A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Acquisition and Semi-Automated Analysis of Respiratory Muscle Surface Electromyography
In This Article
Summary
Here, we describe a protocol to record and analyze respiratory electromyography (EMG) signals. It includes the anatomic references for placing the EMG electrodes over several respiratory muscles, removing electrocardiographic noise from the EMG signals, and acquiring the EMG root mean square (RMS) and onset timing of activity.
Abstract
Evaluating respiratory drive presents challenges due to the obtrusiveness and impracticality of current methods like functional magnetic resonance imaging (fMRI). Electromyography (EMG) offers a surrogate measure of respiratory drive to the muscles, allowing the determination of both the magnitude and timing of muscle activation. The magnitude reflects the level of muscle activation, while the timing indicates the onset and offset of muscle activity relative to specific events, such as inspiratory flow and activation of other muscles. These metrics are critical for understanding respiratory coordination and control, especially under varying loads or in the presence of respiratory pathophysiology. This study outlines a protocol for acquiring and analyzing respiratory muscle EMG signals in healthy adults and patients with respiratory health conditions. Ethical approval was obtained for the studies, which included participant preparation, electrode placement, signal acquisition, preprocessing, and postprocessing. Key steps involve cleaning the skin, locating muscles via palpation and ultrasound, and applying electrodes to minimize electrocardiography (ECG) contamination. Data is acquired at a high sampling rate and gain, with synchronized ECG and respiratory flow recordings. Preprocessing includes filtering and transforming the EMG signal, while postprocessing involves calculating onset and offset differences relative to the inspiratory flow. Representative data from a healthy male participant performing incremental inspiratory threshold loading (ITL) illustrate the protocol's application. Results showed earlier activation and prolonged duration of extradiaphragmatic muscles under higher loads, correlating with increased EMG magnitude. This protocol facilitates a detailed assessment of respiratory muscle activation, providing insights into both normal and pathophysiologic motor control strategies.
Introduction
Respiratory drive (i.e., the output of respiratory centers to respiratory muscles) is challenging to evaluate due to the obtrusive, often impractical nature of evaluative methods such as functional magnetic resonance imaging (fMRI). Moreover, the small size of the respiratory centers located in the brain stem is difficult to localize and is sensitive to alterations by physiologic noise1,2. Measurements of respiratory drive are important because of their association with important clinical outcomes such as dyspnea, an indication of respiratory distress. Electromyography (EMG) is a surrogate of respiratory drive to the respiratory muscles3. Respiratory muscle EMG allows the determination of muscle activity and its intensity by way of the root mean square (RMS) of the EMG signal. Additionally, the timing of muscle activation can be assessed by identifying the onset and offset of their activity (EMG, onset and EMG, offset, respectively)1,2,3,4,5,6,7,8,9,10,11.
The magnitude of the EMG signal refers to the electrical potential generated by muscle cells when they contract, indicating their level of muscle activation12. The magnitude of the EMG signal can vary depending on factors such as the intensity of muscle contraction, the number of motor units recruited, the electrode placement, the movement of muscle and subcutaneous tissues, and the specific characteristics of the muscle being measured12.
The timing of the EMG signal refers to when the electrical activity occurs relative to a specific event or action (e.g., relative to inspiratory flow for breathing)13. The onset timing indicates when muscle activation begins, while the offset timing indicates when muscle activity decreases, ceases, or is in the relaxation phase13. Timing among the activation of several respiratory muscles will facilitate an understanding of coordination and control mechanisms during breathing. Assessing the consistency or variability of timing patterns over time or in individuals can help identify physiologic and pathophysiologic motor control strategies associated with acute or chronic ventilatory failure.
Both the magnitude and timing of the respiratory muscle EMG have been associated with important clinical outcomes12,13,14. The diaphragm generates the majority of ventilation at rest15. When the respiratory demand increases, such as during exercise or increased inspiratory loading associated with lung diseases (e.g., chronic obstructive pulmonary disease, interstitial lung disease, or acute respiratory distress syndrome), extradiaphragmatic respiratory muscles boost ventilation, which can augment or offset diaphragm contractile requirements15. Thus, in addition to the increasing magnitude of diaphragm EMG, the magnitude of extradiaphragmatic muscle EMG will also increase.
Activation of extradiaphragmatic respiratory muscles can protect the diaphragm from developing fatigue16. However, early activation (onset) and prolonged activation have been associated with acute and chronic ventilatory failure14,17,18. The objective here is to describe a protocol to acquire and analyze both the timing and magnitude of respiratory muscle EMG signals in both healthy adults and patients with suspected or confirmed respiratory pathophysiology. This protocol includes previously validated steps from data acquisition to quantify the timing and magnitude of EMG activity13,19.
Protocol
Studies employing this technique have received ethical approval from the University of Toronto and St. Michael's Hospital located in Toronto, Canada, and the University Hospital Gasthuisberg, Leuven, Belgium. One specific protocol is described here. General discussion about several alternative surface EMG (sEMG) approaches have been proposed for the respiratory muscles and are reported elsewhere12.
1. Participant preparation and placement of sEMG electrodes
- To ensure adequate visualization, ask males not to wear a shirt and females to wear a sports bra or singlet. Use a hospital gown with a front opening to provide adequate access and maintain modesty.
- If the participant has long hair, have it tied back and pinned out of place so the scalene and sternocleidomastoid can be evaluated.
- Position the participant in sitting or half-lying position.
- If excess chest or neck hair is found, shave the area for electrode placement for sEMG.
- To reduce skin impedance, clean the skin of oil and dead skin.
- Do this by rubbing with an alcohol wipe and allowing the alcohol to evaporate (i.e., air dry) before the electrode is applied.
- If the skin is lighter, it may appear slightly red, but more importantly, ensure no obvious dirt, oil, or dry skin is apparent where electrodes will be placed. However, avoid excessive rubbing to prevent skin damage. Avoid application of electrodes to areas of broken skin or other skin lesions.
- Locate muscles of interest by landmarking, palpation and/or ultrasound.
NOTE: Ultrasound may be useful in landmarking the costal diaphragm20. Figure 1 shows examples of locations where electrodes can be placed for sEMG of respiratory muscles. - Place sEMG electrodes on the right side of the thorax, farther from the heart, to decrease the amplitude of the ECG signal and minimize its contamination.
- Apply paired EMG electrodes with a 2 cm inter-electrode distance at the center of the muscle belly along the longitudinal alignment of muscle fibers.
- For the costal diaphragm/intercostals, landmark the anterior axillary line and midclavicular line and place the paired electrodes vertically between these two lines at the level of the seventh or eighth intercostal space.
- For the scalene, landmark the posterior triangle of the neck and place the paired electrodes along the longitudinal axis of the muscle at the level of the cricoid process.
- For the parasternal intercostals, landmark the second intercostal space 1-2 cm lateral to the right side of the sternum and place the paired electrodes along the longitudinal axis of the muscle.
- For the sternocleidomastoid, landmark the suprasternal notch and mastoid process. Accentuate the right sternocleidomastoid muscle belly by placing the operator's hand on the left side of the participant's chin and asking the participant to gently perform isometric left rotation against the hand. Place the paired electrodes at the midpoint of the muscle belly along its longitudinal axis.
- Some EMG systems may require a ground sensor. If required, place the ground sensor on a bony structure close to respiratory muscles (e.g., clavicle, C7 cervical spinous process).
- Attach EMG sensor clips to EMG electrodes. Be sure that wires from EMG sensors from two different muscles (even if wireless) do not overlap and contaminate or provide crosstalk between the two muscles.
NOTE: Wires from the same sensor can overlap, but wires from two different sensors should not. - Apply further fixation of EMG electrodes and sensors by using double-sided tapes that secure the underside of the sensor to the skin.
- Apply medical-grade hypoallergenic tape over the top of the sensors to further secure each sensor to the skin. Avoid applying excessive pressure, and as mentioned above, ensure that the wires from different sensors do not overlap.
2. Signal acquisition
- Select the pre-set template on the data acquisition software and press Open. The template will have the following pre-set parameters: A high pass filter (0.5-20 Hz) in the EMG signal to reduce low-frequency artifacts to facilitate real-time visualization.
- Set the sampling rate of the EMG signal of at least 1 kHz.
- Set the gain of the EMG signal to 1000.
- Set the template to acquire a synchronized recording of ECG and respiratory flow.
- Acquire sEMG and ECG data according to protocol, e.g., during a spontaneous breathing trial in a mechanical ventilation patient.
- After the protocol is complete, stop recording and save the data file.
NOTE: Figure 2 shows screenshots of the software showing applied filtering.
3. Preprocessing after data acquisition
- Open the software and confirm the parameters to be used for analysis of the EMG signal (a bi-directional high pass filter of 5 Hz, the Least Mean Square (LMS) Adaptive Filter to remove ECG contamination, root mean square transform with a moving window for 0.02 s) and press Continue.
- Select the file to be analyzed and press OK.
- Define the time interval to be analyzed (if the total duration of the file is to be analyzed, it will be from 0 s to maximum time), press Select the Range and Continue, and then press Conditioning.
- Press the Analyze button to apply the pre-selected parameters (see step 3.1). Visualize the analyzed EMG signal. Press Rescaled on 1 button to show the EMG signal normalized by its maximum value during the recorded period.
- Press the Continue to Calculate On Off button. Based on the derivative function of the EMG signal it will detect the onset timing of the EMG activity. Press the On and Off button.
- Select the EMG signal from the muscle that needs to be visualized. The visualization can be alternated between muscles to allow the visual inspection of all recorded EMG signals. Press the STOP Looking and Go To Saving button. Press Saving.
- Select the data to be saved. It is possible to reduce the signals before saving (e.g., from 1000Hz to 100Hz). Press Save Processed Data, select the computer folder in which the file is to be saved, and give it a name. Press Save again to confirm.
4. Postprocessing
- Open the saved file using software that provides the ability to compute calculations (e.g., Excel, R, Phyton, Matlab). Determine each breath either by the on and off time of the flow signal and calculate the EMG peak RMS and the EMG mean RMS for each breath.
- For EMG onset, calculate the Absolute difference (in milliseconds) between EMG onset and inspiratory flow onset (INSP, onset):
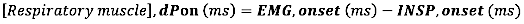
- For EMG offset, calculate the Absolute difference (in milliseconds) between EMG offset and the end of inspiratory flow (INSP,offset)
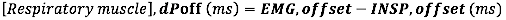
- For EMG onset relative to the duration of inspiratory time, calculate the relative difference (to the duration of Ti) between EMG onset and INSP,onset:

- For EMG offset relative to the duration of inspiratory time, calculate the relative difference (to the duration of Ti) between EMG offset and INSP,offset:

where dP is the time difference between the EMG,onset and inspiratory flow onset (INSP,onset) or between the EMG,offset and inspiratory flow offset (INSP,offset).)
Results
Data is provided for a male participant (22 years old; weight: 100 kg; height: 185 cm; BMI: 29 kg/m2) with normal spirometry and inspiratory muscle strength (FEV1: 4.89 L/s [97% of predicted]; maximal inspiratory pressure: 151 cmH2O [136% of predicted]). He performed an incremental inspiratory threshold loading (ITL) up to task failure using a protocol previously described21,22,23. An overview of...
Discussion
Removal of cardiac activity artifacts from the EMG signal is complex due to their overlapping bandwidth spectrums. The majority of the EMG frequency spectrum is between 20 and 250 Hz, while the ECG frequency spectrum is between 0 Hz and 100 Hz. For some analyses (i.e., timing), it is essential to derive the EMG signal without ECG contamination to achieve accuracy and interpretability of the EMG magnitude and timing. The least mean square (LMS) adaptive filter by utilizing frequencies, is an algorithm that recognizes a pa...
Disclosures
The authors declare they have no conflict of interest to disclose.
Acknowledgements
AR is supported by a Canadian Institutes of Health Research (CIHR) Fellowship (#187900) and UM was funded by Mitacs (IT178-9 -FR101644).
Materials
| Name | Company | Catalog Number | Comments |
| Adjustable table | Amazon | VIVO Electric Height Adjustable 102 cm x 61 cm Stand Up Desk | Enables fine adjustment for trunk and mouthpiece position |
| Air filters | Cardinal | https://cardinalfilters.com/ | |
| Analog output cable | A-Tech Instruments Ltd. | 25 pin D-sub Female to 16xBNC male; 16xRG-174 -16 x 3ft cable | To connect EMG (Noroxan) to data acquisition system (PowerLab) |
| Bioamp for ECG | ADInstruments | ML138 | |
| Desktop or Laptop | N/A | N/A | Capacity for data acquisition system including EMG |
| Double sticks for EMG probes | Noraxon | https://shop.noraxon.com/products/dual-emg-electrodes | |
| Electromyography | Noraxon | Noraxon Ultium Myomuscle with 8 smart leads. https://www.noraxon.com/our-products/ultium-emg/ | |
| EMG electrodes | Duotrode | N/A | |
| Gas analyzer | ADInstruments | ML206 | |
| Gloves | Medline | https://www.medline.com/jump/category/x/cat1790003 | |
| Metricide or protocol to disinfect valves & mouthpieces | Medline | https://www.medline.com/product/MetriCide-28-Disinfectant/Disinfectants/Z05-PF27961?question=metricide | |
| Oximeter pod | ADInstruments | ML320/F | https://www.adinstruments.com/products/oximeter-pods |
| Pneumotach | ADInstruments | MLT3813H-V | https://www.adinstruments.com/products/heated-pneumotach-800-l-heater-controller |
| Powerlab and Labchart Data Acquisition System | ADInstruments, Inc. | https://m-cdn.adinstruments.com/brochures/Research_PowerLab _Brochure_V2-1.pdf | Acquires mouth pressure, ECG, end-tidal CO2, flow (to derive respiratory rate, tidal volume, minute ventilation) and EMG. |
| Pressure transducer with single or dual channel demodulator | Validyne.com | Www.Validyne.Com/Product/Dp45_Low_Pressure_ Variable_Reluctance_Sensor/ | Range depends on population being tested i.e. patients or healthy (Www.Validyne.Com/Product/Cd280_Multi_Channel_Carrier_ Demodulator/; www.Validyne.Com/Product/Cd15_General_Purpose_Basic _Carrier_Demodulator/) |
| Silicone mouthpieces | Hans Rudolph | https://www.rudolphkc.com/ | Small bite size |
| Table model chin rest | Sacor Inc. | Model 600700 | https://sacor.ca/products/head-chin-rest-table-model-with-white-chin-rest-cup |
| Two-way t-piece nonrebreathing valve with sampling port | Hans Rudolph | 1410 Small | |
| Ultrasound | GE Healthcare | Vivid i BT12 Cardiac system with Respiration and 12L-RS Linear Array Transducer | Requires resolution to landmark respiratory muscles including appositional region of diaphragm |
References
- Vaporidi, K., et al. Respiratory drive in critically ill patients. Pathophysiology and clinical implications. Am J Respir Crit Care Med. 201 (1), 20-32 (2020).
- Ciumas, C., Rheims, S., Ryvlin, P. fMRI studies evaluating central respiratory control in humans. Front Neural Circuits. 16, 982963 (2022).
- Domnik, N. J., Walsted, E. S., Langer, D. Clinical utility of measuring inspiratory neural drive during cardiopulmonary exercise testing (CPET). Front Med (Lausanne). 7, 483 (2020).
- Hudson, A. L., et al. Activation of human inspiratory muscles in an upside-down posture. Respir Physiol Neurobiol. 226, 152-159 (2016).
- Hodges, P. W., Gandevia, S. C. Pitfalls of intramuscular electromyographic recordings from the human costal diaphragm. Clin Neurophysiol. 111 (8), 1420-1424 (2000).
- Nguyen, D. a. T., et al. Differential activation of the human costal and crural diaphragm during voluntary and involuntary breaths. J Appl Physiol (1985). 128 (5), 1262-1270 (2020).
- Hudson, A. L., Gandevia, S. C., Butler, J. E. Common rostrocaudal gradient of output from human intercostal motoneurones during voluntary and automatic breathing. Respir Physiol Neurobiol. 175 (1), 20-28 (2011).
- Epiu, I., et al. Inspiratory muscle responses to sudden airway occlusion in chronic obstructive pulmonary disease. J Appl Physiol (1985). 131 (1), 36-44 (2021).
- Sinderby, C., et al. An automated and standardized neural index to quantify patient-ventilator interaction. Crit Care. 17 (5), R239 (2013).
- Estrada, L., Sarlabous, L., Lozano-Garcia, M., Jane, R., Torres, A. Neural offset time evaluation in surface respiratory signals during controlled respiration. 2019, 2344-2347 (2019).
- Luo, Y. M., Moxham, J. Measurement of neural respiratory drive in patients with COPD. Respir Physiol Neurobiol. 146 (2-3), 165-174 (2005).
- Jonkman, A. H., et al. Analysis and applications of respiratory surface EMG: Report of a round table meeting. Crit Care. 28 (1), 2 (2024).
- Rodrigues, A., et al. Semi-automated detection of the timing of respiratory muscle activity: Validation and first application. Front Physiol. 12, 794598 (2021).
- Parthasarathy, S., Jubran, A., Tobin, M. J. Cycling of inspiratory and expiratory muscle groups with the ventilator in airflow limitation. Am J Respir Crit Care Med. 158 (5 Pt 1), 1471-1478 (1998).
- De Troyer, A., Boriek, A. M. Mechanics of the respiratory muscles. Compr Physiol. 1 (3), 1273-1300 (2011).
- Laghi, F., et al. Diaphragmatic neuromechanical coupling and mechanisms of hypercapnia during inspiratory loading. Respir Physiol Neurobiol. 198, 32-41 (2014).
- Parthasarathy, S., Jubran, A., Laghi, F., Tobin, M. J. Sternomastoid, rib cage, and expiratory muscle activity during weaning failure. J Appl Physiol (1985). 103 (1), 140-147 (2007).
- Parthasarathy, S., Jubran, A., Tobin, M. J. Assessment of neural inspiratory time in ventilator-supported patients. Am J Respir Crit Care Med. 162 (2 Pt 1), 546-552 (2000).
- Dacha, S. R. A., Louvaris, Z., Janssens, L., Janssens, W., Gosselink, R., Langer, D. Effects of inspiratory muscle training (IMT) on dyspnea, respiratory muscle function and respiratory muscle activation in patients with COPD during endurance cycling. Eur Respir J. 54 (Suppl 63), PA2199 (2019).
- Bellissimo, C. A., Morris, I. S., Wong, J., Goligher, E. C. Measuring diaphragm thickness and function using point-of-care ultrasound. J Vis Exp. 201, e65431 (2023).
- Basoudan, N., et al. Scalene and sternocleidomastoid activation during normoxic and hypoxic incremental inspiratory loading. Physiol Rep. 8 (14), e14522 (2020).
- Basoudan, N., Shadgan, B., Guenette, J. A., Road, J., Reid, W. D. Effect of acute hypoxia on inspiratory muscle oxygenation during incremental inspiratory loading in healthy adults. Eur J Appl Physiol. 116 (4), 841-850 (2016).
- Melo, L. T., et al. Prefrontal cortex activation during incremental inspiratory loading in healthy participants. Respir Physiol Neurobiol. 296, 103827 (2022).
- Dacha, S., et al. Comparison between manual and (semi-)automated analyses of esophageal diaphragm electromyography during endurance cycling in patients with COPD. Front Physiol. 10, 885 (2019).
- Hermens, H. J., Freriks, B., Disselhorst-Klug, C., Rau, G. Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol. 10 (5), 361-374 (2000).
- Petersen, E., Sauer, J., Graßhoff, J., Rostalski, P. Removing cardiac artifacts from single-channel respiratory electromyograms. IEEE Access. 8, 30905-30917 (2020).
- Hutten, G. J., van Thuijl, H. F., van Bellegem, A. C., van Eykern, L. A., van Aalderen, W. M. A literature review of the methodology of EMG recordings of the diaphragm. J Electromyogr Kinesiol. 20 (2), 185-190 (2010).
- van Leuteren, R. W., Hutten, G. J., de Waal, C. G., Dixon, P., van Kaam, A. H., de Jongh, F. H. Processing transcutaneous electromyography measurements of respiratory muscles, a review of analysis techniques. J Electromyogr Kinesiol. 48, 176-186 (2019).
- Jonkman, A. H., Juffermans, R., Doorduin, J., Heunks, L. M. A., Harlaar, J. Estimated ECG subtraction method for removing ECG artifacts in esophageal recordings of diaphragm EMG. Biomed Signal Process Control. 69, 102861 (2021).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved