, como secagem, filtração e transporte de sólidos. Para obter mais informações sobre cristalização, consulte um livro didático especializado ou um livro didático de Operações unitárias. 9
A unidade cristalizadora (Figura 1) permite o estudo de: (a) os efeitos dos parâmetros-chave, como taxas de supersaturação e resfriamento/aquecimento, sobre conteúdo de sólidos, morfologia e distribuição do tamanho do cristal; b E o controle on-line dos processos de cristalização. A supersaturação pode ser controlada alterando condições como taxa de agitação e temperatura. As diferentes classificações de cristalização incluem resfriamento, evaporação, balanço de pH e modificação química. Neste experimento, um microscópio offline medirá a partir de cristais que variam em tamanho de 10-1000 μm, uma faixa de tamanho típico para biológicos.

Figura 1: Esquema P&ID (esquerda) e imagem (direita)de Cristalizador. Clique aqui para ver uma versão maior desta figura.
Este experimento demonstrará uma cristalização "modificação química", ou "pH-swing", para gerar cristais de ácido salicílico (SAL) (precursor de aspirina) a partir da reação rápida de soluções aquosas de salicilato básico de sódio (NaSAL), que são ácido básico e sulfúrico (H2SO4) em qualquer lugar entre 40 e 80°C.
Na+SAL + 0,5 H2SO4 SAL (ppt) + Na+ + 0,5 SO42-
O subproduto sulfato de sódio permanece solúvel. O aparelho é composto por dois tanques de ração, três bombas de velocidade variável (peristáltica), o cristalizador (tanque mexido para temperatura e concentração uniforme aproximado, ~5 L), um banho circulante para controle de temperatura, controlador de energia, tanque de produto e um tanque de maquiagem para regeneração de ração com solução NaOH (se desejar). As amostras serão analisadas por um espectrômetro UV-Vis para o íon salicílico solúvel residual, e o produto de cristal ácido salicílico será seco e pesado. Uma sonda de pH pode ser usada para determinar estado estável quando as condições de reação são alteradas.
Procedimento
Soluções orgânicas (saliciilato de sódio, NaSAL) e ácido (ácido sulfúrico, 0,25 M = 0,50 N) serão alimentados com o cristalizador. Certifique-se de usar luvas de látex ao manusear NaSAL, ácido salicílico ou suas soluções, e o ácido sulfúrico de 0,25 M.
Todo o sistema é controlado a partir de um PC usando um controlador distribuído comercial com uma interface semelhante à da Figura 1. Todas as válvulas solenoides on-off ou de 3 vias e os set points do controlador p
Resultados
A Figura 2 apresenta dados representativos que sugerem desvios modestos da distribuição do tamanho de cristal do ideal do MSMPR mesmo em velocidades relativamente altas e baixas concentrações de alimentação.

Figura 2. Distribuição de tamanho de cristal para alimentação NaSAL de 0,16 M, 540 rpm, 60 ° C
Os cristais...
Aplicação e Resumo
Este experimento demonstrou como fazer medições de concentração bruta, fluxo e temperatura e usar a teoria do MSMPR para estimar os parâmetros-chave necessários para projetar um sistema cristalizador grande e complexo. O papel crítico que o tempo de residência desempenha na obtenção de altos rendimentos de cristais e no controle do tamanho médio dos cristais, foi explorado. Muitas vezes há um tempo de residência ideal porque cristais muito grandes raramente são desejáveis. O mesmo vale para a mistura - a m...
Referências
- C. Wibowo, L. O’Young and K.M. Ng, Chem. Eng. Prog., Jan. 2004, pp. 30-39.
- W.J. Genck, Chem. Eng. Prog., Oct. 2004, pp. 26-32.
- S. Takamatsu and D.D.Y. Ryu, Biotechnol. Bioeng., 32, 184-191 (1988).
- F. Wang and K.A. Berglund, Ind. Eng. Chem. Res., 39, 2101-2104 (2000).
- Y. Kim, S. Haam, Y.G. Shul, W.-S. Kim, J.K. Jung, H.-C. Eun and K.-K. Koo, Ind. Eng. Chem. Res., 42, 883-889 (2003).
- K. Hussain, G. Thorsen and D. Malthe-Sorenssen, Chem. Eng. Sci., 56, 2295-2304 (2001).
- H. Gron, A. Borissova and K.J. Roberts, Ind. Eng. Chem. Res., 42, 198-206 (2003).
- F. Lewiner, G. Fevotte, J.P. Klein and F. Puel, Ind. Eng. Chem. Res., 41, 1321-1328 (2002).
- For example: W.L. McCabe, J.C. Smith, and P. Harriott, “Unit Operations of Chemical Engineering”, 7th Ed., McGraw-Hill, New York, 2005, Ch. 27, or C.J. Geankoplis, “Transport Processes and Unit Operations”, 3rd Ed., 1993, Ch. 12.
- P. Barrett, Chem. Eng. Prog., Aug. 2003, pp. 26-32.
- R. Franck, R. David, J. Villermaux and J.P. Klein, Chem. Eng. Sci., 43, 69-77 (1988).
- J. Garside, Chem. Eng. Sci., 40, 3-26 (1985).
- H. Zhao, J.-X. Wang, Qi-An Wang, J.-F. Chen and J. Yun, Ind. Eng. Chem. Res. 46, 8229-8235 (2007).
- J.S. Kwon, M. Nayhouse, G. Orkoulas and P.D. Christofides, Ind. Eng. Chem. Res., 53, 15538-15548 (2014).
Pular para...
Vídeos desta coleção:

Now Playing
Cristalização de Ácido Salicílico via Modificação Química
Chemical Engineering
24.3K Visualizações

Teste de eficiência da transferência de calor de um trocador de calor de tubos aletados
Chemical Engineering
17.9K Visualizações

Uso de um secador de bandeja para investigar a transferência de calor por convecção e condução
Chemical Engineering
43.9K Visualizações

Viscosidade de soluções de propilenoglicol
Chemical Engineering
32.8K Visualizações

Porosimetria de um pó de sílica alumina
Chemical Engineering
9.6K Visualizações

Demonstração do modelo de lei de potência por meio de extrusão
Chemical Engineering
10.1K Visualizações

Absorvedor de gás
Chemical Engineering
36.7K Visualizações

Equilíbrio vapor-líquido
Chemical Engineering
89.1K Visualizações

O efeito da taxa de refluxo na eficiência da destilação em bandejas
Chemical Engineering
77.8K Visualizações

Eficiência da Extração Líquido-Líquido
Chemical Engineering
48.5K Visualizações

Reator de fase líquida: Inversão de sacarose
Chemical Engineering
9.7K Visualizações

Fluxo monofásico e bifásico em um reator de leito compactado
Chemical Engineering
19.0K Visualizações

Cinética da Polimerização por Adição ao Polidimetilsiloxano
Chemical Engineering
16.1K Visualizações

Reator Catalítico: Hidrogenação de etileno
Chemical Engineering
30.4K Visualizações

Avaliação a Transferência de Calor de um Equipamento de Resfriamento e Agitação
Chemical Engineering
7.4K Visualizações
Copyright © 2025 MyJoVE Corporation. Todos os direitos reservados
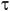 residência) variando as taxas de fluxo volumétrico, mas mantendo a equivalência estequiométrica.
residência) variando as taxas de fluxo volumétrico, mas mantendo a equivalência estequiométrica.