A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Subcutaneous Infection of Methicillin Resistant Staphylococcus Aureus (MRSA)
In This Article
Summary
Murine skin and soft tissue infection model is utilized for assessing the virulence function of methicillin resistant Staphylococcus aureus (MRSA) and the host immunological responses. Here, we presented a subcutaneous infection model for skin and soft tissue infection.
Abstract
MRSA is a worldwide threat to public health, and MRSA skin and soft-tissue infections now account for more than half of all soft-tissue infections in the United States. Among soft-tissue infections, myositis, pyomyositis, and necrotizing fasciitis have been increasingly reported in association with MRSA arising from the community. To understand the interplay between MRSA and host immunity leading to more severe infection, the availability of animal models is critical, permitting the study of host and bacterial factors. Several infection models have been introduced to assess the pathogenesis of S. aureus during superficial skin infection. Here, we describe a subcutaneous infection model that examines the skin, subcutaneous, and muscle pathologies.
Protocol
1. Preparing the MRSA for Infection (two Days Prior to Infection)
- Inoculate a loopful of MRSA from a stock culture to a blood agar (Trypticase Soy Agar (TSA)) plate.
- Check the hemolysis phenotype (a clear zone around each colony) on the blood agar plate.
- Pick a colony that has a hemolytic phenotype that is consistent with other experiments.
- Inoculate the colony into 3 mL Todd Hewitt Broth (THB),with the appropriated antibiotic when necessary, in a 15 mL snap-capped tube.
- Incubate at 37°C overnight with shaking at 220 rpm.
2. Preparing the Mice for Infection (one Day Prior to Infection)
- Depending on the size of the mice, shave the fur off a 3 x 4 cm area on the back of mice.
- Apply ~5 mm3 hair remover cream (purchased from a local drug store) to the shaved area.
- Allow the hair remover cream to incubate on the skin surface for approximately 1 min.
- Wet paper towels with ddH2O.
- Wipe off the hair remover cream with the wet paper towel.
3. Preparing the MRSA for Infection (on the Day of Infection)
- Dilute the overnight bacterial culture at 1:500 to 1:1000 with 10 mL of pre-warmed THB, in a 50 mL screw-capped tube.
- Incubate at 37°C for approximately 2.5 hours with shaking at 220 rpm, until A540 reaches 2.5.
- Collect MRSA by centrifugation at 3,225 x g for 10 min at 4°C.
- Discard the supernatant.
- Resuspend the bacterial pellet in 10 mL Dulbecco's phosphate buffered saline (DPBS).
- Repeat steps 3 and 4.
- Resuspend the bacterial pellet in DPBS at a desired concentration.
- Serially dilute the bacterial suspension from 101 to 108.
- Plate the diluted bacterial suspension onto blood agar plates.
- Incubate the plates at 37°C overnight.
- Observe and record the hemolytic phenotype of the inocula.
- Count the Colony Forming Units (CFU) number on the plate.
- Calculate the inocula based on the CFU number on the plate.
4. Subcutaneous Infection of MRSA (on the Day of Infection and 3 Days Post-infection)
- Inject subcutaneously 100 μL bacterial suspension in the shaved area.
- Observe the animals for 3 to 5 hours after injection to make sure the mice are alive.
- Observe the lesion on the back of the animals daily and record the lesion area daily when it is needed. Lesion observation should include recording of lesion size and morphology. Both skin and muscle lesions are quantitated by multiplying the length and width of the lesion. Irregularly-shaped lesions need to be broken down into smaller symmetrical pieces, and each piece measured by the same method. Lesion measurements may also use a computer-assisted histomorphometric assessment program (ImageJ; open-source available from the NIH at http://rsb.info.nih.gov/ij/) 2.
- Sacrifice the animals on day 3 post-infection by inhalation of isoflurane followed by cervical dislocation.
- Observe and record the lesion on the skin.
- Nick the skin with a pair of sterile scissors.
- Peel the skin off carefully and observe and record the lesion on the muscle.
- Excise the lesion and approximately 2 - 5 mm of the surrounding area.
- Collect spleen and kidneys.
- Homogenize the tissues using a tissue homogenizer or by repeated application of a plunger from a 1 mL syringe to a microfuge tube containing the tissue and 100 μL of DPBS.
- Add 900 μL of DPBS to each tube.
- Vortex the samples for 5 min.
- Serially dilute the suspension with DPBS 101 to 106.
- Plate the diluted suspensions on THA plates.
5. Representative Results:
- Immediately after each subcutaneous injection, a bleb will be observed on the skin surface when infection is done correctly (Figure 1A). If no bleb is observed on the skin surface, the injection may be too deep, and this may affect the outcome of the infection (lesion size, CFU).
- The bacterial suspension needs to be serially diluted and the CFU need to be examined on blood agar plates for each experiment. This step will yield important information: 1) whether the inocula have homogeneous phenotype; 2) the exact viable bacterial count for the infection.
- The lesions and bacterial survival may be assessed at various time points post-infection. Here, we examined the infection on day 3 post-infection. The lesion sizes are analyzed on the skin surface (Figure 1B: 1x 109; Figure 1D: 1 x 107 CFU) and on the muscle surface (Figure 1C: 1x 109; Figure 1E: 1 x 107 CFU). The lesion area equals length (mm) x width (mm) may be measured for assessing the tissue damage (Figs. 2 A and B). This result may help determine the extent of tissue damage caused by a given virulence factor. Additionally, the lesions are excised and homogenized in DPBS, and plated to quantitate the CFU number (Figure 2C), which indicates the viability of the bacteria at the infection site.
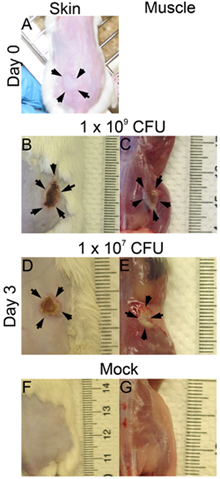
Figure 1. The skin and muscle lesions. CD1 mice were inoculated subcutaneous on one flank with MRSA (LAC). Pictures were taken at time 0 and day 3 post-infection. (A) Time 0 post-infection (p.i.), (B)-(G) Day 3 p.i.; B, D. and F: skin lesions; C, E, and G: muscle lesions; B and C: 1x 109 CFU infected; D and E: 1x 107 CFU infected; F and G: mock.
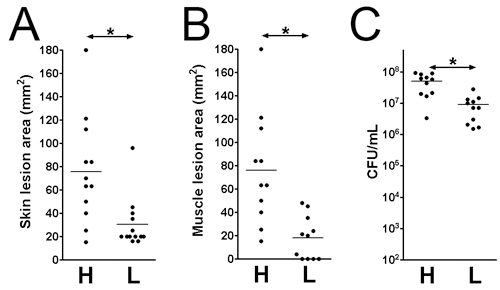
Figure 2. Skin lesion size and viable bacteria count at the infection site on day 3 post-infection. Skin and muscle lesions on MRSA (LAC) infected CD1 mice. (A) Skin lesion size. (B) Muscle lesion size. (C) Total tissue CFU. H: 1 x 109 CFU inocula; L: 1 x 107 CFU inocula. *: p < 0.05, Mann-Whitney test.
Discussion
- The murine skin and soft tissue infection model is a powerful tool for in vivo virulence assessment of a pathogen. The pathogenicity of S. aureus in skin and soft tissue infection may vary depending on a number of parameters. These include inoculum size, bacterial growth phase, depth of inoculation, age of mice, and the mouse genetic background 7, 8. When examining the virulence function using this model, it is critical to control for these parameters to ensure that results will be consistent. We have ...
Disclosures
No conflicts of interest declared.
Acknowledgements
This work was supported by a Burroughs-Wellcome Career Award and by National Institutes of Health grant AI074832 to G. Y. Liu.
Materials
| Material Name | Type | Company | Catalogue Number | Comment |
|---|---|---|---|---|
| Name | Company | Catalog Number | Comments | |
| THB | VWR | 95025-314 | ||
| DPBS | Invitrogen | 21-031-CV | ||
| 1 ml syringe | BD | 309602 | ||
| 27G1/2 needle | BD | 305109 | ||
| Sheep Blood Agar (TSA) | VWR | 90004-328 |
References
- Ahn, J. Y., Song, J. Y., Yun, Y. S., Jeong, G., Choi, I. S. Protection of Staphylococcus aureus-infected septic mice by suppression of early acute inflammation and enhanced antimicrobial activity by ginsan. FEMS Immunol Med Microbiol. 46, 187-197 (2006).
- Girish, V., Vijayalakshmi, A. Affordable image analysis using NIH Image/ImageJ. Indian J Cancer. 41, 47-47 (2004).
- Hahn, B. L., Onunkwo, C. C., Watts, C. J., Sohnle, P. G. Systemic dissemination and cutaneous damage in a mouse model of staphylococcal skin infections. Microb Pathog. 47, 16-23 (2009).
- Hruz, P., Zinkernagel, A. S., Jenikova, G., Botwin, G. J., Hugot, J. P., Karin, M., Nizet, V., Eckmann, L. NOD2 contributes to cutaneous defense against Staphylococcus aureus through alpha-toxin-dependent innate immune activation. Proc Natl Acad Sci U S A. 106, 12873-12878 (2009).
- Ji, Y., Zhang, B., Van, S. F., Horn, P. W. a. r. r. e. n., Woodnutt, G., Burnham, M. K., Rosenberg, M. Identification of critical staphylococcal genes using conditional phenotypes generated by antisense RNA. Science. 293, 2266-2269 (2001).
- Somerville, G. A., Beres, S. B., Fitzgerald, J. R., DeLeo, F. R., Cole, R. L., Hoff, J. S., Musser, J. M. In vitro serial passage of Staphylococcus aureus: changes in physiology, virulence factor production, and agr nucleotide sequence. J Bacteriol. 184, 1430-1437 (2002).
- Tseng, C. W., Kyme, P., Low, J., Rocha, M. A., Alsabeh, R., Miller, L. G., Otto, M., Arditi, M., Diep, B. A., Nizet, V., Doherty, T. M., Beenhouwer, D. O., Liu, G. Y. Staphylococcus aureus Panton-Valentine leukocidin contributes to inflammation and muscle tissue injury. PLoS One. 4, e6387-e6387 (2009).
- von Kockritz-Blickwede, M., Rohde, M., Oehmcke, S., Miller, L. S., Cheung, A. L., Herwald, H., Foster, S., Medina, E. Immunological mechanisms underlying the genetic predisposition to severe Staphylococcus aureus infection in the mouse model. Am J Pathol. 173, 1657-1668 (2008).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved