A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Longitudinal Morphological and Physiological Monitoring of Three-dimensional Tumor Spheroids Using Optical Coherence Tomography
In This Article
Summary
Optical coherence tomography (OCT), a three-dimensional imaging technology, was used to monitor and characterize the growth kinetics of multicellular tumor spheroids. Precise volumetric quantification of tumor spheroids using a voxel counting approach, and label-free dead tissue detection in the spheroids based on intrinsic optical attenuation contrast, were demonstrated.
Abstract
Tumor spheroids have been developed as a three-dimensional (3D) cell culture model in cancer research and anti-cancer drug discovery. However, currently, high-throughput imaging modalities utilizing bright field or fluorescence detection, are unable to resolve the overall 3D structure of the tumor spheroid due to limited light penetration, diffusion of fluorescent dyes and depth-resolvability. Recently, our lab demonstrated the use of optical coherence tomography (OCT), a label-free and non-destructive 3D imaging modality, to perform longitudinal characterization of multicellular tumor spheroids in a 96-well plate. OCT was capable of obtaining 3D morphological and physiological information of tumor spheroids growing up to about 600 µm in height. In this article, we demonstrate a high-throughput OCT (HT-OCT) imaging system that scans the whole multi-well plate and obtains 3D OCT data of tumor spheroids automatically. We describe the details of the HT-OCT system and construction guidelines in the protocol. From the 3D OCT data, one can visualize the overall structure of the spheroid with 3D rendered and orthogonal slices, characterize the longitudinal growth curve of the tumor spheroid based on the morphological information of size and volume, and monitor the growth of the dead-cell regions in the tumor spheroid based on optical intrinsic attenuation contrast. We show that HT-OCT can be used as a high-throughput imaging modality for drug screening as well as characterizing biofabricated samples.
Introduction
Cancer is the second leading cause of death in the world1. Developing drugs targeting cancer is of crucial importance for patients. However, it is estimated that more than 90% of new anti-cancer drugs fail in the development phase because of a lack of efficacy and unexpected toxicity in clinical trials2. Part of the reason can be attributed to the use of simple two-dimensional (2D) cell culture models for compound screening, which provide results with limited predictive values of compound efficacy and toxicity for the following stages of drug discovery2,3,4. Recently, three-dimensional (3D) tumor spheroid models have been developed to provide clinically relevant physiological and pharmacological data for anti-cancer drug discovery3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25. Since these spheroids can mimic tissue-specific properties of tumors in vivo, such as nutrient and oxygen gradient, hypoxic core as well as drug resistance19, the use of these models can potentially shorten drug discovery timelines, reduce costs of investment, and bring new medicines to patients more effectively. One critical approach to evaluating compound efficacy in 3D tumor spheroid development is to monitor the spheroid growth and recurrence under treatments9,26. To do this, quantitative characterizations of the tumor morphology, involving its diameter and volume, with high-resolution imaging modalities, are imperative.
Conventional imaging modalities, such as bright-field, phase contrast7,9,22,24, and fluorescence microscopy8,9,16,18,22 can provide a measurement of the spheroid's diameter but cannot resolve the overall structure of the spheroid in 3D space. Many factors contribute to these limitations, including penetration of the probing light in the spheroid; diffusion of the fluorescent dyes into the spheroid; emitting fluorescent signals from excited fluorescent dyes inside or on the opposite surface of the spheroid due to strong absorption and scattering; and depth-resolvability of these imaging modalities. This often leads to an inaccurate volume measurement. Development of the necrotic core in spheroids mimics necrosis in in vivo tumors6,10,15,19,25. This pathological feature is unlikely reproduced in 2D cell cultures19,25,27,28. With a spheroid size larger than 500 µm in diameter, a three-layer concentric structure, including an outer layer of proliferating cells, a middle layer of quiescent cells, and a necrotic core, can be observed in the spheroid6,10,15,19,25, due to lack of oxygen and nutrients. Live and dead cell fluorescence imaging is the standard approach to label the boundary of the necrotic core. However, again, penetrations of both these fluorescent dyes and visible light hinder the potential to probe into the necrotic core to monitor its development in its actual shape.
An alternative 3D imaging modality, optical coherence tomography (OCT) is introduced to characterize the tumor spheroids. OCT is a biomedical imaging technique that is capable of acquiring label-free, non-destructive 3D data from up to 1-2 mm depths in biological tissues29,30,31,32,33,34. OCT employs low-coherence interferometry to detect back-scattered signals from different depths of the sample and provides reconstructed depth-resolved images at micron-level spatial resolutions in both lateral and vertical directions. OCT has been widely adopted in ophthalmology35,36,37 and angiography38,39. Previous studies have used OCT to observe the morphology of in vitro tumor spheroids in basement membrane matrix (e.g., Matrigel) and evaluate their responses to photodynamic therapy40,41. Recently, our group established a high-throughput OCT imaging platform to systematically monitor and quantify the growth kinetics of 3D tumor spheroids in multi-well plates42. Precise volumetric quantification of 3D tumor spheroids using a voxel counting approach and label-free necrotic tissue detection in the spheroids based on intrinsic optical attenuation contrast were demonstrated. This paper describes the details of how the OCT imaging platform was constructed and employed to obtain high-resolution 3D images of tumor spheroids. The step-by-step quantitative analyses of the growth kinetics of 3D tumor spheroids, including accurate measurements of spheroid diameter and volumes, is described. Also, the method of the non-destructive detection of necrotic tissue regions using OCT, based on the intrinsic optical attenuation contrast is presented.
Protocol
1. Preparation of Cells
- Obtain cell lines from a qualified supplier.
NOTE: Verify that cells from the cell lines of interest can form spheroid in the culture media or with the help of a substrate (basement membrane matrix like Matrigel). Look into the literature9 or perform one round of a pre-experiment for a check. - Thaw the frozen cells following the specific procedure provided by the cell-line supplier. A general procedure can be found elsewhere43.
- Culture the cells for 1-2 passages in 25 cm2 culture flasks. The cells are then ready to use for 3D cell culture.
- Monitor the health status of the cells every day and maintain them in an incubator under standard conditions (37 °C, 5% CO2, 95% humidity). Refresh the media as needed.
NOTE: The culture medium consists of DMEM (4.5 g/L glucose), 1% antibiotic-antimycotic, 10% fetal bovine serum. Subculture cells before they reach confluence in the culture flask. Follow the cell culture guideline provided by the supplier. A general procedure can be found elsehwhere44. - Perform 3D cell culture in multi-well plates based on the following general protocol9.
- Remove the culture media from the culture flask and wash it with sterilized phosphate buffered saline (PBS, heated to 37 °C).
- Resuspend the cells by adding 1 mL of trypsin ethylenediaminetetraacetic acid (EDTA, 0.5%) into the flask for 3 min. Then, add culture media to dilute the trypsin.
- Transfer the cell suspension into a 15 mL centrifuge tube and centrifuge for 5 min at 500 x g and room temperature.
- Remove the supernatant and resuspend cells with 4 mL of pre-warmed culture medium. Pipette one drop of sample onto a hemocytometer for cell counting to determine cell concentration. Dilute the cells to appropriate concentration for seeding (e.g., 3,000 cells/mL).
NOTE: Optimize the initial cell concentration of the spheroid for each cell-line and each type of multi-well plate (96-well, 384-well or 1536-well). - Seed cells into an ultra-low attachment (ULA) round-bottomed multi-well plate. Add 200 µL of cells suspension into each well at the concentration of 3,000 cells/mL so that each well has about 600 cells.
- At RT, centrifuge the whole plate using a plate adapter for 7 min, right after seeding, at a speed of 350 x g or the lowest speed available.
NOTE: The centrifuge helps gather cells to the center of the well to facilitate forming a single, uniform spheroid. The centrifuge step is performed only once at the beginning to form the tumor spheroids. It will not be repeated when the tumor spheroids start growing. - Maintain the multi-well plate at 37 °C and 5% CO2 in a culture incubator and refresh the culture media every 3 days.
NOTE: Growth time may vary for different 3D culture conditions. In our study, 3,000 cells/mL is used for both U-87 MG and HCT 116 cell lines in 96-well plates, so that the spheroid can grow to ~500 μm in 4‒7 days for HCT 116 cells. Consider adding media supplements and growth factors for different spheroid models, based on the general 3D culture protocol. - Perform OCT imaging of tumor spheroids every 3‒4 days for a longitudinal study of their growth.
NOTE: Recommended time points for OCT imaging would be day 4, day 7, day 11, day 14, day 18 and day 21.
2. High-throughput OCT Imaging Platform
NOTE: See referenced work29,30,31,32,33,34 for a thorough review of principles and applications of OCT. See Figure 1 and Huang et al.42 for details of the custom OCT imaging system used in this study.
- Choose an appropriate broadband light source for the OCT system for tumor spheroid imaging.
NOTE: Here, a superluminescent diode (SLD, Figure 1A,B) with a central wavelength of ~1,320 nm and ~110 nm bandwidth was used as a broadband light source. - Construct the reference arm and sample arm of the OCT system following the schematics (See Figure 1A,B for details). See the Table of Materials for a list of optical components to construct the OCT system. Ensure that the optical path length of the reference arm and sample arm are closely matched.
- Construct the spectrometer, including a collimator, a grating, an F-theta lens and a line-scan camera (See Figure 1C for setup34) for details of spectrometer design of OCT. Alternatively, select a commercial spectrometer that matches the center wavelength of the light source. Make sure that the spectrometer is aligned correctly to cover the entire laser bandwidth, to achieve high photon collection efficiency and to provide slow wash-out of the interference pattern.
- Characterize the performance of the OCT system, including the following metrics such as sample arm power, total imaging depth, depth-dependent sensitivity, axial resolution, depth of focus and lateral resolution. Place a weak reflector (e.g., a mirror with a neutral density filter) as a sample to measure the depth-dependent sensitivity, axial resolution, and depth of focus. Place a USAF resolution test chart target as the sample to check the lateral resolution.
NOTE: See references34,45 for definitions of metrics of OCT performance and protocols to characterize these metrics45. See Table 1 for a list of measured parameters for the custom OCT system used in our study. - Select a motorized translation stage to provide horizontal movement of the multi-well plate to image tumor spheroids in different wells (See Figure 1B). Use a stage with a travel range larger than 108 mm x 72 mm to ensure a full scanning of all the wells of the multi-well plate. Use a 2D or 3D motorized translation stage with software control to enable precise location of each well and automation of the OCT system for high-throughput imaging.
- Use a plate adapter or design a plate holder (by 3D printing) to hold the multi-well plate in a fixed position.
- Correct the tilting and rotation of the multi-well plate using a 2D tilting stage and a rotation stage mounted on the translational stage (See Figure 1D), before conducting any OCT imaging to minimize variation of the focus plane from different wells. Use D2, D11, B6, D6, G6 as the guiding wells when monitoring their relative positions in the OCT images (Figure 1A).
- Adjust the rotation of the plate to ensure the edges of the plate are parallel with the direction of stage movement so that the wells remain at the same horizontal positions in the OCT images (Figure 1E). Adjust the tilting of the plate to be parallel to the optical table so that the wells remain at the same vertical locations for OCT imaging (Figure 1F).
NOTE: Adjustment of the tilting angle and focus help optimize the OCT image quality for all the wells. However, variations of the height of culture media in different wells may cause changes in optical paths which may lead to defocusing of the spheroid image. Auto-focus may be implemented to control the focal plane of OCT imaging to achieve optimized image quality. The adjustment step does not resolve poor OCT image quality of the tumor spheroid due to the following issues: the spheroid decentering due to initial seeding location; spheroid elevation when embedded in biofabricated extracellular matrices; poor plate quality with large variations of the height of well bottoms. Additional software control with auto-focus or self-alignment functions can be implemented to optimize the performance of the OCT imaging system. - Use a custom computer program to control the OCT image acquisition and the stage movement to collect data from each well sequentially.
3. OCT Scanning and Processing of Tumor Spheroids
- On the day of the OCT imaging of tumor spheroids, take the multi-well plate from the incubator. Transfer the multi-well plate under the OCT imaging system. Place it on top of the plate adapter.
NOTE: OCT imaging of tumor spheroids may be performed with the polystyrene plate lid on or off. However, the water condensations on the lid due to evaporation from the wells may affect light transmission and distort the light path, yielding less optimal OCT images from the spheroids. - Adjust the height of the plate by moving along the z-direction of the translation stage. Maintain the focal plane position at ~100–200 μm below the top surface of each spheroid, to minimize the effect of the non-uniform depth-wise focal profile.
- Set a proper OCT scanning range (e.g., 1 mm x 1 mm) in the custom software to cover the whole tumor spheroid according to its development stages. Click Save Parameters to save the setting.
- Use the custom software to acquire 3D OCT images of tumor spheroids one by one for all the wells of the plate containing spheroids. Click the Preview button to view the preview image and click the Acquire button to acquire the OCT image.
NOTE: Ensure that the OCT spheroid data are collected when the stage is not in motion. The spheroid is usually located at the center of the U-bottom well. However, the spheroid may be shifted in the culture media when the stage is accelerating or decelerating due to the inertia of the spheroid in the culture media. - Process 3D OCT datasets of tumor spheroids to generate OCT structural images with a custom C++ processing code. See Figure 2A for a flowchart of post-processing of OCT data.
NOTE: See Figure 4A for the generated 3D OCT structural images.- See Chapter 5 of Drexler and Fujimoto34 and Jian et al.46 for detail descriptions of post-processing steps of OCT data. Calibrate the pixel size in all three dimensions. Re-scale the OCT structural images on corrected scales.
NOTE: The distance in the axial direction (z-direction) of OCT images is a measure of the optical path difference between the reference arm and sample arm. Thus, the refractive index of the sample (n) needs to be taken into consideration when calibrating the pixel size in the axial direction for rescaling. In our study, we use n = 1.37 as the refractive index of the tumor spheroid42.
- See Chapter 5 of Drexler and Fujimoto34 and Jian et al.46 for detail descriptions of post-processing steps of OCT data. Calibrate the pixel size in all three dimensions. Re-scale the OCT structural images on corrected scales.
- Generate the collage of spheroid images using 2D OCT Images in three cross-sectional XY, XZ, and YZ planes across the centroid of the spheroid. See Figure 3C–E for the representative output of collages of spheroid images. Perform image registration for all the spheroids, using the MATLAB function dftregistration47, to ensure that the centroids of all the spheroid are located approximately at the same location.
- Obtain 3D rendering of the spheroid using a commercial or custom software.
NOTE: The following steps show how to obtain the 3D rendering of tumor spheroids using a commercial software.- Load the 3D OCT data into the software.
- Click the Surpass panel. Then, click Add New Volume. Choose the Blend mode to use for 3D rendering.
- Adjust the viewing angle by dragging the image using the mouse pointer.
4. Morphological Quantification of 3D Tumor Spheroids
NOTE: A custom written code in MATLAB processes this quantification. Click the Run button to initiate the process. See Figure 2B for the flowchart of the steps of morphological quantification of spheroids.
- Quantify spheroid diameter, height, and diameter-based volume.
- Select 2D OCT Images in three cross-sectional XY, XZ, and YZ planes that cross the centroid of the spheroid.
- Measure the diameter and height of the spheroid in XY and XZ planes, respectively.
- Calculate diameter-based spheroid volume using:
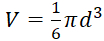 , with a presumption of the spherical shape of tumor.
, with a presumption of the spherical shape of tumor.
- Quantify voxel-based spheroid volume.
- Apply a 3D averaging filter on the OCT structural data of spheroid to remove speckles.
- Segment tumor spheroids using the Canny edge detection48 filter, frame by frame, with a proper threshold separating the tumor spheroid region from the well bottom.
- Group connective voxels for 3D data (see built-in function: bwconncomp).
- Calculate the mean distance between each connective voxel in the group and the spheroid centroid (manually chosen), for each group. Identify the spheroid region as the group with the minimum mean distance.
- Count the number of voxels within the spheroid region and then multiply by the actual volume of an individual voxel (volume/voxel), yielding the total volume of the spheroid.
5. Dead-Cell Region Detection of 3D Tumor Spheroids
NOTE: In a homogeneous medium, OCT back-scattered intensity detected as a function of depth (I(z)) can be described by the Beer-Lambert Law49: 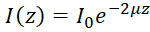 , where z represents the depth, μ is the optical attenuation coefficient, and I0 is the incident intensity to the sample. Hence the derived optical attenuation coefficient can be expressed as:
, where z represents the depth, μ is the optical attenuation coefficient, and I0 is the incident intensity to the sample. Hence the derived optical attenuation coefficient can be expressed as: 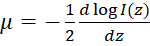 . Since OCT images are often plotted on a logarithmic scale, the slope of the OCT intensity profile can be retrieved to derive the optical attenuation coefficient. See Figure 2C for a flowchart of the generation of the optical attenuation maps.
. Since OCT images are often plotted on a logarithmic scale, the slope of the OCT intensity profile can be retrieved to derive the optical attenuation coefficient. See Figure 2C for a flowchart of the generation of the optical attenuation maps.
- Perform segmentation to remove unwanted regions outside the spheroid. Perform 3D average filter to suppress the speckle noise that is inherent in OCT images.
- Obtain pixel-wise optical attenuation coefficients by linear fitting the log-scale OCT intensity profile over a certain depth range (moving window), extract its slope, and multiply the slope by -1/2.
NOTE: The attenuation coefficient at each voxel within the segmented spheroid region is calculated based on the slope of OCT intensity profile in a 10-voxel depth window (~40 μm in depth), with the voxel located in the middle of the window. - Apply the methods above (steps 5.1 and 5.2) to each axial scan in a frame and each frame in a 3D dataset containing the segmented spheroid region until optical attenuation coefficients for all voxels of the segmented spheroid region are calculated.
- Perform the binary thresholding to highlight the high-attenuation region.
NOTE: See Huang et al.42 for the determination of threshold of the high-attenuation region using histogram analysis. - Highlight the binarized optical attenuation map on the original image to label the dead-cell region (blending). Generate the 3D-rendered image of the blended attenuation map to visualize the 3D distribution of the dead-cell region.
6. Histology and Immunohistochemistry
NOTE: Histology and immunohistochemistry (IHC) stained images of tumor spheroids are obtained to correlate with the corresponding OCT results.
- At selected time points, select 1-2 tumor spheroids from the multi-well plate for histology and IHC staining. Use a pipette with the 1 mL pipette tips to transfer the spheroid from the well to a 1.5 mL centrifuge tube.
NOTE: Cut the 1 mL pipette tip before transfer to ensure that the opening of the tip is larger than the size of the tumor spheroid to avoid damaging the structure of the spheroid. - Collect each tumor spheroid in a single 1.5 mL microcentrifuge tube filled with 10% formaldehyde and fix for 48 h.
- Perform the histology and IHC processes for each spheroid, using standard paraffin embedding techniques.
NOTE: Stain 5 μm thick sections of tumor spheroids for hematoxylin and eosin (H&E) and terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) apoptosis detection. A counterstaining of hematoxylin is applied to TUNEL. A digital slide scanner was used to scan the stained sample and obtain high-resolution histological and IHC images.
Results
High Throughput Optical Coherence Tomography Imaging of Spheroids in a 96-well Plate
Figure 3 exhibits the result of HT-OCT scanning of a 96-well plate with HCT 116 tumor spheroids on Day 3. The sequential scan of the whole plate starts from the bottom-right well (H12). Figure 3B shows the flow chart of the software implementation of the HT-OCT system. After one spheroid d...
Discussion
Tumor activity is highly relevant to its morphological structure. Similar to monitoring characteristic growth curve for 2D cell cultures, tracking the growth curve for 3D tumor spheroids is also a conventional approach to characterize the long-term spheroid growth behavior for different cell lines. Notably, we can characterize the drug response by analyzing tumor degradation or tumor regrowth directly reflected in the growth curve. Therefore, quantitative assessment of 3D tumor spheroids, including the size and volume, t...
Disclosures
The authors disclose no competing interest.
Acknowledgements
This work was supported by NSF grants IDBR (DBI-1455613), PFI:AIR-TT (IIP-1640707), NIH grants R21EY026380, R15EB019704 and R01EB025209, and Lehigh University startup fund.
Materials
| Name | Company | Catalog Number | Comments |
| Custom Spectral Domain OCT imaging system | Developed in our lab | ||
| Superluminescent Diode (SLD) | Thorlabs | SLD1325 | light source |
| 2×2 single mode fused fiber coupler, 50:50 splitting ratio | AC Photonics | WP13500202B201 | |
| Reference Arm | |||
| Lens Tube | Thorlabs | ||
| Adapter | Thorlabs | ||
| Collimating Lens | Thorlabs | AC080-020-C | |
| Focusing Lens | Thorlabs | ||
| Kinematic Mirror Mount | Thorlabs | ||
| Mirror | Thorlabs | ||
| 1D Translational Stage | Thorlabs | ||
| Continuous neutral density filter | Thorlabs | ||
| Pedestrial Post | Thorlabs | ||
| Clamping Fork | Thorlabs | ||
| Sample Arm | |||
| Lens Tube | Thorlabs | ||
| Adapter | Thorlabs | ||
| Collimating Lens | Thorlabs | AC080-020-C | |
| Galvanometer | Thorlabs | ||
| Relay Lens | Thorlabs | AC254-100-C | two Relay lens to make a telescope setup |
| Triangle Mirror Mount | Thorlabs | ||
| Mirror | Thorlabs | ||
| Objective | Mitutoyo | ||
| Pedestrial Post | Thorlabs | ||
| Clamping Fork | Thorlabs | ||
| Polarization Controller | Thorlabs | ||
| 30mm Cage Mount | Thorlabs | ||
| Cage Rod | Thorlabs | ||
| Stage | |||
| 3D motorized translation stage | Beijing Mao Feng Optoelectronics Technology Co., Ltd. | JTH360XY | |
| 2D Tilting Stage | |||
| Rotation Stage | |||
| Plate Holder | 3D printed | ||
| Spectrometer | |||
| Lens Tube | Thorlabs | ||
| Adapter | Thorlabs | ||
| Collimating Lens | Thorlabs | AC080-020-C | |
| Grating | Wasatch | G = 1145 lpmm | |
| F-theta Lens | Thorlabs | FTH-1064-100 | |
| InGaAs Line-scan Camera | Sensor Unlimited | SU1024-LDH2 | |
| Name | Company | Catalog Number | Comments |
| Cell Culture Component | |||
| HCT 116 Cell line | ATCC | CCL-247 | |
| Cell Culture Flask | SPL Life Sciences | 70025 | |
| Pipette | Fisherbrand | 14388100 | |
| Pipette tips | Sorenson Bioscience | 10340 | |
| Gibco GlutaMax DMEM | Thermo Fisher Scientific | 10569044 | |
| Fetal Bovine Serum, certified, US origin | Thermo Fisher Scientific | 16000044 | |
| Antibiotic-Antimycotic (100X) | Thermo Fisher Scientific | 15240062 | |
| Corning 96-well Clear Round Bottom Ultra-Low Attachment Microplate | Corning | 7007 | |
| Gibco PBS, pH 7.4 | Thermo Fisher Scientific | 10010023 | |
| Gibco Trypsin-EDTA (0.5%) | Thermo Fisher Scientific | 15400054 | |
| Forma Series II 3110 Water-Jacketed CO2 Incubators | Thermo Fisher Scientific | 3120 | |
| Gloves | VWR | 89428-750 | |
| Parafilm | Sigma-Aldrich | P7793 | |
| Transfer pipets | Globe Scientific | 138080 | |
| Centrifuge | Eppendorf | 5702 R | To centrifuge the 15 mL tube |
| Centrifuge | NUAIRE | AWEL CF 48-R | To centrifuge the 96-well plate |
| Microscope | Olympus | ||
| Name | Company | Catalog Number | Comments |
| Histology & IHC | |||
| Digital slide scanner | Leica | Aperio AT2 | Obtain high-resolution histological images |
| Histology Service | Histowiz | Request service for histological and immunohistological staining of tumor spheroid | |
| Name | Company | Catalog Number | Comments |
| List of Commerical OCTs | |||
| SD-OCT system | Thorlabs | Telesto Series | |
| SD-OCT system | Wasatch Photonics | WP OCT 1300 nm | |
| Name | Company | Catalog Number | Comments |
| Software for Data Analyses | |||
| Basic Image Analysis | NIH | ImageJ | Fiji also works. |
| 3D Rendering | Thermo Fisher Scientific | Amira | Commercial software. Option 1 |
| 3D Rendering | Bitplane | Imaris | Commercial software. Option 2. Used in the protocol |
| OCT acquisition software | custom developed in C++. | ||
| Stage Control | Beijing Mao Feng Optoelectronics Technology Co., Ltd. | MRC_3 | Incorporated into the custom OCT acquisition code |
| OCT processing software | custom developed in C++. Utilize GPU. Incorporated into the custom OCT acquisition code. | ||
| Morphological and Physiological Analysis | custom developed in MATLAB |
References
- Kola, I., Landis, J. Can the pharmaceutical industry reduce attrition rates?. Nature Reviews Drug Discovery. 3 (8), 711-716 (2004).
- Breslin, S., O'Driscoll, L. Three-dimensional cell culture: the missing link in drug discovery. Drug Discovery Today. 18, 240-249 (2013).
- Hickman, J. A., et al. Three-dimensional models of cancer for pharmacology and cancer cell biology: Capturing tumor complexity in vitro/ex vivo. Biotechnology Journal. 9 (9), 1115-1128 (2014).
- Sutherland, R. M. Cell and environment interactions in tumor microregions: the multicell spheroid model. Science. 240 (4849), 177-184 (1988).
- Mueller-Klieser, W. Three-dimensional cell cultures: from molecular mechanisms to clinical applications. American Journal of Physiology - Cell Physiology. 273, C1109-C1123 (1997).
- Friedrich, J., Seidel, C., Ebner, R., Kunz-Schughart, L. A. Spheroid-based drug screen: considerations and practical approach. Nature Protocols. 4 (3), 309-324 (2009).
- Tung, Y. -. C., et al. High-throughput 3D spheroid culture and drug testing using a 384 hanging drop array. The Analyst. 136 (3), 473-478 (2011).
- Vinci, M., et al. Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC biology. 10, 29 (2012).
- LaBarbera, D. V., Reid, B. G., Yoo, B. H. The multicellular tumor spheroid model for high-throughput cancer drug discovery. Expert Opinion on Drug Discovery. 7, 819-830 (2012).
- Pampaloni, F., Ansari, N., Stelzer, E. H. K. High-resolution deep imaging of live cellular spheroids with light-sheet-based fluorescence microscopy. Cell and Tissue Research. 352, 161-177 (2013).
- Lovitt, C. J., Shelper, T. B., Avery, V. M. Miniaturized three-dimensional cancer model for drug evaluation. Assay and Drug Development Technologies. 11 (7), 435-448 (2013).
- Wenzel, C., et al. 3D high-content screening for the identification of compounds that target cells in dormant tumor spheroid regions. Experimental Cell Research. 323 (1), 131-143 (2014).
- Astashkina, A., Grainger, D. W. Critical analysis of 3-D organoid in vitro cell culture models for high-throughput drug candidate toxicity assessments. Innovative tissue models for drug discovery and development. 69, 1-18 (2014).
- Edmondson, R., Broglie, J. J., Adcock, A. F., Yang, L. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay and Drug Development Technologies. 12 (4), 207-218 (2014).
- Gong, X., et al. Generation of multicellular tumor spheroids with microwell-based agarose scaffolds for drug testing. PLoS ONE. 10 (6), e0130348 (2015).
- Hoffmann, O. I., et al. Impact of the spheroid model complexity on drug response. Journal of biotechnology. 205, 14-23 (2015).
- Martinez, N. J., Titus, S. A., Wagner, A. K., Simeonov, A. High-throughput fluorescence imaging approaches for drug discovery using in vitroand in vivothree-dimensional models. Expert Opinion on Drug Discovery. 10, 1347-1361 (2015).
- Nath, S., Devi, G. R. Three-dimensional culture systems in cancer research: Focus on tumor spheroid model. Pharmacology, Therapeutics. 163, 94-108 (2016).
- Li, L., Zhou, Q., Voss, T. C., Quick, K. L., LaBarbera, D. V. High-throughput imaging: Focusing in on drug discovery in 3D. Methods. 96, 97-102 (2016).
- Ham, S. L., Joshi, R., Thakuri, P. S., Tavana, H. Liquid-based three-dimensional tumor models for cancer research and drug discovery. Experimental Biology and Medicine. 241 (9), 939-954 (2016).
- Kessel, S., et al. High-Throughput 3D Tumor Spheroid Screening Method for Cancer Drug Discovery Using Celigo Image Cytometry. Journal of Laboratory Automation. , 2211068216652846 (2016).
- Stock, K., et al. Capturing tumor complexity in vitro: Comparative analysis of 2D and 3D tumor models for drug discovery. Scientific Reports. 6, 28951 (2016).
- Thakuri, P. S., Ham, S. L., Luker, G. D., Tavana, H. Multiparametric analysis of oncology drug screening with aqueous two-phase tumor spheroids. Molecular Pharmaceutics. 13 (11), 3724-3735 (2016).
- Lin, R. Z., Chang, H. Y. Recent advances in three-dimensional multicellular spheroid culture for biomedical research. Biotechnology Journal. 3 (9-10), 1172-1184 (2008).
- Piccinini, F., Tesei, A., Arienti, C., Bevilacqua, A. Cancer multicellular spheroids: Volume assessment from a single 2D projection. Computer Methods and Programs in Biomedicine. 118 (2), 95-106 (2015).
- Zanoni, M., et al. 3D tumor spheroid models for in vitro therapeutic screening: a systematic approach to enhance the biological relevance of data obtained. Scientific Reports. 6, 19103 (2016).
- Debnath, J., Brugge, J. S. Modelling glandular epithelial cancers in three-dimensional cultures. Nature Reviews Cancer. 5 (9), 675-688 (2005).
- Huang, D., et al. Optical coherence tomography. Science. 254 (5035), 1178-1181 (1991).
- Drexler, W., et al. Optical coherence tomography today: speed, contrast, and multimodality. Journal of Biomedical Optics. 19 (7), 071412 (2014).
- Fujimoto, J., Swanson, E. The development, commercialization, and impact of optical coherence tomography. Investigative Ophthalmology, Visual Science. 57 (9), (2016).
- Vakoc, B. J., Fukumura, D., Jain, R. K., Bouma, B. E. Cancer imaging by optical coherence tomography: preclinical progress and clinical potential. Nature Reviews Cancer. 12 (5), 363-368 (2012).
- Wojtkowski, M. High-speed optical coherence tomography: basics and applications. Applied optics. 49 (16), D30-D61 (2010).
- Drexler, W., Fujimoto, J. G. . Optical coherence tomography: technology and applications. , (2008).
- Geitzenauer, W., Hitzenberger, C. K., Schmidt-Erfurth, U. M. Retinal optical coherence tomography: past, present and future perspectives. British Journal of Ophthalmology. 95 (2), 171 (2011).
- Sakata, L. M., DeLeon-Ortega, J., Sakata, V., Girkin, C. A. Optical coherence tomography of the retina and optic nerve - a review. Clinical, Experimental Ophthalmology. 37 (1), 90-99 (2009).
- van Velthoven, M. E. J., Faber, D. J., Verbraak, F. D., van Leeuwen, T. G., de Smet, M. D. Recent developments in optical coherence tomography for imaging the retina. Progress in Retinal and Eye Research. 26 (1), 57-77 (2007).
- Kashani, A. H., et al. Optical coherence tomography angiography: A comprehensive review of current methods and clinical applications. Progress in Retinal and Eye Research. 60, 66-100 (2017).
- de Carlo, T. E., Romano, A., Waheed, N. K., Duker, J. S. A review of optical coherence tomography angiography (OCTA). International Journal of Retina and Vitreous. 1 (1), 5 (2015).
- Sharma, M., Verma, Y., Rao, K. D., Nair, R., Gupta, P. K. Imaging growth dynamics of tumour spheroids using optical coherence tomography. Biotechnology Letters. 29 (2), 273-278 (2006).
- Jung, Y., Nichols, A. J., Klein, O. J., Roussakis, E., Evans, C. L. Label-Free, Longitudinal Visualization of PDT Response In Vitro with Optical Coherence Tomography. Israel Journal of Chemistry. 52 (8-9), 728-744 (2012).
- Huang, Y., et al. Optical coherence tomography detects necrotic regions and volumetrically quantifies multicellular tumor spheroids. Cancer Research. 77 (21), 6011-6020 (2017).
- Spalteholz, W. . Über das Durchsightigmachen von menschlichen und tierischen Präparaten: nebst Anhang, Über Knochenfärbung. , (1911).
- Dodt, H. -. U., et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nature Methods. 4 (4), 331 (2007).
- Leitgeb, R., Hitzenberger, C., Fercher, A. F. Performance of fourier domain vs. time domain optical coherence tomography. Optics express. 11 (8), 889-894 (2003).
- Jian, Y., Wong, K., Sarunic, M. V. . Optical Coherence Tomography and Coherence Domain Optical Methods in Biomedicine XVII. , 85710Z (2013).
- Guizar-Sicairos, M., Thurman, S. T., Fienup, J. R. Efficient subpixel image registration algorithms. Optics Letters. 33 (2), 156-158 (2008).
- Canny, J. A computational approach to edge detection. IEEE Transactions on Pattern Analysis and Machine Intelligence. (6), 679-698 (1986).
- Vermeer, K. A., Mo, J., Weda, J. J. A., Lemij, H. G., de Boer, J. F. Depth-resolved model-based reconstruction of attenuation coefficients in optical coherence tomography. Biomedical Optics Express. 5 (1), 322-337 (2014).
- Klein, T., et al. Multi-MHz retinal OCT. Biomedical Optics Express. 4, 1890-1908 (2013).
- Klein, T., Huber, R. High-speed OCT light sources and systems [Invited]. Biomedical Optics Express. 8 (2), 828-859 (2017).
- Zhou, C., Alex, A., Rasakanthan, J., Ma, Y. Space-division multiplexing optical coherence tomography. Optics Express. 21, 19219-19227 (2013).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved