A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Spectral Confocal Imaging of Fluorescently tagged Nicotinic Receptors in Knock-in Mice with Chronic Nicotine Administration
In This Article
Summary
We have developed a novel technique of quantifying nicotinic acetylcholine receptor changes within subcellular regions of specific subtypes of CNS neurons to better understand the mechanisms of nicotine addiction by using a combination of approaches including fluorescent protein tagging of the receptor using the knock-in approach and spectral confocal imaging.
Abstract
Ligand-gated ion channels in the central nervous system (CNS) are implicated in numerous conditions with serious medical and social consequences. For instance, addiction to nicotine via tobacco smoking is a leading cause of premature death worldwide (World Health Organization) and is likely caused by an alteration of ion channel distribution in the brain1. Chronic nicotine exposure in both rodents and humans results in increased numbers of nicotinic acetylcholine receptors (nAChRs) in brain tissue1-3. Similarly, alterations in the glutamatergic GluN1 or GluA1 channels have been implicated in triggering sensitization to other addictive drugs such as cocaine, amphetamines and opiates4-6.
Consequently, the ability to map and quantify distribution and expression patterns of specific ion channels is critically important to understanding the mechanisms of addiction. The study of brain region-specific effects of individual drugs was advanced by the advent of techniques such as radioactive ligands. However, the low spatial resolution of radioactive ligand binding prevents the ability to quantify ligand-gated ion channels in specific subtypes of neurons.
Genetically encoded fluorescent reporters, such as green fluorescent protein (GFP) and its many color variants, have revolutionized the field of biology7.By genetically tagging a fluorescent reporter to an endogenous protein one can visualize proteins in vivo7-10. One advantage of fluorescently tagging proteins with a probe is the elimination of antibody use, which have issues of nonspecificity and accessibility to the target protein. We have used this strategy to fluorescently label nAChRs, which enabled the study of receptor assembly using Förster Resonance Energy Transfer (FRET) in transfected cultured cells11.More recently, we have used the knock-in approach to engineer mice with yellow fluorescent protein tagged α4 nAChR subunits (α4YFP), enabling precise quantification of the receptor ex vivo at submicrometer resolution in CNS neurons via spectral confocal microscopy12. The targeted fluorescent knock-in mutation is incorporated in the endogenous locus and under control of its native promoter, producing normal levels of expression and regulation of the receptor when compared to untagged receptors in wildtype mice. This knock-in approach can be extended to fluorescently tag other ion channels and offers a powerful approach of visualizing and quantifying receptors in the CNS.
In this paper we describe a methodology to quantify changes in nAChR expression in specific CNS neurons after exposure to chronic nicotine. Our methods include mini-osmotic pump implantation, intracardiac perfusion fixation, imaging and analysis of fluorescently tagged nicotinic receptor subunits from α4YFP knock-in mice (Fig. 1). We have optimized the fixation technique to minimize autofluorescence from fixed brain tissue.We describe in detail our imaging methodology using a spectral confocal microscope in conjunction with a linear spectral unmixing algorithm to subtract autofluoresent signal in order to accurately obtain α4YFP fluorescence signal. Finally, we show results of chronic nicotine-induced upregulation of α4YFP receptors in the medial perforant path of the hippocampus.
Protocol
1. Pump implantation
- Before pump implantation, fill and prepare the Alzet mini-osmotic pumps (Alzet, Model 2002, Cupertino, USA) being careful not to introduce air bubbles. This model of mini-osmotic pump delivers solution at a rate of 0.5 μl/hr for 14 days. Ensure sterile conditions. Weigh empty and filled pumps. At the conclusion of experiment (10 days after implantation), the remaining liquid in the pump can be removed with a syringe and needle and weighed to calculate the volume pumped.
- Pumps with control solution contain saline (0.9% w/v, Teknova, S5819, Hollister, USA). To prepare nicotine solution, a 1 M stock solution of (-)-nicotine hydrogen tartrate salt (Sigma, cat# N5260) is diluted in saline (0.9% w/v) and sterilized by filtering through a 0.22 μm syringe-end filter. We have previously administered nicotine at 0.4 and 2 mg/kg/hr (calculated as free base of nicotine) for 10 days.
- Prepare three saline baths (three 10 cm saline filled petri dishes). Once the pump has been filled and capped, wash pump thoroughly in each successive saline bath to remove any traces of the drug on the outer shell. Immerse filled pumps in saline solution for storage until surgery, keeping the control and nicotine pumps in separate saline containers.
- To encourage a healthy recovery and reduce the risk of post-surgical infection, have clean cages prepared for long-term recovery.
- For short-term recovery immediately following surgery, prepare a cage containing a heating pad and a heat lamp.
- To examine chronic nicotine effects we have 5 to 6 homozygous α4YFP knock-in mice (2-3 months old) in each group (control or nicotine). The α4YFP knock-in mouse line have been backcrossed for 10 generations to the C57BL/6J mouse strain. We make sure the age and sex of all mice are the same for the study and that all surgeries are performed on the same day to minimize variability. To prevent hypothermia to the mice during and after surgery, equip surgical table with a heating pad covered in sterile surgical drape.
- Induce the α4YFP knock-in mouse with 3 L/min oxygen and 3% isoflurane and then maintain anesthesia at 2.5 L/min oxygen and 1% isoflurane. We prefer isoflurane anesthesia because on completion of the surgery isoflurane clears the system rapidly and the mice are conscious and mobile within ~2 min. Apply eye drops immediately (Tear-Gel, Novartis) to avoid corneal damage.
- Pumps are implanted subcutaneously through a skin incision on the back of the neck and the pump is pushed caudally down the dorsal aspect of the back. Wipe the area on the back between the forelimbs with 95% ethanol to matte the fur. Pinch the skin with 0.8mm Graefe forceps (Fine Science Tools, 11050-10) and make a 1 cm central lateral cut using iris scissors (Fine Science Tools, 14060-10).
- To create a subcutaneous space for the pump, insert standard pattern scissors (Fine Science Tools, 14101-14) into the incision and push them carefully toward the caudal end of the animal.
- Using 1.0 mm Graefe forceps (Fine Science Tools, 11650-10), remove the pump from saline. Hold the incision open using forceps and insert the pump into the incision with the osmotic pump cap facing the caudal end of the animal and push the pump to the caudal end.
- Pinch wound shut using 0.8mm forceps and apply an adequate amount of Vetbond (3M, cat# 1469SB) glue. Hold until wound is sealed.
- Remove the animal from isoflurane mask, inject with the analgesic, meloxicam (0.1 mg/kg s.c.), and place into a short-term recovery cage until conscious and mobile. Then place in a long-term recovery cage with food and water available ad libitum.
2. α4YFP knock-in mouse fixation by intracardiac perfusion
- Make solutions one day prior to procedure and leave at 4° C. To minimize variability all mice will be perfused on the same day and with the same batch of solutions.
- Perform perfusion fixations in a well ventilated area. Flush line of peristatic pump (Masterflex Easy Load, 7518-00; Masterflex Pump Controller, 60648) with ddH2O.
- Add ~0.0015g of heparin (Sigma, cat# H4784), an anticoagulant, to 20 ml PBS pH 7.6 (Invitrogen, cat# 70011).
- Anesthetize α4YFP knock-in mouse by injecting intramuscularly a mixture of ketamine (25 mg/kg, Wyeth Animal Health) and medatomidine hydrochloride (0.25 mg/kg, Pfizer) into the hind leg muscle and immediately place the animal back into its home cage.
- Pin animal to a styrofoam lid inserted into a metal tray. Wipe thorax with 95% ethanol. Pinch skin with adson forceps (Fine Science Tools, 91106-12) and snip open the thoracic cavity with iris scissors. Clamp the ribcage using an ultra fine hemostat (Fine Science Tools, 13021-12) and expose the heart.
- Begin pumping PBS at 4 ml/min and insert a 23G butterfly needle (Becton Dickinson, 367253) into the left ventricle of the animal. Immediately snip the right atrium to allow blood and perfusate to escape.
- Perfuse 20 ml of PBS (pH 7.6), then 30 ml of 4% paraformaldehyde (pH 7.6, diluted with PBS from a 16% PFA stock, Electron Microscopy Sciences, cat# 15710), then 20ml of 5% sucrose (pH 7.6) . We found that over fixation increases autofluorescence. Perfusion with 5% sucrose rinses residual PFA from the brain, which lowers autofluorescence.
- Remove brain and store in 30% sucrose for 3 days.
- To freeze brains for coronal sectioning, cut off the cerebellum with a razor blade and place brain in a plastic embedding mold (VWR, cat# 18986-1) rostral side up and submerge in O.C.T. Mounting Compound (Tissue-Tek, cat# 4583). Freeze in dry ice and store at -20 °C before slicing.
- Section brains (30 μm thick) on a cryostat and then transfer to coated slides. Store the brain sections at -20°C in slide boxes containing one anhydrous calcium sulphate stone. The slide box should be in a zip lock sealed bag to avoid moisture and subsequent freezer burn when stored at -20°C.
3. Imaging fluorescent nAChRs using spectral confocal microscopy
- Ensure slides have minimal exposure to any light source to minimize photobleaching.
- Coverslip the brain section slides with a mounting medium that does not inhibit fluorescent protein fluorescence. We have good success with Mowiol 4-88 (pH 8.5 in Tris-HCl and glycerol, EMD-Calbiochem, cat# 475904), which hardens after a few hours. Make sure the Mowiol equilibrates to room temperature before cover slipping in order to avoid air bubbles. Do not use nail polish when cover slipping as it will quench YFP fluorescence.
- Images are acquired using a Nikon C1si spectral confocal microscope system. Details on the method of spectral confocal imaging and linear unmixing are provided elsewhere 13-15. The rationale for using a spectral confocal microscope is that fixed brain tissue has inherent autofluorescence. Spectral confocal imaging on the Nikon C1si uses an array of 32 photomultiplier tube detectors that sample a specified range of wavelengths of fluorescent emitted light, refracted spatially into its different wavelengths via a grating dispersive element just like a prism refracting white light into a rainbow of colors16. What is collected is a lambda stack of images -- images collected at different wavelengths of light so that an emission spectrum is collected for each pixel of a lambda stack of images. Since YFP and tissue autofluorescence each have characteristic spectral signatures, the lambda stack can be deconvolved using a linear unmixing algebraic algorithm into separate YFP and autofluorescent signals (Fig. 2). Thus, very accurate quantification of YFP fluorescence can be determined even in tissue with significant levels of autofluorescence.
- Various settings can be adjusted to optimize image quality and fluorescence collection efficiency. We will report settings we typically use but these settings can be adjusted depending on the sample and the confocal microscope. To accurately quantify changes in α4YFP subunit expression with chronic nicotine we ensure that the grey scale level intensity of the signals of all pixels are below saturating value (< 4095 for 12 bit grey scale). Moreover, we accommodate for potential increase in signal due to receptor upregulation by adjusting the confocal settings so that pixels are around one third of saturating value (~1300-1400) or less. Once the settings are optimized, the settings are maintained identical for all imaging sessions and samples.
- We use the following settings for imaging α4YFP using a 60X oil CFI Plan Apo VC objective (1.40 N.A., 0.13 mm working distance): 488 nm laser line at 15% maximal transmission of a 40 mW Argon laser, spectral detector gain at 220, spectral range imaged from 496.5 nm to 656.5 nm at 5 nm resolution, 512 x 512 pixels over a 50 μm x 50 μm area, a medium pinhole size (60 μm diam), a 4.08 pixel dwell time, averaging over two scans and 12 bits of grey scale.
4. Linear unmixing of spectral confocal images and image analysis
- To perform linear spectral unmixing on a sample image taken with a spectral confocal microscope, one must first acquire a reference spectrum for YFP and a reference spectrum for tissue autofluorescence.
- An important requisite for any reference spectrum is to obtain a high signal to noise fluorescence spectrum imaged with the same laser line used for imaging of your samples. Although the 514 nm laser line of Argon has a higher excitation efficiency to excite YFP we prefer to image YFP with the 488 nm laser line because there is better separation from the peak emission of YFP and the 488 nm line is less likely to distort the peak and one can obtain the entire YFP emission spectrum including the rise to peak. We image soluble YFP transfected in a cell line so that a strong YFP signal is obtained and the resultant YFP spectrum is stored in our library of reference spectra. Transfected α4YFP over expressed in cell lines may also be used since it will also provide a high signal to background spectrum.
- To obtain a reference spectrum of autofluorescence we image autofluorescence from a wildtype mouse brain section from the various brain regions that we intend to image from the α4YFP mouse and obtain their corresponding spectra. We use the same laser line, 488 nm, and image using nearly identical parameters, although we may alter the detector gain, laser intensity or perform average imaging to maximize signal to noise.
- After obtaining a spectral confocal image from a brain region of the α4YFP mouse, the image is then deconvolved into its YFP and autofluorescence signals by applying a linear spectral unmixing algorithm using the reference spectrum of YFP and the reference spectrum of wildtype mouse autofluorescence from the same brain region.
- The unmixed α4YFP image can then be opened in an image analysis software such as ImageJ (http://rsbweb.nih.gov/ij/) and then the mean pixel intensity is calculated for the region of interest. A plugin for ImageJ called "loci_tools.jar" (http://www.loci.wisc.edu/bio-formats/imagej) can be used to import Nikon ics/ids confocal files.
- We repeat the same linear spectral unmixing and analysis to wildtype mice in the same brain region to obtain a background residual value.
- Then one obtains the corrected mean α4YFP intensity by subtracting the background residual value (4.6) from the mean uncorrected α4YFP value (4.5).
5. Representative Results
We show a representative true color projection of a lambda stack of images of the medial habenula from a homozygous α4YFP mouse (Fig. 3A) taken with a spectral confocal microscope. We also show the emission spectrum from a region of interest containing α4YFP positive neurons from the same lambda stack image (Fig. 3B). A distinct emission peak is evident at ~527 nm, which is the peak fluorescence emission of YFP. The region neighbouring the medial habenula shows an emission spectrum lacking a spectral peak at 527 nm, indicating the absence of α4YFP nAChR subunits. Following linear spectral unmixing using reference spectra of YFP and mouse brain autofluoresence with significant overlap of emission (Fig. 4), separation of YFP and autofluorescent signal is possible yielding an α4YFP unmixed image, an autofluorescent unmixed image and a remainder channel. Clear localization of α4YFP fluorescence can be identified in the tightly packed soma of the medial habenula (Fig. 4).
In the hippocampus α4YFP is localized mainly in the medial perforant path, the tempero-ammonic path and the alveus12. These are all glutamatergic innervation of the hippocampus. We examined the effects of chronic nicotine on α4YFP expression in the hippocampal perforant path (Fig. 5). Chronic administration of nicotine (2 mg/kg/hr for 10 days) resulted in a significant increase (69 ± 14%) in α4YFP fluorescence from control saline treated mice to chronic nicotine treated mice (p = 0.001, Wilcoxon rank sum test) (Fig. 5).
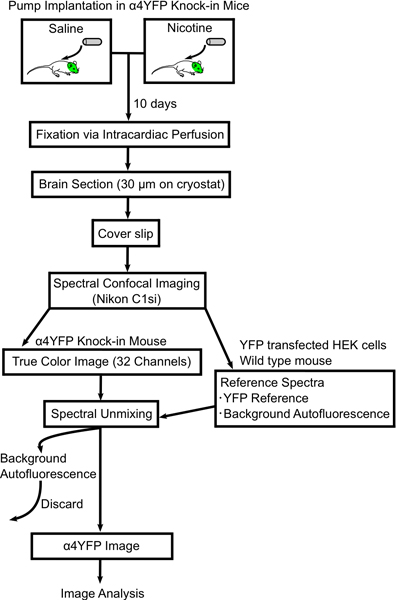
Figure 1. Flow chart showing procedure to image changes in α4YFP nAChR subunits with chronic nicotine. Mini-osmotic pumps are filled with either saline or nicotine and implanted subcutaneously in α4YFP homozygous mice. After 10 days mice are perfused and fixed with 4% paraformaldehyde and the mouse brains are sectioned (30 μm thick) on slides. The brain section is imaged on a spectral confocal microscope (Nikon C1si) and spectrally unmixed into YFP and autofluorescent images. Then the α4YFP images are analyzed further with ImageJ software.
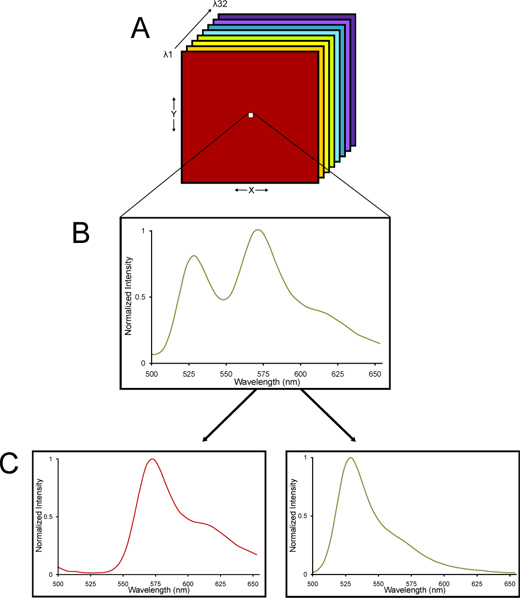
Figure 2. A schematic diagram of a lambda stack imaged from a spectral confocal microscope and linearly unmixed into its spectral components. (A) A lambda stack of images is collected. (B) A lambda stack consists of images collected at different wavelengths of light so that an emission spectrum is collected for each pixel across the entire stack. (C) Since YFP and tissue autofluorescence each have characteristic spectral signatures, the lambda stack can be deconvolved using a linear unmixing algebraic algorithm into separate YFP and autofluorescent signals. Thus, very accurate quantification of YFP fluorescence can be determined even in tissue with high levels of autofluorescence.

Figure 3. Spectral confocal image of a brain region expressing α4YFP nAChRs. (A) A true color projection of a lambda stack of images of the medial habenula from an α4YFP mouse taken with a Nikon C1si spectral confocal microscope. (B) Plots of spectra from a region of interest, which includes α4YFP containing neurons (green), and a region of interest outside of the medial habenula (red).
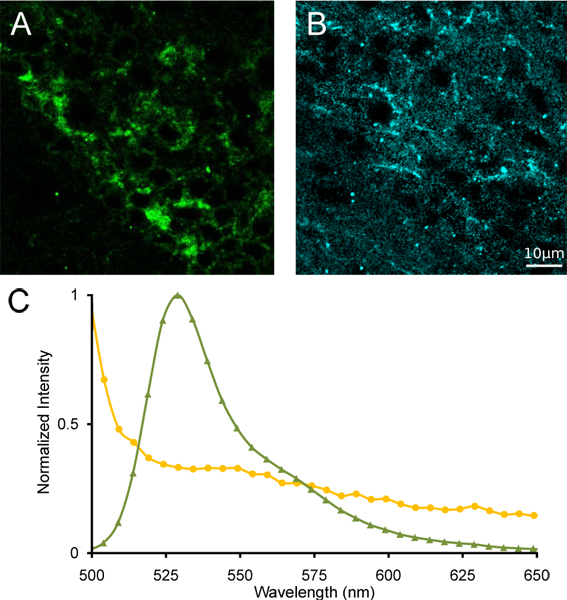
Figure 4. Linear spectral unmixing of the medial habenula. (A) Images of unmixed α4YFP and (B) autofluorescence following linear spectral unmixing. (C) Reference spectra of YFP (green triangles) and autofluorescence (yellow circles) used for spectral unmixing.

Figure 5. Upregulation of α4 nAChRs in the hippocampus of α4YFP knock-in mice exposed to chronic nicotine. (A) A tiled montage of α4YFP fluorescence from the hippocampus. The two dashed selection areas are the approximate locations on the inferior limb of the perforant path of the hippocampus where analyses were performed for each mouse. (B) α4YFP fluorescence was significantly higher in the perforant path of mice exposed to chronic nicotine than chronic saline (*, p = 0.001, Wilcoxon rank sum test). Results represent mean ± SEM from n = 20 measurements for both saline and chronic nicotine treated mice (5 mice for each treatment group).

Figure 6. Better depth expression of α4YFP as compared to antibody labeling. X-Z orthogonal views of α4YFP fluorescence (A) and VGlut2 antibody with Cy5 as a secondary label (B). (C) Plots showing greater fluorescence signal intensity degradation over depth for antibody staining (black squares) as compared to α4YFP (open circles).
Discussion
The use of a fluorescent receptor in a knock-in mouse model to determine quantity and localization of a specific ion channel provides a number of advantages. In contrast to proteins such as actin, which is ubiquitously expressed in all cells, ion channels are present in far fewer numbers and their expression varies between neuronal subtypes making accurate analysis via traditional immunohistochemical techniques challenging. The α4YFP gene product is expressed at WT levels, being under control of the same promoter...
Disclosures
We have nothing to disclose.
Acknowledgements
Anthony Renda was supported by a University of Victoria Graduate Fellowship Award. This research was supported by a Natural Sciences and Engineering Research Council of Canada Discovery Grant, a NARSAD Young investigator Award (to R.N.), a Victoria Foundation - Myre and Winifred Sim Fund, a Canadian Foundation for Innovation grant, a British Columbia Knowledge Development Fund and a Natural Sciences and Engineering Research Council of Canada Research Tools and Instrumentation Grant. We thank Jillian McKay, Christina Barnes, Ariel Sullivan, Jennifer MacDonald and Daniel Morgado for excellent mouse husbandry.
Materials
| Name | Company | Catalog Number | Comments |
| mini-osmotic pumps |  Alzet Alzet | model 2002 | |
| saline | TEKnova, Inc. | S5819 | |
| (-)-nicotine hydrogen tartrate salt | Sigma-Aldrich | N5260 | |
| eye drops | Novartis AG | Tear-Gel | |
| Vetbond glue | 3M | 1469SB | |
| heparin sodium salt | Sigma-Aldrich | H4784 | |
| 10x PBS | Invitrogen | 70011 | |
| ketamine | Wyeth Animal Health | 0856-4403-01 | |
| medatomidine hydrochloride | Pfizer Pharma GmbH | 1950673 | |
| 23G butterfly needle | BD Biosciences | 367253 | |
| paraformaldehyde | Electron Microscopy Sciences | 15710 | |
| plastic embedding mold | VWR international | 18986-1 | |
| O.C.T. Mounting Compound | Tissue-Tek | 4583 | |
| Mowiol 4-88 | EMD Millipore | 475904 | pH 8.5 |
References
- Perry, D. C., Davila-Garcia, M. I., Stockmeier, C. A., Kellar, K. J. Increased nicotinic receptors in brains from smokers: membrane binding and autoradiography studies. J. Pharmacol. Exp. Ther. 289, 1545-1552 (1999).
- Schwartz, R. D., Kellar, K. J. Nicotinic cholinergic receptor binding sites in the brain: regulation in vivo. Science. 220, 214-216 (1983).
- Marks, M. J., Burch, J. B., Collins, A. C. Effects of chronic nicotine infusion on tolerance development and nicotinic receptors. J. Pharmacol. Exp. Ther. 226, 817-8125 (1983).
- Carlezon, W. A. J., Nestler, E. J. Elevated levels of GluR1 in the midbrain: a trigger for sensitization to drugs of abuse. Trends Neurosci. 25, 610-615 (2002).
- Fitzgerald, L. W., Ortiz, J., Hamedani, A. G., Nestler, E. J. Drugs of abuse and stress increase the expression of GluR1 and NMDAR1 glutamate receptor subunits in the rat ventral tegmental area: common adaptations among cross-sensitizing agents. J. Neurosci. 16, 274-2782 (1996).
- Saal, D., Dong, Y., Bonci, A., Malenka, R. C. Drugs of abuse and stress trigger a common synaptic adaptation in dopamine neurons. Neuron. 37, 577-5782 (2003).
- Tsien, R. Y. The green fluorescent protein. Annu. Rev. Biochem. 67, 509-544 (1998).
- Chalfie, M., Tu, Y., Euskirchen, G., Ward, W. W., Prasher, D. C. Green fluorescent protein as a marker for gene expression. Science. 263, 802-805 (1994).
- Feng, G., Mellor, R. H., Bernstein, M., Keller-Peck, C., Nguyen, Q. T., Wallace, M. Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron. 28, 41-51 (2000).
- Livet, J., Weissman, T. A., Kang, H., Draft, R. W., Lu, J., Bennis, R. A. Transgenic strategies for combinatorial expression of fluorescent proteins in the nervous system. Nature. 450, 56-62 (2007).
- Nashmi, R., Dickinson, M. E., McKinney, S., Jareb, M., Labarca, C., Fraser, S. E. Assembly of α4β2 nicotinic acetylcholine receptors assessed with functional fluorescently labeled subunits: effects of localization, trafficking, and nicotine-induced upregulation in clonal mammalian cells and in cultured midbrain neurons. J. Neurosci. 23, 11554-11567 (2003).
- Nashmi, R., Xiao, C., Deshpande, P., McKinney, S., Grady, S. R., Whiteaker, P. Chronic nicotine cell specifically upregulates functional α4* nicotinic receptors: basis for both tolerance in midbrain and enhanced long-term potentiation in perforant path. J. Neurosci. 27, 8202-8218 (2007).
- Dickinson, M. E., Bearman, G., Tilie, S., Lansford, R., Fraser, S. E. Multi-spectral imaging and linear unmixing add a whole new dimension to laser scanning fluorescence microscopy. BioTechniques. 31, 1272-1278 (2001).
- Nashmi, R., Fraser, S. E., Lester, H., Dickinson, M. E., Periasamy, A., Day, R. N. . Molecular imaging: fret microscopy and spectroscopy. , 180-192 (2005).
- Zimmermann, T., Rietdorf, J., Girod, A., Georget, V., Pepperkok, R. Spectral imaging and linear un-mixing enables improved FRET efficiency with a novel GFP2-YFP FRET pair. FEBS Lett. 531, 245-249 (2002).
- Larson, J. M. The Nikon C1si combines high spectral resolution, high sensitivity, and high acquisition speed. Cytometry A. 69, 825-8234 (2006).
- Melvin, N. R., Sutherland, R. J. Quantitative caveats of standard immunohistochemical procedures: implications for optical disector-based designs. J. Histochem. Cytochem. 58, 577-5784 (2010).
- Jones, I. W., Wonnacott, S. Why doesn't nicotinic ACh receptor immunoreactivity knock out. Trends Neurosci. 28, 343-345 (2005).
- Moser, N., Mechawar, N., Jones, I., Gochberg-Sarver, A., Orr-Urtreger, A., Plomann, M. Evaluating the suitability of nicotinic acetylcholine receptor antibodies for standard immunodetection procedures. J. Neurochem. , (2007).
- Whiteaker, P., Cooper, J. F., Salminen, O., Marks, M. J., McClure-Begley, T. D., Brown, R. W., Collins, A. C., Lindstrom, J. M. Immunolabeling demonstrates the interdependence of mouse brain a4 and b2 nicotinic acetylcholine receptor subunit expression. The Journal of Comparative Neurology. 499, 1016-1038 (2006).
- Marks, M. J., McClure-Begley, T. D., Whiteaker, P., Salminen, O., Brown, R. W. B., Cooper, J., Collins, A. C., Lindstrom, J. M. Increased nicotinic acetylcholine receptor protein underlies chronic nicotine-induced up-regulation of nicotinic agonist binding sites in mouse brain. The Journal of Pharmacology and Experimental Therapeutics. , 337-3187 (2011).
- Marks, M. J., Rowell, P. P., Cao, J. Z., Grady, S. R., McCallum, S. E., Collins, A. C. Subsets of acetylcholine-stimulated 86[Rb]+ efflux and 125[I]-epibatidine binding sites in C57BL/6 mouse brain are differentially affected by chronic nicotine treatment. Neuropharmacology. 46, 1141-1157 (2004).
- King, S. L., Caldarone, B. J., Picciotto, M. R. Beta2-subunit-containing nicotinic acetylcholine receptors are critical for dopamine-dependent locomotor activation following repeated nicotine administration. Neuropharmacology. 47, 132-139 (2004).
- Robinson, S. F., Marks, M. J., Collins, A. C. Inbred mouse strains vary in oral self-selection of nicotine. Psychopharmacology (Berl). 124, 332-339 (1996).
- Sparks, J. A., Pauly, J. R. Effects of continuous oral nicotine administration on brain nicotinic receptors and responsiveness to nicotine in C57Bl/6 mice. Psychopharmacology (Berl). , 141-145 (1999).
- Rahman, S., Zhang, J., Engleman, E. A., Corrigall, W. A. Neuroadaptive changes in the mesoaccumbens dopamine system after chronic nicotine self-administration: a microdialysis study. Neuroscience. 129, 415-4124 (2004).
- Picciotto, M. R., Zoli, M., Rimondini, R., Lena, C., Marubio, L. M., Pich, E. M. Acetylcholine receptors containing the β2 subunit are involved in the reinforcing properties of nicotine. Nature. 391, 173-177 (1998).
- Fowler, C. D., Lu, Q., Johnson, P. M., Marks, M. J., Kenny, P. J. Habenular α5 nicotinic receptor subunit signalling controls nicotine intake. Nature. 471, 597-601 (2011).
- Maskos, U., Molles, B. E., Pons, S., Besson, M., Guiard, B. P., Guilloux, J. P. Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature. 436, 103-107 (2005).
- Matta, S. G., Balfour, D. J., Benowitz, N. L., Boyd, R. T., Buccafusco, J. J., Caggiula, A. R., Craig, C. R., Collins, A. C., Damaj, M. I., Donny, E. C., Gardiner, P. S., Grady, S. R., Heberlein, U., Leonard, S. S. Guidelines on nicotine dose selection for in vivo research. Psychopharmacology. 190, 269-319 (2007).
- Lang, T., Rizzoli, S. O. Membrane protein clusters at nanoscale resolution: more than pretty pictures. Physiology (Bethesda). 25, 116-1124 (2010).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved