A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Magnetometric Characterization of Intermediates in the Solid-State Electrochemistry of Redox-Active Metal-Organic Frameworks
In This Article
Summary
Ex situ magnetic surveys can directly provide bulk and local information on a magnetic electrode to reveal its charge storage mechanism step by step. Herein, electron spin resonance (ESR) and magnetic susceptibility are demonstrated to monitor the evaluation of paramagnetic species and their concentration in a redox-active metal-organic framework (MOF).
Abstract
Electrochemical energy storage has been a widely discussed application of redox-active metal-organic frameworks (MOFs) in the past 5 years. Although MOFs show outstanding performance in terms of gravimetric or areal capacitance and cyclic stability, unfortunately their electrochemical mechanisms are not well understood in most cases. Traditional spectroscopic techniques, such as X-ray photoelectron spectroscopy (XPS) and X-ray absorption fine structure (XAFS), have only provided vague and qualitative information about valence changes of certain elements, and the mechanisms proposed based on such information are often highly disputable. In this article, we report a series of standardized methods, including the fabrication of solid-state electrochemical cells, electrochemistry measurements, the disassembly of cells, the collection of MOF electrochemical intermediates, and physical measurements of the intermediates under the protection of inert gases. By using these methods for quantitatively clarifying the electronic and spin state evolution within a single electrochemical step of redox-active MOFs, one can provide clear insight into the nature of electrochemical energy storage mechanisms not only for MOFs, but also for all other materials with strongly correlated electronic structures.
Introduction
Since the term metal-organic framework (MOF) was introduced in the late 1990s, and especially in the 2010s, the most representative scientific concepts concerning MOFs have arisen from their structural porosity, including guest encapsulation, separation, catalytic properties, and molecule sensing1,2,3,4. Meanwhile, scientists were quick to realize that it is essential for MOFs to possess stimuli-responsive electronic properties in order to integrate them into modern smart devices. This idea triggered the spawning and flourishing of the conductive two-dimensional (2D) MOF family in the past 10 years, thereby opening the gate for MOFs to play key roles in electronics5 and, more attractively, in electrochemical energy storage devices6. These 2D MOFs have been incorporated as active materials in alkali metal batteries, aqueous batteries, pseudocapacitors, and supercapacitors7,8,9, and have exhibited tremendous capacity as well as excellent stability. However, to design better-performing 2D MOFs, it is crucial to understand their charge storage mechanisms in detail. Therefore, this article aims to provide a comprehensive understanding of the electrochemical mechanisms of MOFs, which can aid in the rational design of better-performing MOFs for energy storage applications.
In 2014, we first reported the solid-state electrochemical mechanisms of MOFs with redox-active sites on both metal cations and ligands10,11. These mechanisms were interpreted with the help of various in situ and ex situ spectroscopic techniques, such as X-ray photoelectron spectroscopy (XPS), X-ray absorption fine structure (XAFS), X-ray diffraction (XRD), and solid-state nuclear magnetic resonance (NMR). Since then, this research paradigm has become a trend in studies of the solid-state electrochemistry of molecular-based materials12. These methods work fine for identifying the redox events of conventional MOFs with carboxylate bridging ligands, as the molecular orbitals and energy levels of metal cluster building blocks and organic ligands are almost independent of each other in such MOFs12,13.
However, when encountering the strongly correlated 2D MOFs with significant π-d conjugation, the limitations of these spectroscopic methods were exposed. One of these limitations is that the band levels of most aforementioned 2D MOFs cannot be considered as a simple combination of metal clusters and ligands, but are rather a hybridization of them, while most of the spectroscopic methods only provide averaged, qualitative information about the oxidation states14. The other limitation is that the interpretation of these data is always based on the assumption of localized atomic orbitals. Therefore, the intermediate states with metal-ligand hybridization and delocalized electronic states are usually overlooked and described incorrectly with only these spectroscopic methods15. It is necessary to develop new probes for the electronic states of these electrochemical intermediates of not only 2D MOFs, but also other materials with similar conjugated or strongly correlated electronic structures, such as covalent organic frameworks16, molecular conductors, and conjugated polymers17.
The most common and powerful tools for assessing the electronic structures of materials are electron spin resonance (ESR) and superconducting quantum interference device (SQUID) magnetic susceptibility measurements18,19. As both rely on unpaired electrons in the system, these tools can provide tentative information about the spin densities, spin distributions, and spin-spin interactions. ESR offers sensitive detection of unpaired electrons, while magnetic susceptibility measurement gives more quantitative signals for upper properties20. Unfortunately, both techniques unavoidably face great challenges when used to analyze the electrochemical intermediates. This is because target samples are not pure, but rather a mixture of target material, conductive additive, binder, and byproduct from the electrolyte, so the obtained data21,22 are the sum of contributions from both the material and the impurities. Meanwhile, most intermediates are sensitive to the environment, including air, water, certain electrolytes, or any other unpredictable perturbations; extra care is necessary while handling and measuring intermediates. Trial and error is normally necessary while dealing with a new combination of electrode material and electrolyte.
Here, we present a new paradigm, called electrochemical magnetometry, for analyzing the electronic states or spin states of 2D MOFs and similar materials using a series of techniques, utilizing electrochemistry and temperature-variable ex situ ESR spectroscopy as well as ex situ magnetic susceptibility measurements20. To demonstrate the effectiveness of this approach, we use Cu3THQ2 (THQ = 1,2,4,5-tetrahydroxybenzoquinone; referred to as Cu-THQ), a representative 2D MOF, as an example. We explain the selection of conductive additives and electrolytes, the fabrication of electrodes and electrochemical cells, as well as details on sample handling and measurement, including possible issues during measurement. By comparing with classic characterizations such as XRD and XAFS, electrochemical magnetometry can provide a comprehensive understanding of the electrochemical mechanism of most MOFs. This approach is capable of capturing unique intermediate states and avoiding incorrect assignment of redox events. The elucidation of energy storage mechanisms using electrochemical magnetometry can also contribute to a better understanding of the structure-function relationships in MOFs, leading to more intelligent synthetic strategies for MOFs and other conjugated materials.
Access restricted. Please log in or start a trial to view this content.
Protocol
1. Electrode fabrication
- Synthesizing Cu-THQ MOF
NOTE: Cu-THQ MOF polycrystalline powder was synthesized via a hydrothermal method following previously published procedures14,20,23.- Put 60 mg of tetrahydroxyquinone into a 20 mL ampule, then add 10 mL of degassed water. In a separate glass vial, dissolve 110 mg of copper (II) nitrate trihydrate in another 10 mL of degassed water. Add 46 µL of the competing ligand ethylenediamine using a pipette.
NOTE: To degas the deionized water, flow nitrogen gas for 30 min before use. If the reaction mixture is heated for too long, a Cu impurity may form with a diffraction peak appearing around 43° (Cu Kα). - Introduce the copper solution into the ampule containing the tetrahydroxyquinone. The color of the solution immediately changes from red to navy. Freeze, pump, and thaw24 the resulting solution three times to further remove dissolved oxygen.
- Flame seal the ampule using a torch under vacuum. Heat the solution to 60 °C for 4 h.
- After the reaction, carefully open the ampule and remove the supernatant. Wash the precipitate with 30 mL of room-temperature deionized water 3x and 30 mL of hot deionized water (80 °C) by centrifugation 3x at 10,000 rpm for 5 min.
- Disperse the precipitate into acetone by shaking, then filter and wash with acetone. Heat the product under vacuum at 353 K overnight to remove the residual solvent in the Cu-THQ MOF.
- Put 60 mg of tetrahydroxyquinone into a 20 mL ampule, then add 10 mL of degassed water. In a separate glass vial, dissolve 110 mg of copper (II) nitrate trihydrate in another 10 mL of degassed water. Add 46 µL of the competing ligand ethylenediamine using a pipette.
- Preparing CuTHQ electrodes
NOTE: To distinguish between the Cu-THQ MOF and the electrode, the former is referred to as Cu-THQ, while the mixture of Cu-THQ, carbon, and binder is referred to simply as the CuTHQ.- To prepare the Cu-THQ/CB/PVDF electrode, dissolve 10 mg of poly(vinylidene fluoride) (PVDF) in 1.4 mL of N-methyl-2-pyrrolidone (NMP). Disperse 50 mg of Cu-THQ MOF and 40 mg of carbon black (CB) in the solution by vigorously stirring overnight. Coat the homogeneous slurry onto an Al disk with a diameter of 15 mm and a mass of ~9.7 mg.
- To prepare the Cu-THQ/Gr/SP/SA electrode, follow the same procedure as the Cu-THQ/CB/PVDF electrode, but with a different slurry composition: Cu-THQ MOF (80 mg), sodium alginate (SA, 2 mg), and graphene/Super P (Gr/SP, 1:1.8 dilution by weight, total of 18 mg) in water/isopropanol (1:1 dilution by volume, total of 1.2 mL).
- Dry the electrodes under vacuum at 353 K for 12 h. Vent the nitrogen gas after drying and measure the mass loading.
2. Battery assembly and post-treatment
NOTE: Due to the air-sensitive nature of electrochemical intermediates, battery assembly and post-treatment must be performed in an argon glove box with strict air-free manners.
- Assembling Li/CuTHQ coin cells
- Cut several pieces of lithium disks with a diameter of 15.5 mm and Celgard separators with a diameter of 17 mm before assembling the battery.
- Assemble Li/CuTHQ coin cells (CR2032) from bottom to top in the following order: negative shell, spacer (height = 0.5 mm), lithium, separator, CuTHQ electrode (prepared in step 1.2.1 or 1.2.2), spacer, spring, and positive shell (Figure 1A).
- Before and after adding the separator, drop a total of 0.04 mL of electrolyte (1.0 M LiBF4 in ethylene carbonate (EC)/diethyl carbonate (DEC) at 1:1 wt%). Do not use metal tweezers to hold the coin cell after it is assembled.
- Preparing electrochemical intermediates
- Compress the coin cell using the tightening screw (not sealed) with a homemade device (Figure 1B) and connect the device to the measuring cables in the glove box. Connect the instrument (outside the glove box) to the ports corresponding to the coin cell. Perform cyclic voltammetry and galvanostatic charge/discharge measurements20 to achieve the intermediates at different potentials (Figure 2).
- After electrochemical cycling, disassemble the coin cell carefully to avoid any short circuits.
- Rinse the cycled CuTHQ electrode with 5 mL of battery-grade dimethyl carbonate (DMC). Dry the electrode naturally for 30 min. Collect the sample from the Al disk to Al foil using a clean spatula.
- Transfer the sample powder into an ESR tube or SQUID tube through a homemade glass funnel (Figure 1C). Seal the sample tube tightly with a cap and transparent film. Alternatively, connect the sample tube to a rubber tube and seal it with a valve, followed by flame sealing the head of the sample tube under vacuum.
- After magnetic measurements20, open the sample tube and dump the sample onto Al foil. Measure the mass of the sample using an analytical balance with a resolution of 0.01 mg in air. Estimate the mass of Cu-THQ from the total mass of the sample.
NOTE: The mass of the cycled Cu-THQ MOF is estimated to be 50% or 80% of the total mass, depending on the type of electrodes used; this estimate does not take into account the inserted Li ions and residual electrolyte.
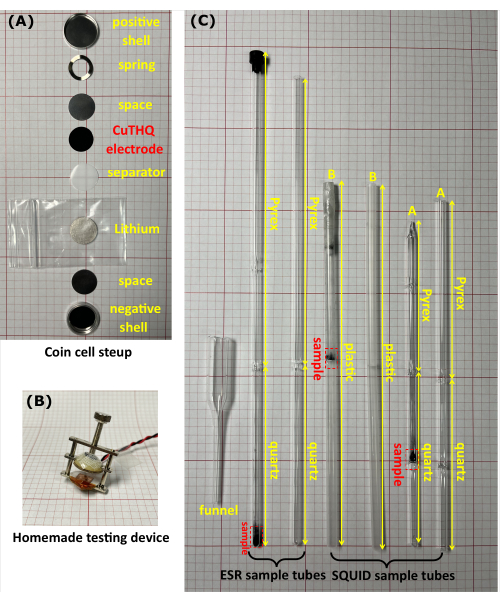
Figure 1: The equipment used for ex situ magnetometry experiments. (A) A Photograph of a CR2032 coin cell. (B) The homemade device was used to evaluate the unsealed coin cell in the glove box. (C) Photographs of ESR and SQUID sample tubes with and without samples inside. The ESR tube consists of a 10 cm high-purity quartz tip (measurement section) and a 17 cm borosilicate glass head. There are two kinds of SQUID tubes. Tube A consists of a 2 cm x 5 cm quartz tip with a quartz diaphragm at the midpoint and a 10 cm borosilicate glass head, and tube B is a plastic tube (20 cm long) with a plastic diaphragm at the midpoint. All sample tubes have an outer diameter of 5 mm. Please click here to view a larger version of this figure.
3. Registration of ESR spectra at variable temperatures
- Recording the ESR spectra at room temperature
- Once the ESR spectrometer is ready, insert the prepared sample tube into the microwave cavity and center the sample. Autotune the microwave phase, coupling, and frequency to reach the cavity's resonance condition. Check the Q-dip in the center of the screen for a symmetric shape and maximal depth.
NOTE: If the sample contains too much conductive carbon, such as carbon black, the autotune process may fail or result in a small quality factor (Q-value) of the cavity. The typical mass of the sample is 3 mg. - Choose the optimal parameters, such as: microwave: power; magnetic field: sweep time; center field: sweep width; modulation: frequency, width; channel: amplitude, time constant. Then, sweep the magnetic field and record the ESR spectrum. The typical values of the measurement parameters are shown in Figure 3 and Figure 4.
- Adjust the insertion amount of the Mn marker to 800. Repeat steps 3.1.1 and 3.1.2 to record an ESR spectrum with the Mn marker. Calibrate the magnetic field by employing six hyperfine lines for the Mn(II) ions.
- Once the ESR spectrometer is ready, insert the prepared sample tube into the microwave cavity and center the sample. Autotune the microwave phase, coupling, and frequency to reach the cavity's resonance condition. Check the Q-dip in the center of the screen for a symmetric shape and maximal depth.
- Line shape analysis of Cu-THQ
- Import the ESR dataset into Python (version 3.9.7). Normalize the ESR spectrum by dividing the intensity by the sample mass, the square root of the microwave power, the modulation width, and the amplitude.
- Fit the calibrated and normalized ESR spectrum of as-prepared Cu-THQ MOF to the axially symmetric Lorentzian function25:
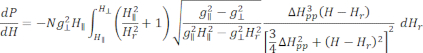
Where N is a scale factor that includes instrument parameters gll and HII constants, (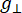 and
and 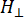 ) are the parallel (perpendicular) component of Lander g-factor and the corresponding resonance magnetic field, ΔHpp is the peak-to-peak line width, and Hr is an integral variable.
) are the parallel (perpendicular) component of Lander g-factor and the corresponding resonance magnetic field, ΔHpp is the peak-to-peak line width, and Hr is an integral variable.
NOTE: The Python codes for the Lorentzian function are available in Supplementary Coding File 1 (which is named as AxialLorentz). - Obtain the anisotropic g-value and peak-to-peak line width for the axially symmetric Cu(II) ions.
- Fit the calibrated and normalized ESR spectrum with the Lorentzian function for the radical samples. Obtain the isotropic g-value and peak-to-peak line width for the radicals.
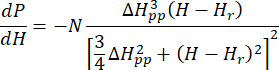
This is named as SymLorentz in Supplementary Coding File 1.
- Quantitating the radical concentration
- Grind 3.45 mg of 4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPOL) and 96.55 mg of KBr together in an agate mortar until a homogeneous mixture is achieved. Place 1 mg (0.2 µmol), 2 mg (0.4 µmol), and 4 mg (0.8 µmol) of TEMPOL/KBr mixtures into three ESR sample tubes, respectively.
- Follow steps 3.1.1 and 3.1.2 to record the ESR spectra for the TEMPOL/KBr standards.
- Conduct a linear baseline fitting between the double integration of the ESR spectra and the number of spins in the TEMPOL/KBr standards. Determine the number of spins in the cycled Cu-THQ using the linear baseline of the TEMPOL/KBr standards26.
- Recording the ESR spectra at low temperatures
NOTE: Use liquid helium to achieve a cryogenic temperature. It is necessary to wear cryogenic gloves when working with liquid helium.- Check the ESR spectrum at room temperature first by following step 3.1.
- Evacuate the thermal shield to a high vacuum level. Purge the microwave cavity using nitrogen gas to avoid condensation.
- Introduce liquid helium from the vessel into the cryostat. Gradually cool the sample to the lowest temperature (around 10 K). Wait 30 min to achieve thermal equilibrium.
- Record the temperature-dependent ESR spectra during warming. Confirm that the ESR spectrum does not suffer from the power saturation effect at low temperature, and the ratio between signal intensity (peak-to-peak height) and the square root of microwave power remains constant in the absence of power saturation.
NOTE: When power is saturated, the signal intensity increases more slowly than the square root of the microwave power. The sampling density could gradually decrease as the temperature rises.
4. Magnetic susceptibility measurements
- Attach the sample tube to the bottom of the sample rod. Make sure that the surface of the sample tube is clean.
- Purge the sample chamber and insert the sample tube into the SQUID. Apply a magnetic field and center the sample within the detection coil. Remove the external magnetic field after centering.
NOTE: If the spin concentration is too low to detect, consider increasing the magnetic field or centering after cooling to 2 K. The typical sample mass for SQUID measurements is approximately 6 mg. - Cool the system to 20 K at a rate of 10 K/min. Pause cooling for 30 min, then further cool to 2 K for 1 h.
- Measure the magnetic susceptibility of the cycled CuTHQ electrode under a magnetic field of 1,000 Oe while warming to 300 K; this is referred to as the zero-field-cooled (ZFC) process. Next, cool to 2 K again and record the magnetic susceptibility in the field-cooled (FC) process.
- Repeat steps 4.1 to 4.4 with CuTHQ electrodes cycled at different reduction degrees.
- Measure the magnetic susceptibility of the carbon materials (Gr/SP) under the same conditions. Use this result to compensate for the magnetic susceptibility of the CuTHQ electrodes.
- Fit the temperature dependence of magnetic susceptibility to the modified Curie-Weiss law:
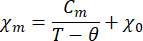
Where χm is the molar magnetic susceptibility, Cm is the molar Curie constant, θ is the Weiss temperature, and χ0 is a temperature-independent term.
Access restricted. Please log in or start a trial to view this content.
Results
Our previous work included a detailed discussion of ex situ ESR spectroscopy and ex situ magnetic susceptibility measurements for electrochemically cycled CuTHQ20. Here, we present the most representative and detailed results that can be obtained following the protocol described in this paper.
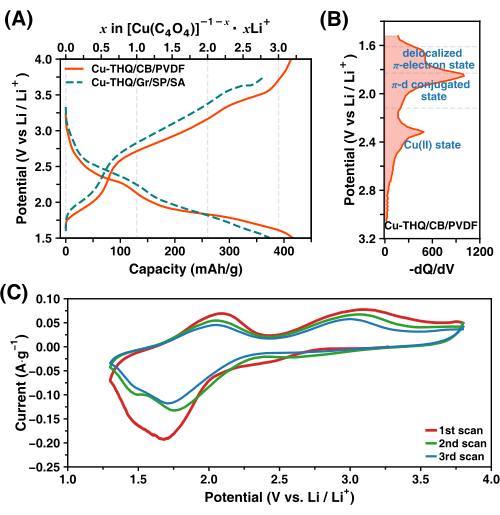
Fi...
Access restricted. Please log in or start a trial to view this content.
Discussion
To produce cathodes, it is necessary to mix the active material with conductive carbon to achieve a low polarization during the electrochemical process. The carbon additive is the first critical point for ex situ magnetometry; if the carbon has radical defects, the emergence of the electrochemically induced organic radical cannot be observed in the ESR spectrum. This makes it difficult to precisely determine the spin concentration or organic radical concentration, since these two types of radicals have similar g...
Access restricted. Please log in or start a trial to view this content.
Disclosures
The authors have no conflicts of interest to declare.
Acknowledgements
This study was supported by a Japan Society for the Promotion of Science (JSPS) KAKENHI Grant (JP20H05621). Z. Zhang also thanks the Tatematsu Foundation and Toyota Riken scholarship for financial support.
Access restricted. Please log in or start a trial to view this content.
Materials
| Name | Company | Catalog Number | Comments |
| 1-Methyl-2-pyrrolidone | FUJIFILM Wako Chemicals | 139-17611 | Super Dehydrated |
| 1mol/L LiBF4 EC:DEC (1:1 v/v%) | Kishida | LBG-96533 | electrolyte |
| 4-Hydroxy-2,2,6,6-tetramethylpiperidine-1-oxyl | FUJIFILM Wako Chemicals | 089-04191 | TEMPOL, for Spin Labeling |
| Ampule tube | Maruemu Corporation | 5-124-05 | 20mL |
| Carbon black, Super P Conductive | Alfa Aesar | H30253 | |
| Conductive Carbon Black | Mitsubishi Chemical | ||
| Copper (II) Nitrate Trihydrate | FUJIFILM Wako Chemicals | 033-12502 | deleterious substances |
| Dimethyl Carbonate | FUJIFILM Wako Chemicals | 046-31935 | battery grade |
| Ethylenediamine | FUJIFILM Wako Chemicals | 053-00936 | deleterious substances |
| Graphene Nanoplatelets | Tokyo Chemical Industry | G0442 | 6-8nm(thick), 15µm(wide) |
| Poly(vinylidene fluoride) | Sigma Aldrich | 182702 | |
| Potassium Bromide | FUJIFILM Wako Chemicals | 165-17111 | for Infrared Spectrophotometry |
| Sodium Alginate | FUJIFILM Wako Chemicals | 199-09961 | 500-600 cP |
| SQUID Magnetometer | Quantum Design | MPMS-XL 5 | |
| Tetrahydroxy-1,4-benzoquinone Hydrate | Tokyo Chemical Industry | T1090 | |
| X-Band ESR | JEOL | JES-F A200 |
References
- Lee, J., et al. Metal-organic framework materials as catalysts. Chemical Society Reviews. 38 (5), 1450-1459 (2009).
- Dolgopolova, E. A., Rice, A. M., Martin, C. R., Shustova, N. B. Photochemistry and photophysics of MOFs: steps towards MOF-based sensing enhancements. Chemical Society Reviews. 47 (13), 4710-4728 (2018).
- Qian, Q., et al. MOF-based membranes for gas separations. Chemical Reviews. 120 (16), 8161-8266 (2020).
- Wang, Q., Astruc, D. State of the art and prospects in metal-organic framework (MOF)-based and MOF-derived nanocatalysis. Chemical Reviews. 120 (2), 1438-1511 (2020).
- Wang, M., Dong, R., Feng, X. Two-dimensional conjugated metal-organic frameworks (2D c-MOFs): chemistry and function for MOFtronics. Chemical Society Reviews. 50 (4), 2764-2793 (2021).
- Baumann, A. E., Burns, D. A., Liu, B., Thoi, V. S. Metal-organic framework functionalization and design strategies for advanced electrochemical energy storage devices. Communications Chemistry. 2 (1), 86(2019).
- Nam, K. W., et al. Conductive 2D metal-organic framework for high-performance cathodes in aqueous rechargeable zinc batteries. Nature Communications. 10 (1), 4948(2019).
- Sheberla, D., et al. Conductive MOF electrodes for stable supercapacitors with high areal capacitance. Nature Materials. 16 (2), 220-224 (2017).
- Wang, Z., et al. Ultrathin two-dimensional conjugated metal-organic framework single-crystalline nanosheets enabled by surfactant-assisted synthesis. Chemical Science. 11 (29), 7665-7671 (2020).
- Zhang, Z., Yoshikawa, H., Awaga, K. Monitoring the solid-state electrochemistry of Cu(2,7-AQDC) (AQDC = anthraquinone dicarboxylate) in a lithium battery: Coexistence of metal and ligand redox activities in a metal-organic framework. Journal of the American Chemical Society. 136 (46), 16112-16115 (2014).
- Zhang, Z., Yoshikawa, H., Awaga, K. Discovery of a "bipolar charging" mechanism in the solid-state electrochemical process of a flexible metal-organic framework. Chemistry of Materials. 28 (5), 1298-1303 (2016).
- Li, C., Hu, X., Hu, B. Cobalt(II) dicarboxylate-based metal-organic framework for long-cycling and high-rate potassium-ion battery anode. Electrochimica Acta. 253, 439-444 (2017).
- Liu, J., et al. Reversible formation of coordination bonds in Sn-based metal-organic frameworks for high-performance lithium storage. Nature Communications. 12 (1), 3131(2021).
- Jiang, Q., et al. A redox-active 2D metal-organic framework for efficient lithium storage with extraordinary high capacity. Angewandte Chemie. 59 (13), 5273-5277 (2020).
- Sakaushi, K., Nishihara, H. Two-dimensional π-conjugated frameworks as a model system to unveil a multielectron-transfer-based energy storage mechanism. Accounts of Chemical Research. 54 (15), 3003-3015 (2021).
- Li, H., et al. 2D organic radical conjugated skeletons with paramagnetic behaviors. Advanced Materials Interfaces. 8 (18), 2100943(2021).
- Peeks, M. D., et al. Electronic delocalization in the radical cations of porphyrin oligomer molecular wires. Journal of the American Chemical Society. 139 (30), 10461-10471 (2017).
- Krug von Nidda, H. A., et al. Anisotropic exchange in LiCuVO4 probed by ESR. Physical Review B. 65 (13), 134445(2002).
- Zeng, Z., et al. Pro-aromatic and anti-aromatic π-conjugated molecules: An irresistible wish to be diradicals. Chemical Society Reviews. 44 (18), 6578-6596 (2015).
- Chen, Q., Adeniran, O., Liu, Z. F., Zhang, Z., Awaga, K. Graphite-like charge storage mechanism in a 2D π-d conjugated metal-organic framework revealed by stepwise magnetic monitoring. Journal of the American Chemical Society. 145 (2), 1062-1071 (2023).
- Julien, C. M., Mauger, A., Groult, H., Zhang, X., Gendron, F. LiCo1-yByO2 as cathode materials for rechargeable lithium batteries. Chemistry of Materials. 23 (2), 208-218 (2011).
- Niemöller, A., Jakes, P., Eichel, R. A., Granwehr, J. In operando EPR investigation of redox mechanisms in LiCoO2. Chemical Physics Letters. 716, 231-236 (2019).
- Park, J., et al. Synthetic routes for a 2D semiconductive copper hexahydroxybenzene metal-organic framework. Journal of the American Chemical Society. 140 (44), 14533-14537 (2018).
- Rondeau, R. E. A technique for degassing liquid samples. Journal of Chemical Education. 44 (9), 530(1967).
- Flores-Llamas, H. Inhomogeneously broadened EPR lineshape of axial powder. Applied Magnetic Resonance. 9 (2), 289-298 (1995).
- Sun, L., et al. Room-temperature quantitative quantum sensing of lithium ions with a radical-embedded metal-organic framework. Journal of the American Chemical Society. 144 (41), 19008-19016 (2022).
- Chen, Y., et al. Successive storage of cations and anions by ligands of π-d-conjugated coordination polymers enabling robust sodium-ion batteries. Angewandte Chemie. 60 (34), 18769-18776 (2021).
- Roessler, M. M., Salvadori, E. Principles and applications of EPR spectroscopy in the chemical sciences. Chemical Society Reviews. 47 (8), 2534-2553 (2018).
- Ji, X., et al. Pauli paramagnetism of stable analogues of pernigraniline salt featuring ladder-type constitution. Journal of the American Chemical Society. 142 (1), 641-648 (2020).
- Noel, M., Santhanam, R. Electrochemistry of graphite intercalation compounds. Journal of Power Sources. 72 (1), 53-65 (1998).
- Wu, K. H., Ting, T. H., Wang, G. P., Ho, W. D., Shih, C. C. Effect of carbon black content on electrical and microwave absorbing properties of polyaniline/carbon black nanocomposites. Polymer Degradation and Stability. 93 (2), 483-488 (2008).
- Yao, M., Taguchi, N., Ando, H., Takeichi, N., Kiyobayashi, T. Improved gravimetric energy density and cycle life in organic lithium-ion batteries with naphthazarin-based electrode materials. Communications Materials. 1 (1), 70(2020).
- Krzystek, J., et al. EPR spectra from "EPR-silent" species: High-frequency and high-field EPR spectroscopy of pseudotetrahedral complexes of nickel(II). Inorganic Chemistry. 41 (17), 4478-4487 (2002).
Access restricted. Please log in or start a trial to view this content.
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved