A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Application of AlDeSense to Stratify Ovarian Cancer Cells Based on Aldehyde Dehydrogenase 1A1 Activity
* These authors contributed equally
In This Article
Summary
Methods to measure ALDH1A1 activity in live cells are critical in cancer research due to its status as a biomarker of stemness. In this study, we employed an isoform-selective fluorogenic probe to determine the relative levels of ALDH1A1 activity in a panel of five ovarian cancer cell lines.
Abstract
Relapse after cancer treatment is often attributed to the persistence of a subpopulation of tumor cells known as cancer stem cells (CSCs), which are characterized by their remarkable tumor-initiating and self-renewal capacity. Depending on the origin of the tumor (e.g., ovaries), the CSC surface biomarker profile can vary dramatically, making the identification of such cells via immunohistochemical staining a challenging endeavor. On the contrary, aldehyde dehydrogenase 1A1 (ALDH1A1) has emerged as an excellent marker to identify CSCs, owing to its conserved expression profile in nearly all progenitor cells including CSCs. The ALDH1A1 isoform belongs to a superfamily of 19 enzymes that are responsible for the oxidation of various endogenous and xenobiotic aldehydes to the corresponding carboxylic acid products. Chan et al. recently developed AlDeSense, an isoform-selective "turn-on" probe for the detection of ALDH1A1 activity, as well as a non-reactive matching control reagent (Ctrl-AlDeSense) to account for off-target staining. This isoform-selective tool has already been demonstrated to be a versatile chemical tool through the detection of ALDH1A1 activity in K562 myelogenous leukemia cells, mammospheres, and melanoma-derived CSC xenografts. In this article, the utility of the probe was showcased through additional fluorimetry, confocal microscopy, and flow cytometry experiments where the relative ALDH1A1 activity was determined in a panel of five ovarian cancer cell lines.
Introduction
Cancer stem cells (CSCs) are a subpopulation of tumor cells that exhibit stem cell-like properties1. Similar to their non-cancerous counterparts, CSCs possess the extraordinary ability to self-renew and proliferate. Together with other built-in mechanisms, such as the upregulation of ATP-binding cassette transporters, CSCs are often spared from initial surgical debulking efforts, as well as subsequent adjuvant therapy2. Owing to their critical role in treatment resistance3, relapse4, and metastasis5, CSCs have become a priority in cancer research. Al....
Access restricted. Please log in or start a trial to view this content.
Protocol
1. Measure total ALDH1A1 activity in ovarian cancer cell homogenates via fluorimetry
- Thaw 1 × 106 cells in a T25 cell culture flask in 5 mL of the following cell culture media:
- IGROV-1 and PEO4: Roswell Park Memorial Institute (RPMI) 1640 medium with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin (P/S).
- BG-1 and Caov-3: Dulbecco's Modified Eagle's Medium (DMEM) with 10% FBS and 1% P/S.
- OVCAR-3: RPMI 1640 with 20% FBS, 1% P/S, and 0.01 mg/mL insulin.
- Maintain the cells in an incubator at 37 °C and 5% CO2 for two to three p....
Access restricted. Please log in or start a trial to view this content.
Results
Total ALDH1A1 activity of ovarian cancer cell homogenates
The average fold turn-ons for each cell line obtained from this assay are: BG-1 (1.12 ± 0.01); IGROV-1 (1.30 ± 0.03); Caov-3 (1.72 ± 0.06); PEO4 (2.51 ± 0.29); and OVCAR-3 (10.25 ± 1.46) (Figure 2).
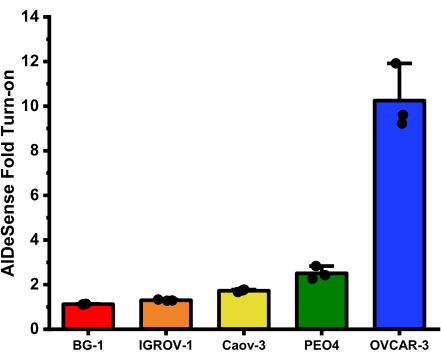
Figure 2<.......
Access restricted. Please log in or start a trial to view this content.
Discussion
Pan-selectivity is a major limitation of many ALDH probes; however, several isoform-selective examples have recently been reported32,33,34,35,36,37,38,39,40,41. The isoform-selective fluo.......
Access restricted. Please log in or start a trial to view this content.
Disclosures
We disclose a pending patent (US20200199092A1) for the AlDeSense technology.
Acknowledgements
This work was supported by The National Institutes of Health (R35GM133581 to JC) and the Cancer Center at Illinois Graduate Scholarship (awarded to SG). JC thanks the Camille and Henry Dreyfus Foundation for support. The authors thank Dr. Thomas E. Bearrood for his initial contribution to preparing stocks of AlDeSense and AlDeSense AM. We thank Mr. Oliver D. Pichardo Peguero and Mr. Joseph A. Forzano for their assistance with preparing various synthetic precursors. We thank Prof. Erik Nelson (Department of Molecular and Integrative Physiology, UIUC) for Caov-3, IGROV-1, and PEO4 cells. We thank Prof. Paul Hergenrother (Department of Chemistry, UIUC) for BG-1 cells. We....
Access restricted. Please log in or start a trial to view this content.
Materials
| Name | Company | Catalog Number | Comments |
| 0.25% Trypsin, 0.1% EDTA in HBSS w/o Calcium, Magnesium and Sodium Bicarbonate | Corning | 25-050-CI | |
| 1x Phosphate Buffer Saline | Corning | 21-040-CMX12 | |
| AccuSpin Micro 17R | Fisher Scientific | 13-100-675 | |
| AlDeSense | Synthesized in-house | ||
| BG-1 | A gift provided by the Prof. Paul Hergenrother Lab, University of Illinois Urbana-Champaign | ||
| BioLite 25cm2 Flask | Thermo Fisher Scientific | 130189 | |
| Biosafety Cabinet 1300 series A2 | Thermo Fisher Scientific | ||
| Caov-3 | A gift provided by the Prof. Erik Nelson Lab, University of Illinois Urbana-Champaign | ||
| Cell homogenizer | Fisher Scientific | ||
| Centrifuge 5180R | Eppendorf | 22627040 | |
| Contrl-AlDeSense | Synthesized in-house | ||
| DMEM, 10% FBS, 1% P/S | Prepared by UIUC cell media facility | ||
| Falcon Round-Bottom Polystyrene Test Tubes with Cell Strainer Snap Cap, 5mL | Corning | 352003 | |
| FCS Express 6 | Provided by UIUC CMtO | ||
| FL microscope | EVOS | ||
| Fluorometer | Photon Technology International | ||
| Forma Series II Water-Jacketed CO2 Incubator | Fisher Scientific | 3110 | |
| IGROV-1 | A gift provided by the Prof. Erik Nelson Lab, University of Illinois Urbana-Champaign | ||
| ImageJ | NIH | ||
| Innova 42R Incubated Shaker | |||
| LSM 700 | Zeiss | ||
| LSR II | BD | ||
| Nunc Lab-Tek Chambered #1.0 Borosicilate Coverglass System | Thermo Fisher Scientific | 155383 | |
| OVCAR-3 | ATCC | HTB-161 | |
| PEO4 | A gift provided by the Prof. Erik Nelson Lab, University of Illinois Urbana-Champaign | ||
| Pierce Protease Inhibitor Tablets | Thermo Scientific | A32963 | |
| Poly-L-Lysine | Cultrex | 3438-100-01 | |
| Rocker | VWR | ||
| RPMI, 10% FBS, 1% P/S | Prepared by UIUC cell media facility | ||
| RPMI, 20% FBS, 1% P/S, 0.01 mg/mL Insulin | Prepared by UIUC cell media facility |
References
- Bonnet, D., Dick, J. E. Human acute myeloid leukaemia is organised as a heirarchy that originates from a primitive haematopoetic cell. Nature Medicine. 3 (7), 730-737 (1997).
- Begicevic, R. R., Falasca, M. ABC tr....
Access restricted. Please log in or start a trial to view this content.
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved