Method Article
Multifactorial Assessment of Motor Behavior in Rats after Unilateral Sciatic Nerve Crush Injury
In This Article
Summary
We provide a protocol for the assessment of motor behavior via a behavioral test battery in rats after sciatic nerve crush injury.
Abstract
The induction of a peripheral nerve injury is a widely used method in neuroscience for the assessment of repair and pain mechanisms among others. In addition, in the research field of movement disorders, sciatic crush injury has been employed to trigger a dystonia-like phenotype in genetically predisposed DYT-TOR1A rodent models of dystonia. To achieve consistent, reproducible and comparable results after a sciatic nerve crush injury, a standardized method for inducing the nerve crush is essential, in addition to a standardized phenotypical characterization. Attention must be paid not only to the specific assortment of behavioral tests, but also to the technical requirements, the correct execution and consecutive data analysis. This protocol describes in detail how to perform a sciatic nerve crush injury and provides a behavioral test battery for the assessment of motor deficits in rats that includes the open field test, the CatWalk XT gait analysis, the beam walking task, and the ladder rung walking task.
Introduction
Rodents are excellent model organisms to deepen the understanding of human diseases1,2 by testing hypotheses on multiple biological levels. One fundamental biological level for the characterization of rodent models is the phenotype level, measured by behavioral assessments. Depending on the animal model and the scientific research question, the selection of a powerful and reliable behavioral test battery is essential to cover a wide range of behavioral aspects such as for animal models of Parkinson's disease and dystonia3,4,5,6.
The sciatic nerve is the largest nerve in the human body with motor as well as sensory fibers. Injuries of the sciatic nerve can result easily from a variety of events such as traffic accidents and surgeries7,8. Therefore, research activities using rodent models with sciatic nerve injuries, are of translationally relevant value. Even though the translational aspect of nerve regeneration from rat to human has to be regarded critically9, the sciatic nerve crush injury (axonotmesis) in rodent models is a commonly used method to analyze degeneration and regeneration processes of peripheral nerves10,11. In case of a crush injury the nerve is not completely transected. It damages the axon, resulting in conduction block directly after crush injury followed by regenerative processes 4,12,13.
Moreover, in dystonia research, the unilateral sciatic nerve crush injury is an established method to trigger dystonia-like movements (DLM) in genetically predisposed dystonia rodent models, which do not show DLM per se4,14. It is assumed that the peripheral nerve trauma disturbs the sensorimotor integration by affecting the sciatic nerve fibers, which are responsible for motor and sensory functions15.
We here provide a detailed description for a standardized crush injury of the sciatic nerve and a battery of motor behavior assessments that is composed of the open field test (OFT), CatWalk XT gait analysis, beam walking task and ladder rung walking task in naïve wild type (wt) rats (n= 8-9) and wt rats five weeks after unilateral sciatic nerve crush injury (n= 10). The OFT provides information about the general locomotor activity, while a detailed gait analysis is achieved by the automated gait analysis system CatWalk XT. The beam walking task is used to assess the motor coordination by evaluating the time to cross the beam and the number of foot placement errors. For gait performance analysis the ladder rung walking task gives information about foot or paw placement and errors on a horizontal ladder rung apparatus with a constant but irregular rung pattern.
Protocol
All animal experiments were approved by the local authorities at the Regierung von Unterfranken (Würzburg, Germany) and performed according to applicable international, national, and/or institutional guidelines for care and use of animals.
1. Sciatic nerve crush injury
NOTE: Maintain a sterile environment during the whole surgical procedure. Set up the surgery table with the necessary equipment.
- Deeply anesthetize the rat in a closed cabinet with isoflurane 3.0% in O2 (2 L/min). Remove the rat from the cabinet. Shave an extensive area of the right hindlimb.
- Position the rat into the anesthesia mask and continue deep anesthesia with isoflurane 2.0% in O2 (2 L/min). Control the anesthesia depth by pinching the interdigital webbing of the hind feet. The absence of withdrawal reflexes indicates an adequate anesthesia.
- Fix the torso and both hindlimbs of the rat with tape. Place both hindlimbs in a symmetrical and outstretched position by turning the paw flat onto the surgery table.
- Apply ophthalmic ointment on eyes to prevent dry eyes. Disinfect the skin of the shaved area with an antiseptic.
- Search for the sciatic notch of the ilium.
- Make a skin incision from the sciatic notch into the direction of the paw with a scalpel. The skin incision should be as small as possible (approximately 1 to 2 cm).
- If the hindlimbs are fixed and the skin incision is performed correctly, a cavity in the fascial plane between the gluteus maximus muscle and biceps femoris muscle can be seen that resembles a "white line". Insert closed super-fine hemostatic forceps (No. 5) into the cavity and spread the forceps. The fascial plane should open up without injuring any muscle tissue.
- Place the rubber band retractors underneath the muscles, to keep the skin incision open.
- Gently remove any surrounding tissue and blood vessels from the sciatic nerve until the nerve is completely exposed. It is important to not stretch or pull the nerve during the whole procedure.
- Crush the sciatic nerve with a non-serrated clamp (ultra-fine hemostat) with constant and reproducible pressure. For this, open the clamp, place the nerve onto the bottom jaw of the clamp and close the clamp by locking it into the first position for three times ten seconds. The position of the sciatic nerve crush is located close to the sciatic notch, proximal to the division site of the main sciatic nerve bundle. After the crush injury, reopen the clamp carefully. The crush site of the sciatic nerve appears translucent.
- Remove the rubber band retractors.
- Close the fascial plane incision with resorbable 4-0 suture. Close the skin incision with body skin staples.
- Apply Rimadyl according to the GV-SOLAS guidelines (5 mg/kg body weight, subcutaneous injection) for postoperative pain relief every 24 hours after surgery for two days.
- Remove the rat from the surgery setup. Place the rat into a clean cage without bedding on a heating plate (37 °C) until the rat is awake. Move the rat back into their clean home cage.
- Remove body skin staples four to six days after surgery.
2. Open field test (OFT)
NOTE: Locomotor activity as well as behavioral activity can be analyzed by the OFT.
- Setup
- Set up the OFT (Figure 1A) in a dark and quiet environment. It consists of the automated video tracking system EthoVision XT (computer, software with license) and an arena measuring 58.5 cm (length) x 58.5 cm (width) x 45 cm (height) with a scratch resistant, cleanable black surface. The black surface is important to increase the contrast when tracking white animals.
- Assessment
- Place the arena and the camera into the correct position. Adjust the camera that the entire open field box with the best resolution is recorded. Perform the experiment in a dark environment. If light is needed for the setup, use a small and diffuse light to avoid light spots, reflections and shades in the arena. Ensure equal light conditions by measuring the illuminance with a lux meter in different areas of the arena.
- Set up the EthoVision XT software. The most important settings are listed in the following. In the Experiment Settings choose Live tracking for the Video Source and the Center point detection for the Tracked Features. Validate the size of the arena in the Arena Settings. Set the start condition for data acquisition to three seconds after the rat was placed into the middle of the arena and the total run time to five minutes in Trial Control Settings. Choose Static subtraction for the Method in Detection Settings. Checkmark Save video for Method in Acquisition Settings.
- Place the rat gently in the middle of the testing arena (Figure 1B).
- Press the Start Trial button in Acquisition Control to start the recording.
- During the recording, stay away from the OFT setup in order to avoid distracting the rat.
- After each trial, remove the rat gently from the testing arena and clean the setup with 0.1% acetic acid to avoid distraction by the smell of the previously recorded rat.
- Data analysis
- For the data analysis of the OFT with the EthoVision XT software, go to the Analysis section in the left side bar and choose Track Visualization under the Results tab (Figure 1C). Next, export the needed parameters to Excel. Within the software, choose a number of variables from different categories for data analysis. Important variables for this specific scientific objective are "Distance moved" and "Velocity" under the category "Distance and Time". Perform a statistical analysis of the selected parameters (Figure 1D).
3. CatWalk XT gait analysis
NOTE: A gait analysis via the CatWalk XT system can help to assess many different parameters concerning the footprints, stance and gait of animal models. A glass walkway is illuminated with green light and the light scattered by the footprints of the animals is captured with a high-speed video camera, which is located underneath the walkway. The signals can be analyzed with the CatWalk XT software.
- Setup
- For gait analysis with the CatWalk XT, use the CatWalk system and the corresponding software (computer, software with license) (Figure 2A).
- Perform the experiment under dark conditions, because data acquisition depends on the illumination of the walkway of the CatWalk system with green LED light. To facilitate the experimental procedure under dark conditions, illuminate the experimental room with red light.
- Use a defined walkway that measures 65 cm in length and 7 cm in width; however, the size of the walkway depends on the size of the rats. Set up the walkway as large as possible to record as many footprints as possible for each paw.
- Capture a minimum number of three footprints per paw for each run. When defining the length of the walkway, consider the rat's body and tail, as the start or stop signal might not be detected correctly and the runs may not be classified as compliant, if the body/tail enters or remains on the defined walkway before or after completion of the run.
- Training
NOTE: Training the rats for the CatWalk system is necessary to habituate the animals to the setup and allow them to learn to cross the walkway without any interruption. Proper training provides the advantages of saving time during experimental assessment and obtaining better results. Starting the data acquisition of the CatWalk system during training sessions allows the rats to get used to the assessment conditions (noise/light).- Start to set up the CatWalk system.
- Clean the glass walkway with distilled water and a lint-free soft cloth. At the beginning and at the end of the experiment, or in between if the glass walkway is dirty, use glass-cleaning fluid and lint-free soft cloth to clean the glass walkway. After the use of glass cleaning fluid, clear the walkway from any residues of the fluid to avoid it distracting the animal.
- Choose the experimental settings. An important parameter is Run Criteria. Set appropriate values for Minimum run duration, Maximum run duration, and Minimum number of compliant runs to acquire, which are specific for every research project. Checkmark the box of Use maximum allowed speed variation and set the value. The Run criteria can be ignored for the first four to five days of training.
- Place the camera in position and adjust the focus. Find the optimal camera position to achieve an appropriate length of the walkway and the best resolution of the recorded paws simultaneously. Label the camera position on the CatWalk system to ensure identical camera placement between recordings.
- Set up the detection settings by using the auto-detection for a new experiment. Be sure that all footprints can be detected with minimal background noise . If necessary, optimize the detection settings manually and change the Green Intensity Threshold. Use the same detection settings for the entire experiment.
- Set up the corridor walls of the CatWalk system. The corridor walls should be as close as possible to the rat. Make sure the corridor walls remain parallel to the walkway.
- Define the length of the walkway: Click the Define Walkway icon. Adjust the size of the white rectangle in length and width, according to the specific research project. Click OK.
- Calibrate the walkway: Click the Calibrate Walkway icon. Position a rectangular calibration sheet measuring 20 x 10 cm in the middle of the walkway. Adapt the size of the white rectangle to the calibration sheet. Click OK.
- Next, snap a background image: Check beforehand that the walkway is clean and empty. Click the Snap Background button to generate a background image.
- Train animals for at least eight days before starting the actual experiment. The training on successive days is recommended.
- Day 1 of training: For rats to get used to the CatWalk system, allow the animal to freely explore the walkway and the goal box. Let the rats practice crossing the walkway and enter the goal box. Pick up the rat at the end of the walkway or in the goal box and bring the rat back to the starting point of the walkway. Five runs are recommended for the first day of training without need for compliance to the experimental settings.
- Day 2 of training: Rats can freely explore the walkway and the goal box. Five runs are recommended without compliance to the experimental settings.
- Day 3 of training: Eight runs are recommended without compliance to the experimental settings.
- Day 4 of training: Ten runs are recommended without compliance to the experimental settings.
- Day 5 of training: Ten runs are recommended. The experimental settings should be kept in mind. Motivate the rats to cross the walkway without any interruption.
- Day 6 of training: Ten runs are recommended. The experimental settings should be kept in mind. Motivate the rats to cross the walkway without any interruption.
- Day 7 of training: Ten runs are recommended. A minimum of three compliant runs should be achieved. Add more runs for animals, if they were unable to reach this goal.
- Day 8 of training: Ten runs are recommended. A minimum of three compliant runs should be achieved. Add more runs for animals, if they were unable to reach this goal.
- Start to set up the CatWalk system.
- Assessment
- According to the defined run criteria, perform three compliant runs per rat for data analysis. For the assessment, please follow steps 3.2.1. - 3.2.1.8. as described in the training section. Even if the rat reaches three compliant runs within the first three runs, carry out a minimum of six runs per session/per week for training purposes.
- Perform at least one (training) session with six runs per week for a stable gait pattern for experiments with multiple time points. Experimental and detection settings stay consistent for the whole experiment.
- Data analysis
- For data analysis, only evaluate the compliant runs. Delete non-compliant runs.
- Verify the Green Intensity Threshold and increase or decrease Green Intensity Threshold before classification of the paw prints if necessary. The Green Intensity Threshold must be consistent for all animals and all runs.
- Classify paw prints automatically with the CatWalk XT software (Figure 2B).
- Review paw print labels manually. Correct wrong paw print labels, add labels of non-detected paw prints, and delete noise and wrong labels manually. Move the video to a position, which has to be reviewed manually. For correction of wrong labeled paw prints, select the rectangle of the specific paw print, click Reset, select the same rectangle again, and assign the correct label from the list. For labeling non-detected paw prints, draw a rectangle around the non-detected paw, click Add Print, select the new generated rectangle, and assign the correct label from the list. In case the software labeled nose or body prints automatically, select the rectangle of the specific label, and click Remove Print.
- Review the numerical results. The numerical results are displayed in an excel sheet, showing a number of basic parameters. Choose pre-defined specific parameters, depending on the research interest and perform statistical analysis as usual (Figure 2D).
- For more detailed information about each footprint, classify the toes of the hind paws. This analysis requires the Interactive Footprint Measurements module.
- Adapt the Green Intensity Threshold for the Interactive Footprint Measurements analysis if necessary. The Green Intensity Threshold must be consistent for all animals and all runs.
- Set the markers for the footprint analysis manually. Analyze every hind paw print in all three compliant runs. Draw a line from the center of the first toe to the center of the fifth toe to measure "Toe Spread". Draw a line from the center of the second toe to the center of the fourth toe to measure "Intermediate Toe Spread". Draw a line from the center of the third toe to the heel of the hind paw to measure "Manual Print Length" (Figure 2C).
- Review the numerical results of the "Interactive Footprint Measurements" displayed in a separate sheet. Select specific parameters of the "Interactive Footprint Measurements" and perform statistical analysis as usual (Figure 2E).
4. Beam walking task
NOTE: Gait deficits can be determined by the beam walking task. The focus of the beam walking task in this specific research topic will be the analysis of motor coordination, defined as the ability to coordinate muscle activation from multiple body parts, and not the assessment of motor balance, defined as the ability for postural control during body movements.
- Setup
- For the beam walking task, use a beam, spacer, table, uniform background and a camcorder (Figure 3A).
- Use a wooden beam of 90 cm in length, 1.7 cm in width and 2 cm in height. A platform of 20.5 cm in length, 15 cm in width and 2 cm in height at both ends of the beam is recommended. Use the same material for the platforms and the beam, avoid any barriers between the platforms and the beam.
- Have the distance between the beam and the table be at least 44 cm. A familiar environment such as a home cage motivates the rats to cross the beam, which can be placed at the end of the beam platform.
- Training
- Set up the beam with spacer and home cage on the table.
- Train animals for seven days. The training on successive days is recommended.
- Day 1 of training
- Place all rats from one home cage on the start platform of the beam.
- Let the rats explore the environment (platform/beam).
- Hold one rat carefully by the tail and lead the rat to the beam by pushing the rat gently onto the beam.
- Assist the rat in traversing the beam by holding the rat by its tail for at least two runs.
- Let the rat traverse the beam for three more runs without assistance. Observe the rat and provide assistance if necessary. If the rat fails to traverse the beam, intercept the fall in order to avoid injuries and the development of fear to traverse the beam.
- Continue with this procedure for all rats.
NOTE: Sometimes rats follow each other to traverse the beam, in which case no assistance is needed. However, it is important to observe the rats, intercept falls and to provide assistance if necessary.
- Day 2 of training
- Place all rats from one home cage on the start platform of the beam.
- Let the rats traverse the beam six times.
- If necessary, provide assistance and intercept falls.
- Day 3 of training
- Place one rat on the start platform of the beam.
- Let the rat traverse the beam six times.
- If necessary, provide assistance and intercept falls.
- Day 4-7 of training
- Place one rat on the start platform of the beam.
- Let the rat traverse the beam ten times.
- If necessary, provide assistance and intercept falls.
- At the end of the training, the rat should traverse the beam without any interruption for at least three runs. It is permissible to gently push the rat on the start platform to trigger initiation of movement.
- Day 1 of training
- Assessment
- Set up the beam with spacer and home cage on the table.
- Place the camcorder in position, aligned in parallel to the beam with the animal in focus. The position of the camcorder should be as close as possible to the animal to achieve optimal resolution of the recorded movements. The beam and parts of both platforms should be captured by the recording.
- Start the recording and first identify the session and the animal.
- Place the rat on the start platform of the beam.
- The rat should cross the beam three times without any interruption. Even if the rat reaches three compliant runs within the first three runs, carry out a minimum of six to ten runs for continuous task performance.
- Always, observe the animal and intercept falls, if needed.
- After the task, clean the beam and the table with 0.1% acetic acid to avoid distraction by the smell of the previously recorded rat.
NOTE: Within the first two weeks after nerve crush injury, the rats are unable to traverse the beam without assistance. Therefore, assistance has to be provided for six to eight runs in the first two weeks after nerve crush injury. From week three to week five, five runs are performed with assistance and ten further runs were performed without assistance.
- Data analysis
- Use the free video analysis software Kinovea for data analysis.
- Select the video sequences of three compliant runs from the recording. For this, choose the first three compliant runs that were performed without assistance by the animal. Be consistent in the compliant run selection for all rats.
- Define the start time point and the end time point of the selected three compliant runs (Figure 3D-E). In this setup, the start point was labelled by a black line on the beam and the placement of the first hindlimb behind the black line defined the start time point of the run. The placement of the first hindlimb on the platform at the end of the beam defines the end time point.
- Next, calculate the time needed for the rat to traverse the beam. Report data as latency time to cross the beam in seconds and perform statistical analysis as usual (Figure 3B).
- Score the number of steps and errors from three compliant runs for both hindlimbs separately by using the zoom and slow motion function of the software. Errors include total foot slips and half foot slips. A total foot slip is defined as a foot placement that is followed by a deep slip causing a loss of contact of the affected paw with the beam (Figure 3F). A half slip is defined as a paw sliding off the sidewall of the beam without losing complete contact with the beam (Figure 3G).
- Calculate the percentage of foot slips in relation to the number of steps to cross the beam ((number of foot slips of the limb x 100%)/ number of steps of the same limb). Present data as percentage foot slips and perform statistical analysis as usual (Figure 3C).
5. Ladder rung walking task
NOTE: The ladder rung walking task can assess motor function, placement of both frontlimbs and hindlimbs, and interlimb coordination.
- Setup
- Use a ladder rung apparatus, spacer, table, uniform background and camcorder for this behavioral test (Figure 4A). The horizontal ladder rung apparatus consists of metal rungs and clear polycarbonate sidewalls. The apparatus has a length of 119.5 cm and the width is adjusted to 7.4 cm. The walkway to be analyzed has a length of 100 cm.
- Label the start and end point with a black line on the sidewall. Place placeholders for the rungs on the apparatus at 1 cm intervals. Arrange an irregular pattern of the rungs for the 100 cm walkway with a distance between 1 and 5 cm between the rungs. The first 10 cm at the beginning and the last 9.5 cm at the end of the apparatus, which are excluded from analysis, have a regular pattern of the rungs with a distance of 1 cm.
- Use a distance between the walkway and the table of approximately 30 cm (Figure 4A-B). A goal box or a familiar environment at the end of the apparatus, like a home cage, motivates the rats to cross the ladder rung apparatus.
- Training
- Set up the ladder rung apparatus with spacer and goal box on the table.
- Train animals for eight days. The training on successive days is recommended.
- Day 1 of training
- Place all rats from one home cage on the ladder rung apparatus.
- Let the rats explore the environment (ladder rung apparatus/goal box).
- Gently push the rats into the direction of the goal box. Assist the rats in entering the goal box. Let the rats explore the goal box for a while.
- After all rats entered the goal box. Take the first rat from the goal box and place the rat on the start zone of the apparatus again. Continue with the same procedure for all rats of one home cage. Gently push the rats into the direction of the goal box and give assistance to enter the goal box, if needed.
- Let the rat traverse the apparatus four times.
- Day 2 of training
- Perform the same protocol as listed for first day of training.
- Let the rat traverse the apparatus six times.
- Day 3 of training
- Perform the same protocol as listed for first day of training.
- Let the rat traverse the apparatus eight times.
- Day 4 of training
- Place one rat on the start of the ladder rung apparatus.
- If the rat does not traverse the apparatus and enters the goal box voluntarily, give assistance by gently pushing the rat from behind.
- Let the rat traverse the apparatus eight times.
- Day 5-8 of training
- Place one rat on the start of ladder rung apparatus.
- If the rat does not traverse the apparatus and enters the goal box voluntarily, give assistance by gently pushing the rat from behind.
- Let the rat traverse the apparatus ten times.
- At the end of the training, the rat should be able to traverse the walkway without any interruption and assistance for a minimum of three runs. It is permissible to give the rat a gentle push in the start zone to trigger initiation of movement.
- Day 1 of training
- Assessment
- Set up the ladder rung apparatus with spacer and goal box on the table.
- Place the camcorder in position, aligned in parallel to the apparatus with the animal in focus. Position the camcorder as close as possible to the animal to achieve optimal resolution of the recorded movements and ensure that the entire ladder rung apparatus is captured in the recording.
- Start the recording and first identify the session and the animal.
- Place the rat on the start zone of the ladder rung apparatus.
- The rat has to traverse the 100 cm walkway of the ladder rung apparatus three times without any interruption in order for it to qualify as a compliant run. Even if the rat reaches three compliant runs within the first three runs, a minimum of ten runs should be carried out for continuous task performance.
- After the task, clean the apparatus and the table with 0.1% acetic acid to avoid distraction by the smell of the previously recorded rat.
- Data analysis
- Use the free video analysis software Kinovea for data analysis.
- Select the video sequences of three compliant runs from the recording. Choose the first three compliant runs for data analysis.
- Define the start time point and the end time point of the selected three compliant runs. The placement of the first hindlimb behind the first black line on the sidewall of the apparatus, which labels the start time point of the 100 cm walkway, defines the start time point of the run. The placement of the first frontlimb behind the second black line on the sidewall of the apparatus, which labels the end point of the 100 cm walkway, defines the end time point of the run.
- Identify the start and end time point. Next, calculate the duration of the run across the walkway. Report data as latency time to traverse the walkway in seconds and perform statistical analysis as usual (Figure 4C).
- Score the three compliant runs with the 7-category scale from Metz et al. by using the slow motion or the frame-by-frame function of the software (Figure 5)16,17. Determine the number of steps and the number of errors in accordance with the categories of the scale for each limb separately. The scale distinguishes between the following categories: (0) total miss (1) deep slip (2) slight slip (3) replacement (4) correction (5) partial placement, and (6) correct placement. Only the error of the initiating limb was rated. Further errors, triggered by the initial error, should not be rated.
- Calculate the errors/step by considering the following requirements. The categories (0) total miss (1) deep slip (2) slight slip count as an error. Divide the number of errors by the number of steps for each hindlimb and each run separately. Determine the mean value of all three compliant runs for each animal and each hindlimb separately and perform statistical analysis as usual (Figure 4D).
Results
The representative results of the five minutes OFT show that the nerve crush injury five weeks post surgery has no effect on the locomotor activity (Figure 1).
Gait analysis with the CatWalk XT system (Figure 2) generates many different parameters. Selective parameters were statistically analyzed by comparing wt naïve rats with nerve-injured wt rats five weeks after the nerve crush (Figure 2D). Significant alterations could be detected for the run average speed, the stride length and the print area of the nerve-injured (right) hind paw. A more detailed analysis of the nerve-injured hind paw was performed with the "Interactive Footprint Measurements" module. A significant reduction of the parameters toe spread, intermediate toe spread and print length were observed in nerve-injured wt rats compared to wt naïve rats. In addition, the paw angle body axis and the paw angle movement vector significantly differ when comparing nerve-injured wt rats with wt naïve rats (Figure 2E).
Figure 3 presents data of motor coordination obtained through beam walking task assessment. Nerve-injured wt rats showed a significantly increased latency time to cross the beam compared to wt naïve rats five weeks post injury (Figure 3B). As an additional read out of the beam walking task, full slips and half slips of the nerve-injured hindlimb were counted and considered as an error for statistical analysis. The percentage of errors per step of the nerve-injured (right) hindlimb was significantly increased in nerve-injured wt rats compared to wt naïve rats.
Representative data of the ladder rung walking task (Figure 4) does not show significant alterations in the latency time to cross the walkway of the ladder rung apparatus (Figure 4C) or in the percentage of errors per step of the nerve-injured (right) hindlimb (Figure 4D). The analysis of the error percentage per step of the nerve-injured hindlimb considered only the score from 0 to 2 of the 7-category scale from Metz et al. The distribution of all score categories per step from the 7-category scale of the nerve-injured hindlimb and the non-nerve-injured (left) hindlimb is illustrated in Figure 4E.
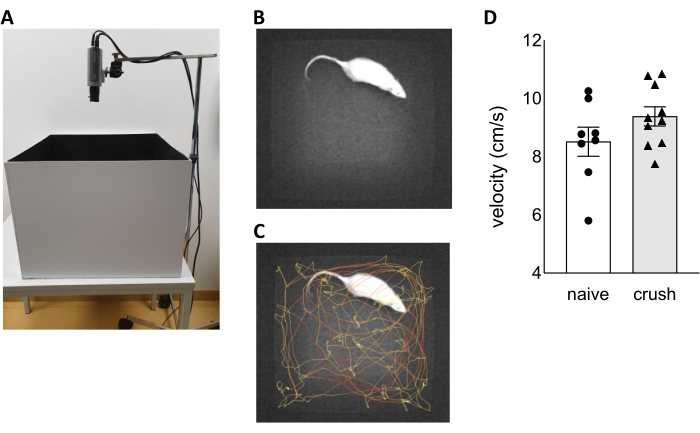
Figure 1: Assessment of locomotor activity during open field test. (A) Picture of the open field test setup. Selected picture subtracted from a recorded video during open field test showing a rat in the open field arena without (B) and with (C) tracking. (D) The velocity during a five minutes open field test recording was investigated in wt naïve rats and wt rats five weeks after nerve crush injury. Data are shown as mean ± SEM. Statistical analysis was performed using the unpaired t test of the normally distributed data. Please click here to view a larger version of this figure.
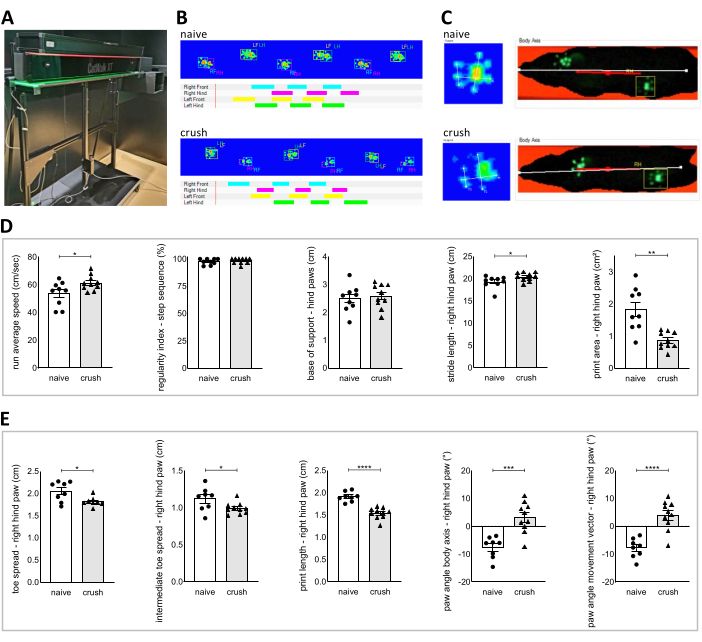
Figure 2: Gait analysis with the CatWalk XT system. (A) Picture of the CatWalk XT apparatus. (B) Examples of the print view showing the labeled paw prints in false color mode and examples of the timing view showing time-based gait diagram of wt naïve rats and wt rats five weeks after nerve crush injury. (C) Examples of the toe classification showing the toe spread (TS), intermediate toe spread (ITS) and print length (PL) as well as examples of the body axis view showing the body axis (white line) and the movement vector (red line) of wt naïve rats and wt rats five weeks after nerve crush injury. (D) Data of selected parameters from the "standard" classification comparing wt naïve rats and wt rats five weeks after nerve crush injury. (E) Data of selected parameters from the "Interactive Footprint Measurements module" comparing wt naïve rats and wt rats five weeks after nerve crush injury. Data are shown as mean ± SEM. Statistical analysis was performed using the unpaired t test of the normally distributed data, unpaired t test with Welch's correction of normally distributed data with unequal variance and Mann-Whitney U test of the non-normal distributed data. P-value < 0.05 was defined as statistically significant labeled as *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Please click here to view a larger version of this figure.

Figure 3: Gait analysis with the beam walking task. (A) Picture and schematic drawings of the beam walking task setup. The latency time to cross the beam (B) and the percentage foot slip errors per step of the nerve-injured hindlimb during the beam walking task (C) was analyzed in wt naïve rats and wt rats five weeks after nerve crush injury. Representative picture for the start time position (D) and the end time position (E) of the beam walking task. Representative image sequence of a full slip error (F) and a half slip error (G) of the beam walking task. Data are shown as mean ± SEM. Statistical analysis was performed using the Mann-Whitney U test of the non-normal distributed data. P-value < 0.05 was defined as statistically significant labeled as *p < 0.05, **p < 0.01. Please click here to view a larger version of this figure.
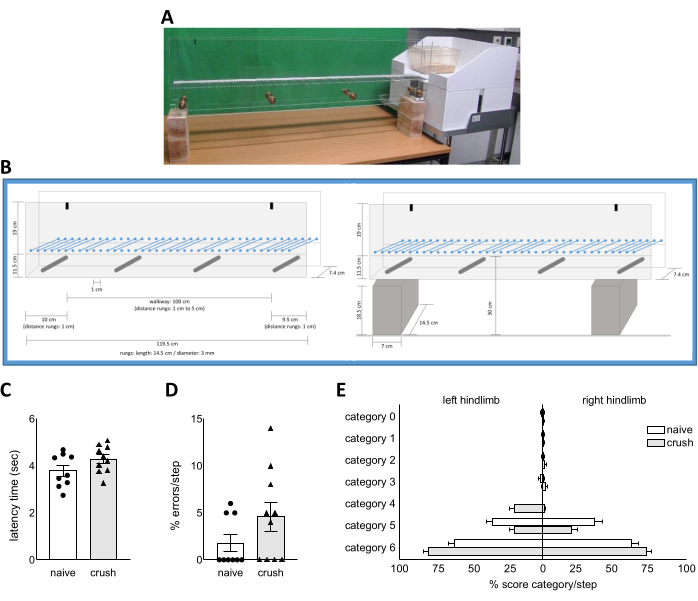
Figure 4: Gait analysis using the ladder rung walking task. Picture (A) and schematic drawings (B) of the ladder rung walking task setup. The latency time of traversing the ladder rung apparatus (C) and the percentage foot slip errors per step of the nerve-injured hindlimb during the ladder rung walking task (D) was assessed in wt naïve rats and wt rats five weeks after nerve crush injury. (E) The percentage distribution of the score category per step according to the 7-category scale from Metz et al. for the left and right hindlimb of wt naïve rats and wt rats five weeks after nerve crush injury. Data are shown as mean ± SEM. Statistical analysis was performed using the unpaired t test of the normally distributed data and Mann-Whitney U test of the non-normal distributed data. Please click here to view a larger version of this figure.
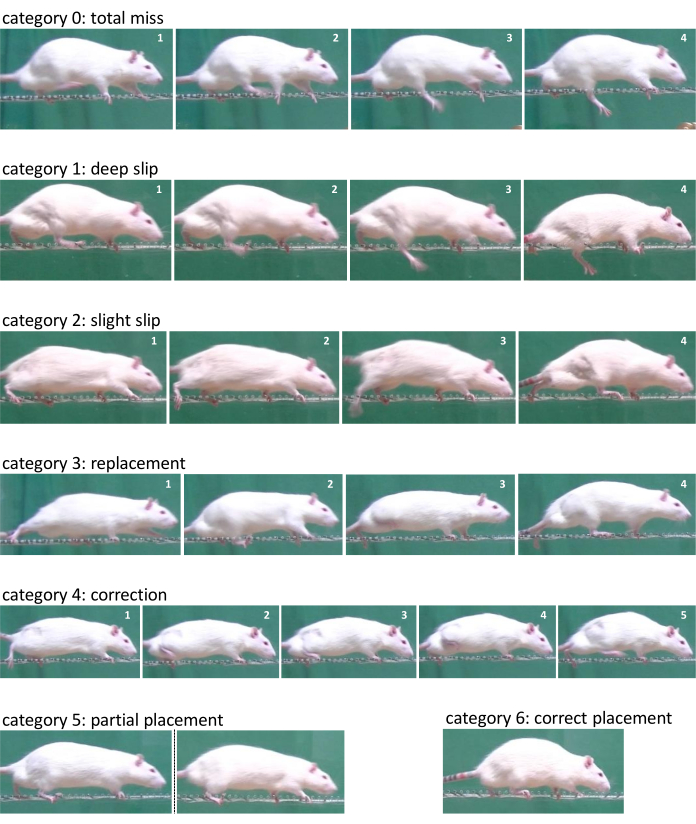
Figure 5: Exemplary representation of each category according to the 7-category scale from Metz et al. during the ladder rung walking task. Representative image sequence from the right hindlimb of category 0 - total miss, category 1 - deep slip, category 2 - slight slip, category 3 - replacement, category 4 - correction, during the ladder rung walking task. Representative pictures for category 5 - partial placement and category 6 - correct placement. Please click here to view a larger version of this figure.
Discussion
This behavioral assessment protocol provides an overview of advantages and disadvantages as well as possible readouts of the selected behavioral test battery in a rodent model after sciatic nerve crush injury.
To obtain a comparative outcome of the sciatic nerve crush injury, a consistent crush technique is mandatory. The use of a non-serrated clamp (Ultra Fine Hemostat) instead of forceps can improve the consistency of the crush. Use the same clamp as well as the same crush position to guarantee equal nerve compression. The exclusive use of the clamp for the crush injury and handling of the clamp with care improves consistency. Also, perform the procedure of the crush injury with care. Additional damage to the nerve during surgery such as unwanted traction of the nerve can lead to undesired side effects like automutilation. Therefore, a careful nerve preparation as well as an administration of a pain reliever for a minimum of two days is recommended.
Multifactorial assessment of motor behavior can characterize the phenotype after nerve crush injury in rats at various levels. We used the OFT, CatWalk XT gait analysis, beam walking task and ladder rung walking task. A blinded experimental procedure and data analysis to experimental groups is essential for these experiments. Before behavior assessment, animals were acclimated in the testing room under testing conditions for at least 30 minutes. All the behavioral tests applied herein have the advantage that food or water deprivation are not required. The same group set of animals were used in all described behavioral tests. A maximum of two different behavioral tests per day were performed for each animal. If behavioral tests are performed in regular intervals, pay attention of a comparable procedure, like performing the test in the same animal order and at the same time of the day. A further important aspect for behavioral analysis is the day-night cycle of rats. Consider a reversed day-night cycle to obtain more natural and higher levels of activity at the day cycle (dark cycle). This has to be considered especially for measurement of spontaneous behavior, like the OFT. In this experiment, a reversed day-night cycle could not be implemented, but an adequate acclimatization to the testing conditions was ensured. A perfect illumination is essential for high-resolution videos for the beam walking task and the ladder rung walking task. This high video quality cannot be reached when performing experiments in the dark.
The assessment of gait requires a continuous task performance. The first important aspect of a continuous task performance is to convince the animals to cross the setup. To increase the motivation, place small food pellets (45 mg) at the end of the setup. In order for animals to get familiarized with the food pellets, the pellets should be fed to them prior to testing. Also, a goal box at the end of the setup can be helpful. The setup of the CatWalk already includes a goal box, but rats sometimes hesitate to enter the goal box. Alternatively, you can add a small cage into the goal box, but the home cage from rats does not fit into the goal box. Let the rat habituate in the cage for a few minutes before acquisition. Additionally, another rat from the same home cage may be placed into the goal box or into the cage inside the goal box. Make sure that the second rat remains in the box and does not block the entrance to the goal box. Furthermore, it is also possible to remove the goal box from the CatWalk system and to place the rat home cage at the end of the walkway, which allows the rat to enter their "home territory" after each run. For the setup of the beam walking task and the ladder rung walking task, we recommend to add a goal box or the home cage at the end of the setup. To ensure consistency, the CatWalk, the beam walking task, and the ladder rung walking task should be performed at least once a week with six to ten runs.
Although not every analysis yielded significant differences in this study, consider that an inclusion of genetically modified animals or treatment groups could produce valuable data that can distinguish between groups from the same behavioral tests.
The nerve crush injury had no effect on the locomotor activity of the rat, which was measured in a five minutes OFT. Catwalk XT gait analysis is a more objective and sensitive tool to analyze gait, paw and toe placement. After an intensive training, the rats learn to cross the walkway of the CatWalk XT apparatus to the default settings. The nerve injury does not reduce the ability of the rats to cross the walkway. The automatic computation of various parameters presents the data objectively. Additional information can be gained by using the "Interactive Footprint Measurements" module and indeed, these analyzes yielded significant differences in various parameters of toe spread, print length and paw angle to body axis comparing rats with and without nerve injury.
Rats can be trained easily for the beam walking task. Differences in the latency time to cross the beam and in the number of foot slips per step of the nerve-injured hindlimb were detected by comparing naïve with crush-injured rats. A disadvantage of analyzing nerve-injured rats with the beam walking task is the size of the beam. Within the first two weeks after the sciatic nerve crush injury, the rats need assistance to cross the beam as their balance is impaired. Although some rats may be capable of crossing the beam, the risk of injuries caused by a fall is high. Nerve-crushed animals should therefore be assisted to cross the beam for the first two weeks after sciatic nerve crush injury or longer, if necessary. However, it is difficult to compare runs with and without assistance. Also, the motor balance is an important parameter assessed by the beam walking task. We considered this parameter not to be relevant to our nerve crush rat model. Therefore, scores described by Ohwatashi et al. and Johansson & Ohlsson could not be used and runs with an incomplete beam traverse were excluded for data analysis18,19.
The 7-category scale from Metz et al. can analyze both fore- and hindlimbs and distinguish between different severity levels of errors of all limbs during the ladder rung walking task16,17. By analyzing the most prominent errors, which include the categories from 0 to 2, no differences of errors per step could be detected in the hindlimb when comparing nerve-injured wt rats with naïve wt rats. Furthermore, the latency time of traversing the ladder rung apparatus did not differ between nerve-injured wt rats and wt naïve rats. Deep learning models could improve and speed up data analysis of the ladder rung walking task through an automated approach.
It is important to mention that the nerve crush injury as well as all described behavioral tests can easily translated to mice, by adapting the settings and sizes of the setups. The use of mice as a model organism has the beneficial effect that transgenic models for many human diseases exist.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by the German Federal Ministry of Education and Research (BMBF DysTract to C.W.I.) and by the Interdisciplinary Center for Clinical Research (IZKF) at the University of Würzburg (N-362 to C.W.I.; Z2-CSP3 to L.R.). In addition, this project has received funding from the European Union's Horizon 2020 research and innovation programme under the EJP RD COFUND-EJP N° 825575 (EurDyscover to J.V.), and from the VERUM Foundation. Moreover C.W.I. is funded by the Deutsche Forschungsgemeinschaft (DFG, German Reseach Foundation) Project-ID 424778381-TRR 295, by the Deutsche Stiftung Neurologie and ParkinsonFonds. L.R. is additionally supported by the Dystonia Medical Reseach Foundation.
The authors thank Keali Röhm, Veronika Senger, Heike Menzel and Louisa Frieß for their technical assistance as well as Helga Brünner for the animal care.
Materials
| Name | Company | Catalog Number | Comments |
| Acetic acid, ≥99.8% | Sigma-Aldrich | 33209-1L | |
| Appose ULC skin stapler 35W | Covidien | 8886803712 | |
| Beam | self made | ||
| Bepanthen eye cream | Bayer Vital GmbH | 81552983 | |
| Box for OFT | self made | ||
| Camcorder GC-PX100 | JVC | ||
| Catwalk XT | Noldus | setup and software | |
| Chamber for isofluran | GT-Labortechnik | custom made | |
| Disposable scalpel No. 11 | Feather | 20.001.30.011 | |
| Dräger Vapor 19.3 isoflurane system | Dr. Wilfried Müller GmbH | ||
| Dumont #2 - laminectomy forceps | Fine Science Tools | 11223-20 | |
| Dumont #5 forceps | Fine Science Tools | 11251-30 | super-fine |
| Dustless precision pellets 45 mg | Bio-Serv | F0021 | |
| EthoVision XT | Noldus | setup and software | |
| Forceps 160 mm | Hartenstein | PZ09 | |
| Gas anesthesia mask, rat | Dr. Wilfried Müller GmbH | ||
| Goal box for ladder rung walking task apparatus | self made | ||
| Hair clipper Magnum 5000 | Wahl GmbH | ||
| Hardened fine scissors | Fine Science Tools | 14090-11 | |
| Heating table | MEDAX | 13801 | |
| Isofluran CP 1ml/ml, 250 ml | cp-pharma | 1214 | prescription needed |
| Kinovea | www.kinovea.org | ||
| Ladder rung walking task apparatus | self made | ||
| Needleholder | KLS Martin | 20-526-14-07 | |
| Octeniderm | Schülke | 118211 | |
| Rimadyl 50 mg/ml, injectable | Zoetis | Carprofen, prescription needed | |
| Rubber band retractors | self made | ||
| Spacer for beam | self made | ||
| Spacer for ladder rung walking task apparatus | self made | ||
| Suture Silkam 4/0 DS 19 | B. Braun | C0762202 | |
| Ultra fine hemostats (non-serrated clamp) | Fine Science Tools | 13020-12 |
References
- Iannaccone, P. M., Jacob, H. J. Rats. Disease Models & Mechanisms. 2 (5-6), 206-210 (2009).
- Phifer-Rixey, M., Nachman, M. W. Insights into mammalian biology from the wild house mouse Mus musculus. Elife. 4, (2015).
- Musacchio, T., et al. Subthalamic nucleus deep brain stimulation is neuroprotective in the A53T alpha-synuclein Parkinson's disease rat model. Annals of Neurology. 81 (6), 825-836 (2017).
- Ip, C. W., et al. Tor1a+/- mice develop dystonia-like movements via a striatal dopaminergic dysregulation triggered by peripheral nerve injury. Acta Neuropathologica Communications. 4 (1), 108 (2016).
- Rauschenberger, L., et al. Striatal dopaminergic dysregulation and dystonia-like movements induced by sensorimotor stress in a pharmacological mouse model of rapid-onset dystonia-parkinsonism. Experimental Neurology. 323, 113109 (2020).
- Klein, A., Wessolleck, J., Papazoglou, A., Metz, G. A., Nikkhah, G. Walking pattern analysis after unilateral 6-OHDA lesion and transplantation of foetal dopaminergic progenitor cells in rats. Behavioural Brain Research. 199 (2), 317-325 (2009).
- Kim, D. H., Murovic, J. A., Tiel, R., Kline, D. G. Management and outcomes in 353 surgically treated sciatic nerve lesions. Journal of Neurosurgery. 101 (1), 8-17 (2004).
- Kline, D. G., Kim, D., Midha, R., Harsh, C., Tiel, R. Management and results of sciatic nerve injuries: a 24-year experience. Journal of Neurosurgery. 89 (1), 13-23 (1998).
- Kaplan, H. M., Mishra, P., Kohn, J. The overwhelming use of rat models in nerve regeneration research may compromise designs of nerve guidance conduits for humans. Journal of Materials Science: Materials in Medicine. 26 (8), 226 (2015).
- Bauder, A. R., Ferguson, T. A. Reproducible mouse sciatic nerve crush and subsequent assessment of regeneration by whole mount muscle analysis. Journal of Visualized Experiments. (60), e3606 (2012).
- Savastano, L. E., et al. Sciatic nerve injury: a simple and subtle model for investigating many aspects of nervous system damage and recovery. Journal of Neuroscience Methods. 227, 166-180 (2014).
- Menorca, R. M., Fussell, T. S., Elfar, J. C. Nerve physiology: mechanisms of injury and recovery. Hand Clinics. 29 (3), 317-330 (2013).
- Luis, A. L., et al. Neural cell transplantation effects on sciatic nerve regeneration after a standardized crush injury in the rat. Microsurgery. 28 (6), 458-470 (2008).
- Knorr, S., et al. The evolution of dystonia-like movements in TOR1A rats after transient nerve injury is accompanied by dopaminergic dysregulation and abnormal oscillatory activity of a central motor network. Neurobiology of Disease. , 105337 (2021).
- Quartarone, A., Hallett, M. Emerging concepts in the physiological basis of dystonia. Movement Disorders. 28 (7), 958-967 (2013).
- Metz, G. A., Whishaw, I. Q. The ladder rung walking task: a scoring system and its practical application. Journal of Visualized Experiments. (28), e1204 (2009).
- Metz, G. A., Whishaw, I. Q. Cortical and subcortical lesions impair skilled walking in the ladder rung walking test: a new task to evaluate fore- and hindlimb stepping, placing, and co-ordination. Journal of Neuroscience Methods. 115 (2), 169-179 (2002).
- Johansson, B. B., Ohlsson, A. L. Environment, social interaction, and physical activity as determinants of functional outcome after cerebral infarction in the rat. Experimental Neurology. 139 (2), 322-327 (1996).
- Ohwatashi, A., Ikeda, S., Harada, K., Kamikawa, Y., Yoshida, A. Exercise enhanced functional recovery and expression of GDNF after photochemically induced cerebral infarction in the rat. EXCLI Journal. 12, 693-700 (2013).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved