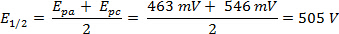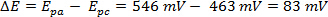循环伏安法 (CV)
Overview
资料来源: 实验室的博士凯拉绿色 — — 德克萨斯基督教大学
循环伏安法 (CV) 实验涉及一系列潜在电压测量时当前的扫描。在 CV 实验中,电极电势的沉管、 固定从预定的起始电位扫描到最终值 (称为开关潜在),得出反向扫描。这给 '循环' 扫的电位和电流与电位曲线所得的数据称为循环伏安图。第一次被称为 '向前扫描',回波被称为反向扫描。潜在的极端被称为 '扫描窗口'。氧化与还原电流的大小和形状的伏安是高度依赖分析物的浓度、 扫描速率和实验条件。通过改变这些因素,循环伏安法可以产生稳定的过渡金属氧化态中的电子转移反应的可逆性,复合体,资料及反应性的资料。这个视频会解释包括分析物制备和设置电化学电池的循环伏安法实验的基本设置。将提出一个简单的循环伏安法实验。
Procedure
1.电解质溶液的制备
- 准备股票电解液 (10 毫升) 组成的 0.1 M [埠4N] [高炉4] 在 CH3CN。
- 电解质溶液置于电化学瓶、 小搅拌棒,加到小瓶上盖,如图 1所示。
- 检查以确保氮铅在电解质溶液中。搅拌和脱气一连串温柔干 N2气 (~ 10 分钟) 要删除氧化还原活性分子氧的电解质溶液。
- 在步骤 1.3 中,小心地插入工作电极 (例如玻碳)、 计数器 (Pt) 和参比电极 (Ag/AgNO3) 铁氟龙单元格顶部。连接单元格站到适当的电极。
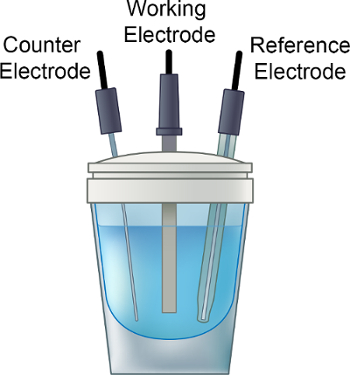
图 1。安装程序的一种电化学电池。
Results
References
- Bard, A. J., Faulkner, L. A. Electrochemical methods: Fundamentals and Applications. 2nd ed. New York: Wiley; 833 p. (2001).
- Geiger, W. E., Connelly, N. G. Chemical Redox Agents for Organometallic Chemistry. Chem Rev. 96 (2), 877-910, (1996).
Tags
跳至...
此集合中的视频:

Now Playing
循环伏安法 (CV)
Analytical Chemistry
124.3K Views

分析制备的样品制备
Analytical Chemistry
84.2K Views

内部标准
Analytical Chemistry
204.3K Views

标准加入的方法
Analytical Chemistry
319.4K Views

校准曲线
Analytical Chemistry
794.7K Views

(紫外-可见) 的紫外-可见光谱法
Analytical Chemistry
621.7K Views

拉曼光谱化学分析
Analytical Chemistry
51.0K Views

X 射线荧光光谱 (XRF)
Analytical Chemistry
25.3K Views

气相色谱 (GC) 与火焰电离检测
Analytical Chemistry
281.1K Views

高性能液相色谱法 (HPLC)
Analytical Chemistry
383.1K Views

离子交换色谱法
Analytical Chemistry
263.9K Views

毛细管电泳 (CE)
Analytical Chemistry
93.3K Views

质谱分析法导论
Analytical Chemistry
111.9K Views

扫描电子显微镜 (SEM)
Analytical Chemistry
86.8K Views

负载型催化剂,用恒电位仪/结合电化学测量
Analytical Chemistry
51.3K Views
版权所属 © 2025 MyJoVE 公司版权所有,本公司不涉及任何医疗业务和医疗服务。