Komplexchemie
Überblick
Quelle: Labor von Dr. Neal Abrams – SUNY College of Environmental Science and Forestry
Übergangsmetalle befinden sich überall von Vitamin-Ergänzungen zur galvanischen Bädern. Übergangsmetalle auch bilden die Pigmente in vielen Farben und alle Mineralien zu komponieren. In der Regel sind Übergangsmetalle in kationischen Form gefunden, da sie leicht oxidieren, oder Elektronen verlieren und Elektron Spender genannt Liganden umgeben sind. Diese Liganden nicht ionische Form oder kovalente Bindungen mit dem Metallzentrum, vielmehr nehmen sie auf eine dritte Art von Bindung bekannt als Koordinate kovalente. Die Koordinate kovalente Bindung zwischen einem Liganden und ein Metall ist dynamisch, d.h. Liganden sind ständig austauschen und neu zu koordinieren, um das Metall Zentrum. Die Identität des Metalls und der Liganden diktiert die Liganden bevorzugt gegenüber einem anderen verpfänden. Darüber hinaus sind Farbe und magnetischen Eigenschaften auch aufgrund der Arten von komplexen, die gebildet werden. Die Koordination-Verbindungen, die bilden werden analysiert, mit einer Vielzahl von Instrumenten und Werkzeugen. Dieses Experiment untersucht, warum so viele komplexe möglich sind und verwendet eine Spektrochemische (Farbe und chemische) Methode, um die Art der Koordination Komplex zu identifizieren, die bildet.
Verfahren
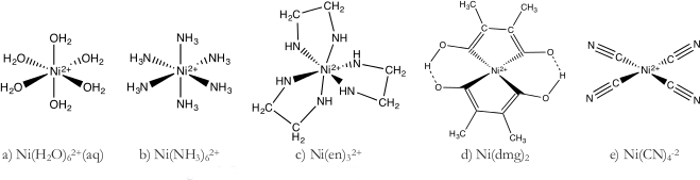
(1) nickel-komplexe und Farben
- NI (H2O)62 + Komplex (Abbildung 1a)
- Bereiten Sie eine 1 M Lösung Ni (H2O)62 + durch NiSO4 in das entsprechende Volumen des Wassers auflösen.
- Weiter verdünnen Sie die Ni (H2O)62 +Lösung durch Zugabe von 70 mL der Lösung 1 M bis 1.000 mL entionisiertem Wasser.
- Teilen Sie die Ni (H2O)62 + unte
Anwendung und Zusammenfassung
Aus Pigmenten, Menschen sind Übergangsregelungen Metalle in Bereichen der Chemie, Biologie, Geologie und Technik gefunden. Verständnis des Verhaltens von Übergangsmetallen unter verschiedenen chemischen Zuständen kann so einfach wie das monitoring Farbe oder magnetische Verhalten sein. Fast jeder 3d (4ten Zeile) Übergangsmetall ist entscheidend für die physiologische Funktion und in allen Fällen sind diese Metalle zu Formular Koordination komplexen Liganden gebunden. Eisen ist beispielsweise wichtig fü...
Tags
pringen zu...
Videos aus dieser Sammlung:

Now Playing
Komplexchemie
General Chemistry
91.8K Ansichten

Labor-Glaswaren und deren Verwendung
General Chemistry
659.1K Ansichten

Lösungen und Konzentrationen
General Chemistry
275.4K Ansichten

Bestimmung der Dichte von Fest- und Flüssigstoffen
General Chemistry
557.0K Ansichten

Bestimmung der prozentualen Massenanteile in einer wässrigen Lösung
General Chemistry
383.9K Ansichten

Ermittlung der Summenformel
General Chemistry
183.8K Ansichten

Bestimmung der Löslichkeit von ionischen Verbindungen
General Chemistry
141.6K Ansichten

Verwendung eines pH-Meters
General Chemistry
347.0K Ansichten

Einführung in die Titration
General Chemistry
425.8K Ansichten

Ideale Gasgleichung
General Chemistry
79.4K Ansichten

Spektrophotometrische Bestimmung der Gleichgewichtskonstante
General Chemistry
158.9K Ansichten

Das Prinzip von Le Chatelier
General Chemistry
265.8K Ansichten

Gefrierpunktserniedrigung zur Bestimmung einer unbekannten Substanz
General Chemistry
160.8K Ansichten

Bestimmung der Ratengleichung und der Reaktionsreihenfolge
General Chemistry
196.4K Ansichten

Verwendung der dynamischen Differenzkalorimetrie zur Messung von Enthalpieänderungen
General Chemistry
44.8K Ansichten
Copyright © 2025 MyJoVE Corporation. Alle Rechte vorbehalten