A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Molten-Salt Synthesis of Complex Metal Oxide Nanoparticles
In This Article
Summary
Here, we demonstrate a unique, relatively low-temperature, molten-salt synthesis method for preparing uniform complex metal oxide lanthanum hafnate nanoparticles.
Abstract
The development of feasible synthesis methods is critical for the successful exploration of novel properties and potential applications of nanomaterials. Here, we introduce the molten-salt synthesis (MSS) method for making metal oxide nanomaterials. Advantages over other methods include its simplicity, greenness, reliability, scalability, and generalizability. Using pyrochlore lanthanum hafnium oxide (La2Hf2O7) as a representative, we describe the MSS protocol for the successful synthesis of complex metal oxide nanoparticles (NPs). Furthermore, this method has the unique ability to produce NPs with different material features by changing various synthesis parameters such as pH, temperature, duration, and post-annealing. By fine-tuning these parameters, we are able to synthesize highly uniform, non-agglomerated, and highly crystalline NPs. As a specific example, we vary the particle size of the La2Hf2O7 NPs by changing the concentration of the ammonium hydroxide solution used in the MSS process, which allows us to further explore the effect of particle size on various properties. It is expected that the MSS method will become a more popular synthesis method for nanomaterials and more widely employed in the nanoscience and nanotechnology community in the upcoming years.
Introduction
Molten-salt synthesis (MSS) involves the use of a molten salt as the reaction medium for preparing nanomaterials from their constituent precursors. The molten salt acts as the solvent and facilitates the enhanced reaction rate by increasing the contact area between reactants and their mobility. The choice of molten salts is of paramount importance for the success of the MSS method. The salt must meet some important quality requirements such as low melting point, compatibility with reacting species, and optimum aqueous solubility. Molten salt has been used previously to enhance the rate of solid-state reactions; however, in a flux system, only a small amount of molten salt is used (unlike in MSS, in which a large quantity is added to form a soluble medium for the reaction and control the properties of the synthesized nanomaterials, such as particle size, shape, and crystallinity, etc.). In this sense, MSS is a modification of the powder metallurgical method and different from the flux method1,2,3. The employment of molten salt can (1) increase reaction kinetic rate4 while decreasing synthesis temperature5, (2) increase the degree of reactant homogeneity6, (3) control crystalline size and morphology7, and (4) reduce the level of agglomeration.
Nanomaterials have been in high demand in scientific research and novel industrial applications because of their superior electrical, chemical, magnetic, optical, electronic, and thermal properties. Their properties are highly dependent on the particle size, shape, and crystallinity. Compared with other synthesis methods for nanomaterials, MSS has several obvious advantages; although, it is not yet as well-known as other synthesis methods in the nanoscience and nanotechnology community. As described below, these advantages include its simplicity, reliability, scalability, generalizability, environmental friendliness, cost effectiveness, relative low synthesis temperature, and free agglomeration of NPs with clean surface8.
Simplicity: The MSS process can be easily carried out in a simple laboratory with basic facilities. No sophisticated instrumentation is needed. Precursors and molten salts are air stable with no need for glove box handling.
Reliability: Once all initial synthesis parameters such as concentration, pH, processing time, and annealing temperature are optimized, high-quality and pure products are assured when using the MSS method. If all synthesis steps are carried out properly, the final products may attain all basic criteria needed for good-quality NPs. A novice to the MSS method will not change the synthesis outcome, as long as all synthesis parameters are properly and carefully followed.
Scalability: The MSS method's ability to produce large quantities of size- and shape-controlled particles is crucial. This critical factor is important because it allows for the determination of industrial usefulness and efficiency. Compared to other synthesis techniques, MSS can easily generate a sufficient amount of products by adjusting stoichiometric amounts during the process. This is an important feature of the method because it allows for convenience at the industrial level, making it a more desired approach due to this scalability9,10.
Generalizability: The MSS method is also a generalizable technique to produce nanoparticles with various compositions. Other than simple metal oxides and some fluorides, nanomaterials of complex metal oxides that have been successfully synthesized by the MSS method include perovskites (ABO3)10,11,12,13,14, spinel (AB2O4)15,16, pyrochlore (A2B2O7)4,17,18,19, and orthorhombic structures (A2B4O9)2,3,20. More specifically, these nanomaterials include ferrites, titanates, niobates, mullite, aluminium borate, wollastonite, and carbonated apatite7,9,21. The MSS method has also been used to produce nanomaterials of various morphologies such as nanospheres4, ceramics powder bodies22, nanoflakes23, nanoplates7, nanorods24, and core-shell nanoparticles (NPs)25, depending on synthesis conditions and crystal structure of the products.
Environmental friendliness: Several traditional methods for making nanomaterials involve the use of large amounts of organic solvents and toxic agents that generate environmental issues. The partial or total elimination of the use of them and the generation of waste by sustainable processes is in demand of green chemistry nowadays8. The MSS method is an environmentally friendly approach to synthesize nanomaterials by employing nontoxic chemical and renewable materials and minimizing waste, byproducts, and energy.
Relative low synthesis temperature: The processing temperature of the MSS method is relatively low compared to that required in a conventional solid-state reaction26 or a sol-gel combustion reaction27. This lower temperature saves energy while producing high-quality NPs.
Cost effectiveness: The MSS method does not require any harsh or costly reactants or solvents nor any specialized instrumentation. Water is the main solvent used for washing away the used molten salts, which are also cheap. Moreover, experimental setup needed includes only simple glassware and a furnace without specialized instrumentation, while nanomaterials with complex composition and refractory nature can be produced.
Agglomeration free with clean surface: During the MSS process, the formed nanoparticles are well-dispersed in the molten salt medium due to its large quantity, used along with its high ionic strength and viscosity1,6,8. Unlike colloidal synthesis and most hydrothermal/solvothermal processes, no protective surface layer is necessary to prevent the continuous growth and agglomeration of the formed NPs.
Exemplary synthesis of complex metal oxide NPs by the MSS method: The MSS method as a universal and cost-effective approach to rationally and large-scale synthesize nanomaterials for a sufficiently wide spectrum of material may be highly welcomed by scientists working in nanoscience and nanotechnology. Here, lanthanum hafnate (La2Hf2O7) was selected because of its multifunctional applications in the areas of X-ray imaging, high k-dielectric, luminescence, thermographic phosphor, thermal barrier coating, and nuclear waste host. La2Hf2O7 is also a good host for doped scintillators due to its high density, large effective atomic number, and the possibility of its crystal structure to be engineered along with an order-disorder phase transition. It belongs to the A2B2O7 family of compounds, in which "A" is a rare-earth element with a +3 oxidation state, and "B" represents a transition metallic element with a +4 oxidation state. However, due to the refractory nature and complex chemical composition, there has been a lack of proper low-temperature and large-scale synthesis methods for La2Hf2O7 NPs.
For fundamental scientific investigation and advanced technological applications, it is a prerequisite to make monodisperse, high-quality, and uniform A2B2O7 NPs. Here we use the synthesis of highly crystalline La2Hf2O7 NPs as an example to demonstrate the advantages of the MSS method. As schematically shown in Figure 1, La2Hf2O7 NPs were prepared by the MSS method with a two-step process following our previous reports. First, a single-source complex precursor of La(OH)3·HfO(OH)2·nH2O was prepared via a coprecipitation route. In the second step, size-controllable La2Hf2O7 NPs were synthesized through the facile MSS process using the single-source complex precursor and nitrate mixture (NaNO3:KNO3 = 1:1, molar ratio) at 650 °C for 6 h.
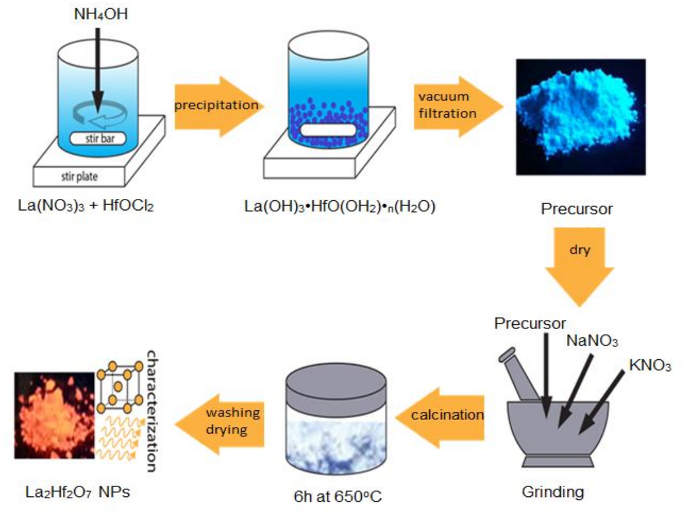
Figure 1: Schematic of the synthesis steps for La2Hf2O7 NPs via the MSS method. Please click here to view a larger version of this figure.
Protocol
1. Preparation of Single-Source Complex Precursor via a Coprecipitation Route
- Preparation of lanthanum and hafnium precursor solution
- Measure 200 mL of distilled water in a 500 mL beaker and start stirring at 300 rpm.
- Dissolve lanthanum and hafnium precursors in the stirring water [i.e., 2.165 g of lanthanum nitrate hexahydrate (La(NO3)3•6H2O) and 2.0476 g of hafnium dichloride oxide octahydrate (HfOCl2•8H2O)].
- Let the solution stir for 30 min before starting the titration.
- Preparation of diluted ammonia solution
- Prepare 200 mL of diluted ammonia solution with different concentrations, including 0.75%, 1.5%, 3.0%, 6.0%, and 7.5%. For example, add 20 mL of concentrated ammonia solution (NH4OH (aq), 28-30%) to 180 mL of distilled water in a separate beaker to make 3.0% diluted ammonia solution.
- Titration and washing the single source complex precursor
- Add the diluted ammonia solution prepared in the previous step into a burette and ensure that the burette is covered at all times, since ammonia solution tends to evaporate which decreases its concentration.
- Add the diluted ammonia solution in the burette into the stirring solution of lanthanum nitrate and hafnium dichloride oxide dropwise.
- Adjust the dropping speed of the ammonia solution accordingly so that it will be added over a period of 2 h.
- After several mL of ammonia solution have been delivered, ensure that the solution becomes cloudy. This is a simple sign that the precipitate of the single-source complex precursor of La(OH)3·HfO(OH)2·nH2O is forming.
- After 2 h, remove the stirring bar and allow the precipitate to age overnight.
- Check the pH of the coprecipitated solution before washing. Wash the precipitate with distilled water until the supernatant reaches a neutral pH, which normally takes 5-8 washes.
- Vacuum filtration and drying of precursor
- Vacuum filter the coprecipitated solution using a filter paper with a coarse porosity (40-60 µm; see Table of Materials) to separate the solid precipitate from the supernatant.
- Ensure that all complex precursor remnants are washed from the walls of the beaker.
- Air dry the resulting single-source complex precursor La(OH)3·HfO(OH)2·nH2O at room temperature overnight.
2. Molten-Salt Synthesis of Lanthanum Hafnate NPs
- Preparation of salt and precursor mixture
- Measure 30 mmol (3.033 g) of potassium nitrate (KNO3) and 30 mmol (2.549 g) of sodium nitrate (NaNO3).
- Combine the measured salts with 0.35 g of the as-prepared single-source complex precursor La(OH)3·HfO(OH)2·nH2O.
- If necessary, add 1-5 mL of acetone or ethanol to the mixture to facilitate the grinding. Ensure that all the solvent is evaporated before placing the mixture into a crucible.
- Grind the mixed salts and precursor as fine as possible for about 30 min using a mortar and pestle.
- Molten-salt processing
- Place the resulting mixture in a corundum crucible, then place it in a muffle furnace.
- Set the furnace at 650 °C for 6 h with a ramp rate of 10 °C/min.
- After the sample and furnace have cooled to room temperature, take out the crucible and soak the sample in a beaker filled with distilled water overnight.
- Washing and drying the La2Hf2O7 NPs
- Empty the sample from the crucible into a 1 L beaker.
- Wash the sample with distilled water 5-8 times until the supernatant is clear of salts and not cloudy anymore.
- Purify the product by centrifugation or vacuum filtration to remove any residual impurity.
- Dry the product in the oven at 90 °C overnight.
Results
The as-synthesized La2Hf2O7 NPs may exist in the ordered pyrochlore phase. However, chemical doping, pressure, and temperature could modify the phase to defect fluorite. It is possible for our material to have multiple phases; however, here we focus only on the pyrochlore phase for simplicity. X-ray diffraction (XRD) and Raman spectroscopy have been used to systematically characterize their phase purity, structure, and phase. The crystalline size can ...
Discussion
The chart in Figure 4 provides several reliable controlling factors of the MSS method and accounts for alternative pathways to fine-tune the features of synthesized nanomaterials. In addition, it helps identify critical steps in the MSS process.
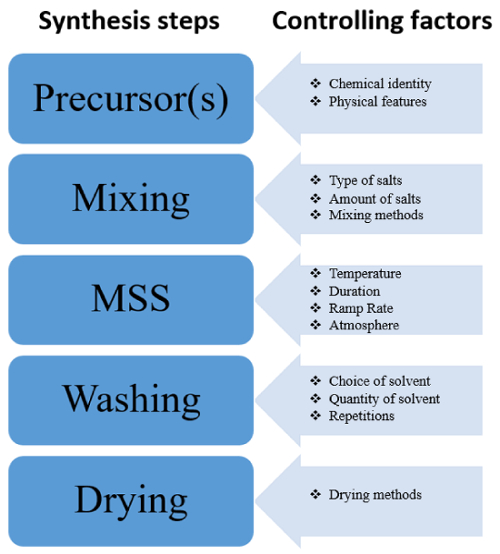
Figure 4: Flowchart of the critical steps of MSS indi...
Disclosures
There are no conflicts to declare.
Acknowledgements
The authors thank the financial support provided by the National Science Foundation under CHE (award #1710160) and the USDA National Institute of Food and Agriculture (award #2015-38422-24059). The Department of Chemistry at the University of Texas Rio Grande Valley is grateful for the generous support provided by a Departmental Grant from the Robert A. Welch Foundation (Grant No. BX-0048). S.K.G. would like to thank the United States-India Education Foundation (USIEF) and the Institute of International Education (IIE) for his Fulbright Nehru Postdoctoral Fellowship (award #2268/FNPDR/2017).
Materials
| Name | Company | Catalog Number | Comments |
| Acetone, ACS, 99.5+% | Alfa Aesar | 67-64-1 | Dried over 4A sieves |
| Hafnium dichloride oxide octahydrate, 98+% (metals basis excluding Zr), Zr <1.5% | Alfa Aesar | 14456-34-9 | Hygroscopic |
| Lanthanum(III) nitrate hexahydrate | Aldrich | 10277-43-7 | Hygroscopic |
| Potassium nitrate, ReagentPlus R, ≥99.0% | Sigma-Aldrich | 7757-79-1 | Hygroscopic |
| Sodium nitrate, ReagentPlus R, ≥99.0% | Sigma-Aldrich | 7631-99-4 | |
| Ammonium hydroxide, 28% NH3, NH4OH | Alfa Aesar | 1336-21-6 | |
| Filter paper, P8 grade | Fisherbrand |
References
- Kimura, T. Molten salt synthesis of ceramic powders. Advances in Ceramics. , 75-100 (2011).
- Mao, Y., Park, T. J., Wong, S. S. Synthesis of classes of ternary metal oxide nanostructures. Chemical Communications. (46), 5721-5735 (2005).
- Mao, Y., Zhou, H., Wong, S. S. Perovskite-phase metal oxide nanostructures: Synthesis, properties, and applications. Material Matters. 5, 50-53 (2010).
- Mao, Y., Guo, X., Huang, J. Y., Wang, K. L., Chang, J. P. Luminescent nanocrystals with A2B2O7 composition synthesized by a kinetically modified molten salt Method. The Journal of Physical Chemistry C. 113 (4), 1204-1208 (2009).
- Yu, Y., Wang, S., Li, W., Chen, Z. Low temperature synthesis of LaB6 nanoparticles by a molten salt route. Powder Technology. 323, 203-207 (2018).
- Liu, X., Fechler, N., Antonietti, M. Salt melt synthesis of ceramics, semiconductors and carbon nanostructures. Chemical Society Reviews. 42 (21), 8237-8265 (2013).
- Chang, Y., Wu, J., Zhang, M., Kupp, E., Messing, C. L. Molten salt synthesis of morphology controlled alpha-alumina platelets. Ceramics International. 43 (15), 12684-12688 (2017).
- Mao, Y., Park, T. J., Zhang, F., Zhou, H., Wong, S. S. Environmentally friendly methodologies for nanostructure synthesis. Small. 3 (7), 1122-1139 (2007).
- Liu, J. R., Hong, R. Y., Feng, W. G., Badami, D., Wang, Y. Q. Large scale production of strontium ferrite by molten salt assited coprecipitation. Powder Technology. 262, 142-149 (2014).
- Yuanbing, M., Banerjee, S., Wong, S. S. Large-scale synthesis of single-crystalline perovskite nanostructures. Journal of the American Chemical Society. 125 (51), 15718-15719 (2003).
- Mao, Y. Facile synthesis of ferromagnetic double perovskite oxide La2BMnO6 nanoparticles. RSC Advances. 2 (33), 12675-12678 (2012).
- Hailili, R., Wang, C., Lichtfouse, E. Perovskite nanostructures assembled in molten salt based on halogen anions KX (X = F, Cl and Br): Regulated morphology and defect-mediated photocatalytic activity. Applied Catalysis B: Enviromental. 232, 531-543 (2018).
- Yuanbing Mao, J. P., McCloy, J. S. Magnetic properties of double perovskite oxide La2BMnO6 nanocrystals. Nanoscale. 5 (11), 4720-4728 (2013).
- Mao, Y., Wong, S. S. Reproducible composition and shape control of crystalline Ca1-xSrxTiO3 perovskite nanoparticles. Advanced Materials. 17 (18), 2194-2199 (2005).
- Rojas-Hernandez, R. E., et al. Original synthetic route to obtain a SrAl2O4 phosphor by the molten salt method: insights into the reaction mechanism and enhancement of the persistent luminescence. Inorganic Chemistry. 54 (20), 9896-9907 (2015).
- Reddy, M. V., Xu, Y., Rajarajan, V., Ouyang, T., Chowdari, B. V. R. Template free facile molten synthesis and energy storage studies on MCo2O4 (M = Mg, Mn) as anode for Li-ion batteries. ACS Sustainable Chemistry and Engineering. 3 (12), 3035-3042 (2015).
- Zuniga, J. P., Gupta, S. K., Pokhrel, M., Mao, Y. Exploring optical properties of La2Hf2O7:Pr3+ nanoparticles under UV and X-ray excitations for potential lighting and scintillating applications. New Journal of Chemistry. 42 (12), 9381-9392 (2018).
- Pokhrel, M., Wahid, K., Mao, Y. Systematic studies on RE2Hf2O7:5%Eu3+ (RE = Y, La, Pr, Gd, Er, and Lu) nanoparticles: Effects of the A-Site RE3+ cation and calcination on structure and photoluminescence. The Journal of Physical Chemistry C. 120 (27), 14828-14839 (2016).
- Wahid, K., Pokhrel, M., Mao, Y. Structural, photoluminescence and radioluminescence properties of Eu3+ doped La2Hf2O7 nanoparticles. Journal of Solid State Chemistry. 245, 89-97 (2017).
- Park, T. J., Papaefthymiou, G. C., Moodenbaugh, A. R., Mao, Y., Wong, S. S. Synthesis and characterization of submicron single-crystalline Bi2Fe4O9 cubes. Journal of Materials Chemistry. 15 (21), 2099-2105 (2005).
- Gilbert, M. R. Molten salt synthesis of titanate pyrochlore waste-forms. Ceramics International. 42 (4), 5263-5270 (2016).
- Huang, Z., et al. Molten salt synthesis of La2Zr2O7 ultrafine powders. Ceramics International. 42 (5), 6221-6227 (2016).
- Huang, Z., Duan, H., Liu, J., Zhang, H. Preparation of lanthanum cerate powders via a simple molten salt route. Ceramics International. 42 (8), 10482-10486 (2016).
- Wang, G., et al. Fabrication of rod-like Ti4O7 with high conductivity by molten salt synthesis. Materials Letters. 186, 361-363 (2017).
- Pokhrel, M., Burger, A., Groza, M., Mao, Y. Enhance the photoluminescence and radioluminescence of La2Zr2O7:Eu3+ core nanoparticles by coating with a thin Y2O3 shell. Optical Materials. 68, 35-41 (2017).
- Ramesh, G., Subramanian, V., Sivasubramanian, V. Dielectric properties of lead indium niobate ceramics synthesized by conventional solid state reaction method. Materials Research Bulletin. 45 (12), 1871-1874 (2010).
- Gupta, S. K., et al. Role of various defects in the photoluminescence characteristics of nanocrystalline Nd2Zr2O7: An introspection through spectroscopic and DFT calculations. Journal of Materials Chemistry C. 4 (22), 4988-5000 (2016).
- Wang, X., Zhu, Y., Zhang, W. Preparation of lanthanum zirconate nano-powders by molten salt method. Journal of Non-Crystalline Solids. 356 (20-22), 1049-1051 (2010).
- Popov, V. V., et al. Fluorite-pyrochlore phase transition in nanostructured Ln2Hf2O7 (Ln = La-Lu). Journal of Alloys and Compounds. 689, 669-679 (2016).
- Rybarczyk, M. K., Gontarek, E., Lieder, M., Titirici, M. M. Salt melt synthesis of curved nitrogen-doped carbon nanostructures: ORR kinetics boost. Applied Surface Science. 435, 543-551 (2018).
- Ozen, M., Mertens, M., Snikers, F., D'Hondt, H., Cool, P. Molten-salt synthesis of tetragonal micron-sized barium titanate from a peroxo-hydroxide precursor. Advanced Powder Technology. 28 (1), 146-154 (2017).
- Fazli, R., Fazli, M., Safaei-Naeini, Y., Golestani-Fard, F. The effects of processing parameters on formation of nano-spinel (MgAl2O4) from LiCl molten salt. Ceramics International. 39 (6), 6265-6270 (2013).
- Bortolani, F., Dorey, R. A. Molten salt synthesis of PZT powder for direct write inks. Journal of the European Ceramic Society. 30 (10), 2073-2079 (2010).
- Zhou, H., Mao, Y., Wong, S. S. Probing structure-parameter correlations in the molten synthesis of BaZrO3 perovskite submicron-sized particles. Chemistry of Materials. 19 (22), 5238-5249 (2007).
- Kimura, T., Machida, M., Yamaguchi, T., Newnham, R. E. Products of reaction between PbO and Nb2O5 in molten KCl or NaCl. Journal of the American Ceramic Society. 66 (10), 195-197 (1983).
- Liu, S., et al. A novel rechargeable zinc-air battery with molten salt electrolyte. Journal of Power Sources. 342, 435-441 (2017).
- Huang, Z., Li, B., Liu, J. Molten-salt synthesis of oxyapatite La9.33Si6O26 powders as electrolytes for intermediate temperature solid oxide fuel cells. Physica status solidi A - Applicationand Materials Science. 207 (10), 2247-2251 (2010).
- Ahmed, J., Mao, Y. Synthesis, characterization and electrocatalytic properties of delafossite CuGaO2. Journal of Solid State Chemistry. 242 (1), 77-85 (2016).
- Ahmed, J., Mao, Y. Ultrafine iridium oxide nanorods synthesized by molten salt method toward electrocatalytic oxygen and hydrogen evolution reactions. Electrochimica Acta. 212, 686-693 (2016).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved