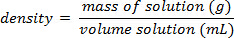קביעת הרכב אחוז המסה בפתרון מימי
Overview
מקור: המעבדה של ד"ר ניל אברמס — מכללת SUNY למדעי הסביבה ויערנות
קביעת הרכב הפתרון היא טכניקה אנליטית וזיהוי משפטית חשובה. כאשר פתרונות נעשים עם מים, הם מכונים מימית, או מכילים מים. המרכיב העיקרי של פתרון מכונה הממס, והרכיב המשני המומס נקרא solute. solute מומס בממס כדי להפוך את הפתרון. מים הם הממס הנפוץ ביותר בחיי היומיום, כמו גם כמעט כל המערכות הביולוגיות. במעבדות כימיה, הממס עשוי להיות נוזל אחר, כמו אצטון, אתר, או אלכוהול. ההולוט יכול להיות נוזל או מוצק, אבל הניסוי הזה מתייחס רק לקביעת המוצקים.
Procedure
1. אחוז לפי מסה - ישיר
- מניחים נפח קטן של תמיסה לתוך נקייה ומיובשת בתנור או צלחת התגבשות.
- לאחר קביעת המסה הכוללת המדויקת של הפתרון, מחממים את הכוס או המנה על כיריים או בתנור כדי לגרש את המים. אידוי איטי הוא השיטה הטובה ביותר, כמו רותחים יכולים לגרום התזה של הפתרון.
- לאחר הממס התאדה, לקרר את מוצק הנותרים (solute) ולקבוע את המסה.
- חשב את אחוז המסה כ:
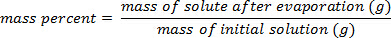
2. אחוז לפי מסה - שימוש בעקומת כיול<
Results
באמצעות הדוגמה המוצגת באיור 1, קבוצה של תקני נתרן כלורי הוכנה עם קומפוזיציות אחוז מסה של 5.000%, 10.00%, 15.00%, 20.00% ו-25.00% של תמיסה. הצפיפות הנמדדת הייתה 1.025, 1.042, 1.060, 1.070 ו-1.090 גרם/מ"ל, בהתאמה. לאחר התוויית נתונים אלה, קו מגמה ליניארי מוחל, מתאים את המשוואה y = 3.446 x 10-3x + 1.0048, כאשר y...
Application and Summary
אחוז הסוכר בסודה, יכול בקלות להיקבע העיקרון של הרכב אחוז המוני. ההליך לביצוע ניסוי זה יהיה למדוד את המסה והנפח של סודה מנויה (ללא בועות) ולחשב את צפיפות הפתרון. עקומת כיול של צפיפות לעומת אחוז במסה עבור מספר פתרונות סוכרוז (סוכר) סטנדרטיים יהיה צורך ליצור, ולאחר מכן כיול זה יכול לשמש כדי לפתור עבור אחוז ...
Skip to...
Videos from this collection:

Now Playing
קביעת הרכב אחוז המסה בפתרון מימי
General Chemistry
383.1K Views

כלי זכוכית ושימושים נפוצים במעבדה
General Chemistry
653.4K Views

פתרונות וריכוזים
General Chemistry
273.1K Views

קביעת הצפיפות של מוצק ונוזל
General Chemistry
554.7K Views

קביעת הנוסחה האמפירית
General Chemistry
180.0K Views

קביעת כללי המסיסות של תרכובות יוניות
General Chemistry
141.1K Views

שימוש במד pH
General Chemistry
344.3K Views

מבוא לתמצית
General Chemistry
423.4K Views

חוק הגז האידיאלי
General Chemistry
78.1K Views

קביעת ספקטרופוטומטריה של קבוע שיווי משקל
General Chemistry
158.3K Views

עקרון לה שאטלייה
General Chemistry
264.0K Views

דיכאון נקודת הקפאה כדי לקבוע תרכובת לא ידועה
General Chemistry
160.4K Views

קביעת חוקי התעריפים וסדר התגובה
General Chemistry
195.7K Views

שימוש בסריקה דיפרנציאלית קלורימטריה למדידת שינויים באנטלפיה
General Chemistry
44.4K Views

מתחמי כימיה של תיאום
General Chemistry
91.2K Views
Copyright © 2025 MyJoVE Corporation. All rights reserved