A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Temperature-Controlled Assembly and Characterization of a Droplet Interface Bilayer
In This Article
Summary
This protocol details the use of a feedback temperature-controlled heating system to promote lipid monolayer assembly and droplet interface bilayer formation for lipids with elevated melting temperatures, and capacitance measurements to characterize temperature-driven changes in the membrane.
Abstract
The droplet interface bilayer (DIB) method for assembling lipid bilayers (i.e., DIBs) between lipid-coated aqueous droplets in oil offers key benefits versus other methods: DIBs are stable and often long-lasting, bilayer area can be reversibly tuned, leaflet asymmetry is readily controlled via droplet compositions, and tissue-like networks of bilayers can be obtained by adjoining many droplets. Forming DIBs requires spontaneous assembly of lipids into high density lipid monolayers at the surfaces of the droplets. While this occurs readily at room temperature for common synthetic lipids, a sufficient monolayer or stable bilayer fails to form at similar conditions for lipids with melting points above room temperature, including some cellular lipid extracts. This behavior has likely limited the compositions—and perhaps the biological relevance—of DIBs in model membrane studies. To address this problem, an experimental protocol is presented to carefully heat the oil reservoir hosting DIB droplets and characterize the effects of temperature on the lipid membrane. Specifically, this protocol shows how to use a thermally conductive aluminum fixture and resistive heating elements controlled by a feedback loop to prescribe elevated temperatures, which improves monolayer assembly and bilayer formation for a wider set of lipid types. Structural characteristics of the membrane, as well as the thermotropic phase transitions of the lipids comprising the bilayer, are quantified by measuring the changes in electrical capacitance of the DIB. Together, this procedure can aid in evaluating biophysical phenomena in model membranes over various temperatures, including determining an effective melting temperature (TM) for multi-component lipid mixtures. This capability will thus allow for closer replication of natural phase transitions in model membranes and encourage the formation and use of model membranes from a wider swath of membrane constituents, including those that better capture the heterogeneity of their cellular counterparts.
Introduction
Cellular membranes are selectively permeable barriers comprised of thousands of lipid types1, proteins, carbohydrates, and sterols that encapsulate and subdivide all living cells. Understanding how their compositions affect their functions and revealing how natural and synthetic molecules interact with, adhere to, disrupt, and translocate cellular membranes are, therefore, important areas of research with wide-reaching implications in biology, medicine, chemistry, physics, and materials engineering.
These aims for discovery directly benefit from proven techniques for assembling, manipulating, and studying model membranes—including lipid bilayers assembled from synthetic or naturally occurring lipids—that mimic the composition, structure, and transport properties of their cellular counterparts. In recent years, the droplet interface bilayer (DIB) method2,3,4 for constructing a planar lipid bilayer between lipid-coated water droplets in oil has received significant attention5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23, and has demonstrated practical advantages over other approaches for model membrane formation: the DIB method is simple to perform, requires no sophisticated fabrication or preparation (e.g., "painting") of a substrate to support the membrane, consistently yields membranes with superior longevity, allows for standard electrophysiology measurements, and simplifies the formation of model membranes with asymmetric leaflet compositions3. Because the bilayer forms spontaneously between droplets and each droplet can be tailored in position and makeup, the DIB technique has also attracted considerable interest in developing cell-inspired material systems that build on the use of stimuli-responsive membranes18,24,25,26,27,28,29, balanced compartmentalization and transport14,30,31, and tissue-like materials17,23,32,33,34,35,36.
The majority of published experiments on model membranes, including those with DIBs, have been performed at room temperature (RT, ~20-25 °C) and with a handful of synthetic lipids (e.g., DOPC, DPhPC, etc.). This practice limits the scope of biophysical questions that can be studied in model membranes and, based on observation, it can also restrict the types of lipids that can be used to assemble DIBs. For example, a synthetic lipid such as DPPC, which has a melting temperature of 42 °C, does not assemble tightly-packed monolayers or form DIBs at RT37. DIB formation at room temperature has also proven difficult for natural extracts, such as those from mammals (e.g., brain total lipid extract, BTLE)38 or bacteria (e.g., Escherichia coli total lipid extract, ETLE)37, which contain many different types of lipids and originate from cells that reside at elevated temperatures (37 °C). Enabling study of diverse compositions thus provides opportunities to understand membrane-mediated processes in biologically relevant conditions.
Raising the temperature of the oil can serve two purposes: it increases the kinetics of monolayer assembly and it can cause lipids to undergo a melting transition to reach a liquid disordered phase. Both consequences aid in monolayer assembly39, a pre-requisite for a DIB. In addition to heating for bilayer formation, cooling the membrane after the formation can be used to identify thermotropic transitions in single lipid bilayers38, including those in natural lipid mixtures (e.g., BTLE) that can be difficult to detect using calorimetry. Aside from assessing thermotropic transitions of lipids, precisely varying the temperature of the DIB can be used to study temperature-induced changes in membrane structure38 and examine how lipid composition and fluidity affect the kinetics of membrane-active species (e.g., pore-forming peptides and transmembrane proteins37), including mammalian and bacterial model membranes at a physiologically relevant temperature (37 °C).
Herein, a description of how to assemble a modified DIB oil reservoir and operate a feedback-temperature controller to enable monolayer assembly and bilayer formation at temperatures higher than RT will be explained. Distinguished from a previous protocol40, explicit detail is included regarding the integration of instrumentation needed for measuring and controlling temperature in parallel to assembly and characterization of the DIB in the oil reservoir. The procedure will thus enable a user to apply this method for forming and studying DIBs across a range of temperatures in a variety of scientific contexts. Moreover, the representative results provide specific examples for the types of measurable changes in both membrane structure and ion transport that can occur as temperature is varied. These techniques are important additions to the many biophysical studies that can be designed and performed effectively in DIBs, including studying the kinetics of membrane-active species in different membrane compositions.
Protocol
1. Heated fixture preparation
- Gather 2 pieces of 1 mm thick insulative rubber trimmed to 25 mm x 40 mm in width and length, respectively, 2 pieces of a 6 mm-thick rubber that are also 25 mm x 40 mm, a prepared aluminum base fixture assembly, and an acrylic oil reservoir that fits in the viewing window of the aluminum base fixture (see Figures S1, S2, and S3 for details on fabrication and an exploded view of assembly). Prepare the aluminum fixture first by attaching to the bottom of the fixture a glass coverslip viewing window with UV curable adhesive and adhering 1 resistive heating element to the top of each 25 mm x 25 mm side flange of the fixture.
- Place the thinner rubber pieces onto the stage of the microscope such that the long edge of each piece is tangential to the stage opening as shown in (Figure 1).
- Position the aluminum-base fixture on top of the insulative pads with the viewing window of the fixture centered above the objective lens. Proper alignment is required for imaging the connected droplets.
- Place a thicker piece of rubber on top of each resistive heating element and use a microscope stage clip to hold it in place. These pieces protect the heating elements from damage caused by the stage clips and insulate against accidental electrical shorting between the heating elements and both the aluminum fixture and the microscope stage.
- Carefully bend the measurement-end of a thermocouple to achieve a 90° angle at ~4 mm from the end.
- Insert the bent tip of the thermocouple into the lower left corner of the aluminum fixture and gently secure it with the locking screw.
- Place the acrylic reservoir into the well of the aluminum fixture. This is done prior to adding hexadecane oil to the well (step 1.8) of the aluminum fixture to minimize the risk of trapping air bubbles between the viewing window and the bottom of the acrylic reservoir, which can obstruct the view of the droplets.
NOTE: Oil added to the viewing compartment of the aluminum fixture is used to match the refractive indices of the acrylic and glass for clearer imaging of the droplets contained within the acrylic reservoir. Thus, it is worth noting that oil in the well of the aluminum fixture does not contact the contents of the acrylic reservoir and rigorous cleaning of the aluminum fixture is not required. - Dispense ~1,000 µL of hexadecane oil into the well of aluminum fixture (i.e., between the walls of the acrylic reservoir and aluminum fixture), taking care to not overfill. The oil level in the well of the aluminum fixture should be as high as allowable to maximize surface area for heat transfer, while not allowing oil to spill over the edges of the fixture onto the microscope stage or objective lens.
- Dispense ~1,000 µL of hexadecane oil into the acrylic reservoir, while remaining mindful to not overfill.
NOTE: The acrylic reservoir should always be thoroughly cleaned between experiments. The user must employ a regiment consisting of successive rinses with ethyl-alcohol and deionized water followed by drying in a desiccator bowl for over 12 h.

Figure 1: Heated stage assembly. Images show the assembly of the thermally conductive fixture and oil reservoir for DIB formation; numbers beneath each image identify the corresponding step of the protocol. Please click here to view a larger version of this figure.
2. Instrumentation for simultaneous feedback temperature control and electrical characterization of a DIB
NOTE: This protocol integrates the following instruments for enabling feedback temperature control and simultaneous electrical characterization of a DIB: a personal computer (PC) with two available universal serial bus (USB) connections, a patch clamp amplifier paired to a dedicated data acquisition (DAQ-1) system, a waveform generator, a second programable DAQ (DAQ-2) with voltage output and temperature input modules, and a power supply/amplifier. The following steps describe the necessary connections of these instruments (as illustrated in Figure 2a) needed for isolating the measurement and control of temperature from simultaneous electrophysiology of a DIB. Substitutions for equivalent instruments may be made as required.
- Establish output and input connections to the DAQ-2 modules.
- Select two pairs of screw terminals on the voltage output module for differential voltage connections and attach wire leads to these locations. Odd number terminals are common ground connections, and the even number terminals are ungrounded outputs, as shown in (Figure 2c). Connect each of these two pairs of lead wires to separate screw-terminal-BNC adapters and then connect each adapter to a separate BNC cable used to route voltage signals to other instruments.
NOTE: In this setup, differential connections at terminals 0 and 1 are assigned for the temperature control output to the power amplifier, while another pair of connections at terminals 6 and 7 are designated for voltage output to be sent to the droplets via the patch clamp amplifier. - Referring to (Figure 2c), select one set of thermocouple terminals (e.g., terminals 2 and 3 are designated as the TC1 pair) on the thermocouple input module and connect to it the thermocouple wires.
- Select two pairs of screw terminals on the voltage output module for differential voltage connections and attach wire leads to these locations. Odd number terminals are common ground connections, and the even number terminals are ungrounded outputs, as shown in (Figure 2c). Connect each of these two pairs of lead wires to separate screw-terminal-BNC adapters and then connect each adapter to a separate BNC cable used to route voltage signals to other instruments.
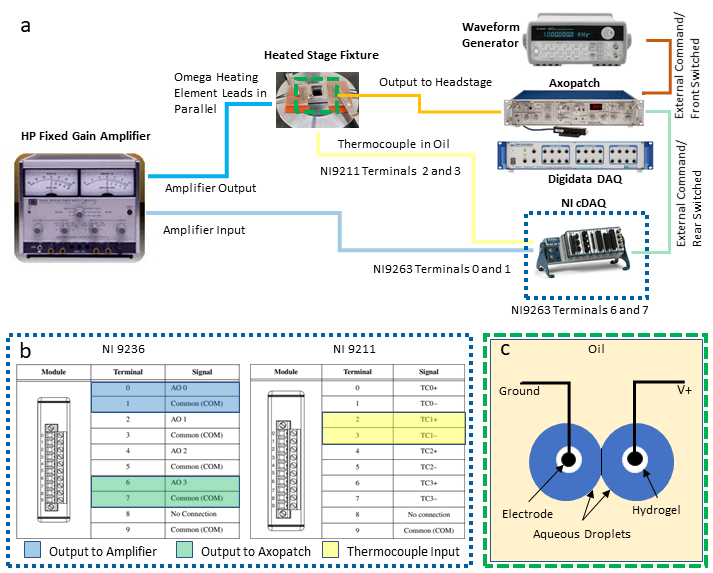
Figure 2: System wiring connections. A schematic of the devices and wiring required for the system is shown in (a), while a detailed look at the DAQ-2 connections is provided in (b). The illustration in (c) shows aqueous droplets on hydrogel-coated electrodes submerged in oil for DIB formation. The two electrodes are connected to the grounded and ungrounded (V+) connections, respectively, on the headstage unit of the patch clamp amplifier. Please click here to view a larger version of this figure.
- After electrical connections to the DAQ-2 modules have been made, connect the DAQ-2 chassis to a PC via a USB connection and connect to an electrical power source. Then confirm successful driver and software installation prior to use with a commercial software.
- Configure and connect a power amplifier between DAQ-2 and resistive heating elements.
- Configure the amplifier to operate in fixed-gain amplification mode with a gain of 10X.
- Using a banana jack-BNC adapter, connect the BNC cable originating from terminals 0 and 1 on the voltage output module (Figure 2b) to the input connections on the power amplifier.
- Using additional BNC adapters and cabling, connect the output terminals of the power amplifier to both sets of heating elements, which are wired in parallel to one another and the amplifier to ensure that both elements maintain the same voltage drop during use.
- Establish necessary connections for the electrophysiology equipment.
- Connect a BNC cable originating from terminals 6 and 7 on the voltage output module (Figure 2b) to the Rear Switched External Command BNC connectors on the rear of the patch clamp amplifier.
- Connect a second BNC cable between the output of waveform generator and the Front Switch External Command connection on the rear of the patch clamp amplifier.
NOTE: These two connections provide alternative methods for generating voltage waveforms that are applied to the droplet electrodes via the patch clamp amplifier. The waveform generator is especially useful for generating triangular waveform voltages used to measure membrane capacitance. The user may decide which, if either, are necessary for their own application. - With a third BNC cable, connect the output of the measured current located on the front panel of the patch clamp amplifier to an available analog input BNC connector on the front of DAQ-1.
- With a fourth BNC cable, connect the output of measured membrane voltage (on the rear of the patch clamp amplifier) to a separate analog input connector on DAQ-1. This enables digitization of the voltage applied across the electrodes.
- With the two droplet electrodes prepared and supported on micromanipulators as described in Steps 7-9 in ref.40, connect the electrode leads to the patch clamp headstage, which is the attached via cable to the patch clamp amplifier.
NOTE: The role of the headstage is to control the voltage between the electrodes and measure the resulting current, which is converted into a proportional voltage that gets output by the patch clamp amplifier to DAQ-1. - Connect DAQ-1 to a PC via a USB connection and connect the corresponding power supply cables to both the patch clamp amplifier and DAQ-1.
- Power on all measurement equipment.
NOTE: Perhaps the most important detail in this setup is to ensure the power amplifier output (mA-A) connections are electrically isolated from the headstage unit of the patch clamp amplifier, which uses a sensitive circuit to measure pA-nA level currents in a DIB.
3. Feedback temperature control of droplet interface bilayers
NOTE: The following steps for operating the feedback temperature control system are based on a custom graphical user interface (GUI) created for implementing proportional-integral (PI) feedback temperature control40,41 (see Supplementary Coding Files). Other software and control algorithms may be used instead. A copy of this program is provided to the reader with the supplementary information for the paper, however the user is responsible to configure it for their own equipment and needs.
- Start the DAQ-2 software on the PC and open the temperature control program file. Once the GUI opens, open the program again by clicking on the folder icon in the bottom left corner of the GUI and selecting the temperature control program (Figure 3).
- Enter appropriate numerical values for the proportional control gain (KP) and integral control gain (KI).
NOTE: KP and KI values of 0.598 and 0.00445, respectively, were found to work well in the setup. These values were determined iteratively through simulation using a system model that incorporates parameters obtained from measured open-loop heating responses (see Figure 4). During open-loop heating, the prescribed heating power is independent of the measured temperature. In contrast, closed-loop heating consists of continually adjusting the applied power to the heaters in a manner that helps drive the measured temperature closer to the desired temperature. This is achieved herein using a PI control scheme. - To test the temperature control scheme, enter a desired set point temperature (above room temperature) and then turn on feedback temperature control within the GUI. Observe the measured temperature signal under feedback (closed-loop) control, which is displayed in the GUI for the next few minutes. If the measured temperature of the oil greatly overshoots the desired temperature, reacts too slowly to changes, or fails to converge to the desired set point, the user will need to adjust the control gains to achieve desired closed loop performance.
NOTE: The program defines a saturation limit for the power (and thus voltage) supplied to the resistive heating elements. For example, two elements reported herein consume up to 5 W power each. Wiring them in parallel means that total power consumption should not exceed 10 W. The user is advised to consider the maximum amount of power that should be supplied to the devices and know that this limit can affect the speed at which the closed-loop system will respond to desired temperature changes. Higher power heating elements enable faster heating and higher set point temperatures but require higher supplied currents for heating. - With the system tuned to acceptable closed-loop performance, enter the desired oil temperature for DIB formation as the set point in the GUI.
NOTE: For example, a set point temperature of 60 °C yielded good results in experiments with BTLE liposomes in the aqueous droplets37. The user is referred elsewhere2,40 for protocols explaining DIB assembly between droplets hanging on wire-type electrodes and configuration of electrophysiology equipment using the patch clamp amplifier, DAQ-1, and electrophysiology measurement software. Specifically, the protocol by Najem, et al.40 can be closely followed until Step 13. Beyond that step, a slightly different approach is employed for successful monolayer and bilayer formation when using lipids that require heating to promote monolayer or bilayer formation. - Lower the tips of the silver/silver chloride (Ag/AgCl) electrodes into the oil until they nearly touch the bottom of the acrylic reservoir. This positioning of the electrode tips is crucial for keeping the droplet on the electrode in heated oil, where convective currents in the oil have been observed to detach droplets from the hydrogel-coated electrodes (Figure 2c).
- Pipette a 250 nL droplet of aqueous lipid solution containing 2 mg/mL of BTLE, 100 mM potassium chloride (KCl), and 10 mM 3-(N-morpholino) propanesulfonic acid (MOPS) onto each electrode tip and let them incubate in the heated oil for a minimum of 10 minutes to promote monolayer formation.
- Cover the headstage and heated stage fixture with a grounded Faraday cage.
- Bring the droplets into gentle contact by slowly
manipulating the horizontal positions of the electrodes until the user sees the droplets deform from contact or begin to displace one another and wait a few minutes until bilayer formation commences. If after several minutes a bilayer has not formed, the droplets can be coerced together more to facilitate bilayer formation. The formation of a thinned interfacial bilayer can be confirmed through visual inspection (Figure 5a) or by measuring the increase in the amplitude of a square-waveform capacitive current induced by a waveform generator outputting a 10 mV, 10 Hz triangular voltage22. Allow the bilayer to equilibrate for a minimum of 10 minutes to reach a steady interfacial area, upon initial formation and prior to subsequent characterization at the initial set point.
NOTE: The type of oil can have a significant impact on bilayer thinning, membrane thickness, and inter-droplet contact angle. In general, the smaller the oil molecule the more easily it can remain in the hydrophobic core of the bilayer occupied by lipid acyl chains. Oil retention increases both monolayer and bilayer tensions and thickness and decreases the area and angle of contact between droplets. These metrics signify a weaker state of adhesion. Larger, bulkier molecules exert the opposite effect. For example, squalene is a bulkier molecule than alkanes such as hexadecane, which enables it to be readily excluded from between monolayers during bilayer thinning. As such, DIBs formed in squalene are thinner, they display higher contact areas and angles, and they exhibit higher free energies of formation22,42 (a measure of droplet-droplet adhesion).
4. Characterization of temperature-dependent behaviors in DIBs
NOTE: Many physical processes can be studied in DIB-based model membranes, including how changes in temperature affect the structure and transport properties of the membrane. The following steps should be performed after successful bilayer formation at a desired temperature.
- Measure the nominal capacitance of the membrane while lowering the temperature of the oil bath from a set point that permits bilayer formation to identify thermotropic phase transitions of the lipids in the membrane38.
- Right click the temperature graph on the GUI and clear the displayed data. This ensures sufficient space in the buffer is available for subsequent recordings.
- Using the waveform generator connected to the patch clamp amplifier, apply a triangular voltage waveform (e.g., 10 mV, 10 Hz) across the DIB electrodes and record the induced current response through the bilayer.
- Cool the bilayer by reducing the set point temperature in 5 °C increments and waiting a minimum of 5 min at the new steady state temperature between temperature changes until the desired temperature is achieved. Alternatively, try passively cooling the bilayer by turning off the feedback control system. Be aware, however, that experiments implementing passive cooling from 50-60 °C resulted in higher rates of coalescence.
- After the oil bath and bilayer cool to the desired minimum temperature, right click the temperature graph in the GUI again and export the temperature data versus time to a spreadsheet software. Stop the current recording.
- From the measured current, calculate the nominal capacitance of the square wave-current response versus time during the cooling period.
- Plot nominal capacitance (C) versus temperature (T) to observe how membrane capacitance changed. Locate nonmonotonic changes in C versus T to identify TM.
NOTE: Nominal capacitance can be calculated from the amplitude of square-wave current43 (|I|) using the relationship |I| = C dv/dt, where dv/dt is equal to four times the product of the voltage amplitude (|V|) and frequency (f) of the applied triangular voltage. From these equations, C = |I|/(4|V|f).
- Similarly, assess the quasi-static specific capacitance (Cm) of the bilayer at fixed temperatures by successively incrementing the temperature of the oil bath and the bilayer area.
- Change the set point temperature in 10 °C increments using the GUI and allow the system to equilibrate to the new temperature.
- Perform Step 4.1.2 to initiate the measurement of capacitive current and recording.
- Change the bilayer area by carefully adjusting the positions of the electrodes using the micro-manipulators (i.e., separating the electrodes reduces bilayer area). Allow for the square-wave current to reach a steady state amplitude and collect images of the DIB to enable calculation of membrane area versus time by using a camera mounted to the microscope to image the bilayer as seen from the aperture of the microscope stage. Simultaneously, add a digital tag in the current recording software to mark the corresponding timepoint for image collection.
NOTE: Micro-manipulators allow for the precise control of the electrodes and thus gentle contact between droplets. Coarse manipulation of the droplets can lead to a failed experiment by coalescence of the droplets or by causing a droplet to fall off the electrode. As discussed elsewhere22, bilayer area is calculated from the contact length between droplets, which appear as overlapping circles in a bottom-view image. The positions and dimensions of the droplets, and the length of the contact line, can be calculated using an image-processing software or with other scientific programming tools. - Repeat Step 4.2.1.2 a minimum of 4 times to obtain a total of 5 DIB images and steady-state regions of bilayer current.
- Repeat Step 4.2.1 at each desired temperature.
- At the tagged timepoints corresponding to steady-state bilayer areas for acquired images, analyze the current recordings and DIB images to extract C and A data for each temperature.
- Plot C versus A data for each temperature and compute the slope of a first-order regression, which represents the Cm of the bilayer at each temperature22.
- Plot values of Cm obtained from Step 4.2.4 versus T.
- Examine the Cm versus T data for non-monotonic variations to identify melting temperatures, TM.
- Change the set point temperature in 10 °C increments using the GUI and allow the system to equilibrate to the new temperature.
- Assess the dynamics of voltage-dependent ion channel formation by generating a dc voltage step input across the bilayer.
- Set Initial Voltage to the desired step value in mV (e.g.,100 mV).
- Set Final Voltage and Step Size to a value higher than the desired step (e.g., 110 mV final voltage and 110 mV step size).
- Set a desired duration time for the step input in seconds (e.g., 90 s).
- Choose the desired polarity for the step input (e.g., positive).
- Switch the patch clamp amplifier to send to the headstage the command voltage originating from the GUI/voltage output module.
- Initiate current recordings.
- Turn on the voltage and record the induced current response, which should exhibit an S -shaped response to a critical voltage (e.g., ~70 mV for 1 µg/mL Mz in 2 mg/mL BTLE).
- Separately, dynamic current-voltage relationships for a membrane can be obtained at desired temperatures to reveal voltage-dependent relationships, such as ion channel behaviors.
- Switch the patch clamp amplifier to send to the headstage the command voltage originating from the waveform generator and initiate current recordings.
- On the waveform generator, output a continuous sinusoidal waveform with a desired amplitude, offset, and frequency.
- Record the induced current response across one or multiple cycles.
- Repeat as desired for different sine wave amplitudes and frequencies and temperatures.
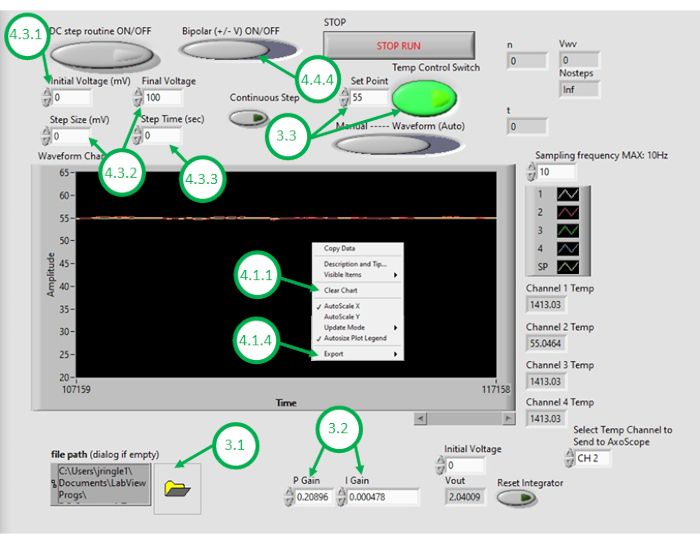
Figure 3: The temperature control GUI. This figure highlights and labels the critical steps required to use the program's GUI to control the temperature of the oil bath. Please click here to view a larger version of this figure.
Results
Figure 1 shows how the aluminum fixture and acrylic oil reservoir are prepared on the microscope stage for DIB formation. Assembly steps 1.2-1.4 serve to thermally insulate the fixture from the stage for more efficient heating. Steps 1.5-1.7 show how to properly attach the thermocouple to the fixture and position the oil reservoir, and steps 1.8 -1.9 show recommended locations for dispensing oil into these pieces.
Figure 2 outlin...
Discussion
The protocol described herein provides instructions for assembling and operating an experimental system to control the temperature of the oil and droplets used to form DIBs. It is especially beneficial for enabling DIB formation using lipids that have melting temperatures above RT. Moreover, by precisely varying the temperature of the oil reservoir, the bilayer temperature can be manipulated to study the effects of elevated temperatures on various membrane properties and characteristics, including capacitance, area, thic...
Disclosures
The authors have no conflicts of interests.
Acknowledgements
Financial support was provided by the National Science Foundation Grant CBET-1752197 and the Air Force Office of Scientific Research Grant FA9550-19-1-0213.
Materials
| Name | Company | Catalog Number | Comments |
| 25 mm x 40 mm x 1 mm insulative rubber (x2) | Any | Insulates the bottom of the aluminum fixture from the stage of the microscope | |
| 25 mm x 40 mm x 6 mm insulative rubber (x2) | Any | Protects heating elements from being damaged by the microscope stage clips and insulates the top of the heating elements. | |
| 3-(N-morpholino) propanesulfonic acid | Sigma Aldrich | M3183 | Buffering agent for lipid solution |
| Acrylic substrate | Fabricated in house | HTD_STG_2 | ~1000 uL acrylic well with a poka-yoke exterior profile to fix orientation |
| Aluminum fixture | Fabricated in house | HTD_STG_1 | Base fixture with an oil well that holds the acylic fixture and includes two flat pads adjacent to the oil well for the heating elements |
| Brain Total Lipid Extract | Avanti | 131101C-100mg | 25 mg/mL porcine lipid extract |
| Compact DAQ Chassis (cDAQ) | National Instruments | cDAQ-9174 | Chassis to house multiple types of sensor measurement or output modules |
| Data Acquisition System (DAQ) | Molecular Devices | Digidata 1440A | High resolution analog to digital converter |
| Fixed gain amplifier/power supply | Hewlitt Packard | HP 6826A | Amplifies DC voltage output from the voltage output module |
| Glass Cover Slip | Corning | CLS284525 | Seals bottom of aluminum base and allows for optical characterization of the bilayer |
| Heating element (x2) | Omega | KHLV-101/5 | 25 mm x 25 mm polymide film kapton heating element with a 5 watt power limit. |
| M3 Stainless Steel Screw | McMaster Carr | 90116A150 | Secures thermocouple to aluminum fixture |
| Patch clamp amplifier | Molecular Devices | AxoPatch 200B | Measures current and outputs voltage to the headstage |
| Personal computer | Any | Computer with mulitiple high speed usb ports and a minimum of 6 Gb of ram | |
| Potassium Chloride | Sigma Aldrich | P3911 | Electrolyte solution of dissociated ions |
| Temperature input module | National Instruments | NI 9211 | Enables open and cold junction thermocouple measurements for the cDAQ chassis |
| Thermocouple | Omega | JMTSS-020U-6 | U-type thermocouple with a diameter of 0.02 inches and 6 inches in length |
| UV Curable Adhesive | Loctite | 19739 | Secures glass coverslip to aluminum base fixture |
| Voltage output module | National Instruments | NI 9263 | Analog voltage output module for use with the cDAQ chassis |
| Waveform generator | Agilent | 33210A | Used to output a 10 mV 10 Hz sinusoidal waveform |
References
- van Meer, G., de Kroon, A. I. P. M. Lipid map of the mammalian cell. Journal of Cell Science. 124 (1), 5-8 (2011).
- Bayley, H., et al. Droplet interface bilayers. Molecular BioSystems. 4 (12), 1191-1208 (2008).
- Hwang, W. L., Chen, M., Cronin, B., Holden, M. A., Bayley, H. Asymmetric droplet interface bilayers. Journal of the American Chemical Society. 130 (18), 5878-5879 (2008).
- Holden, M. A., Needham, D., Bayley, H. Functional bionetworks from nanoliter water droplets. Journal of the American Chemical Society. 129 (27), 8650-8655 (2007).
- Sarles, S. A., Leo, D. J. Physical encapsulation of droplet interface bilayers for durable, portable biomolecular networks. Lab on a Chip. 10 (6), 710-717 (2010).
- Stanley, C. E., et al. A microfluidic approach for high-throughput droplet interface bilayer (DIB) formation. Chemical Communications. 46 (10), 1620-1622 (2010).
- Gross, L. C. M., Heron, A. J., Baca, S. C., Wallace, M. I. Determining membrane capacitance by dynamic control of droplet interface bilayer area. Langmuir. 27 (23), 14335-14342 (2011).
- Huang, J., Lein, M., Gunderson, C., Holden, M. A. Direct quantitation of peptide-mediated protein transport across a droplet, interface bilayer. Journal of the American Chemical Society. 133 (40), 15818-15821 (2011).
- Leptihn, S., Thompson, J. R., Ellory, J. C., Tucker, S. J., Wallace, M. I. In vitro reconstitution of eukaryotic ion channels using droplet interface bilayers. Journal of the American Chemical Society. 133 (24), 9370-9375 (2011).
- Castell, O. K., Berridge, J., Wallace, M. I. Quantification of membrane protein inhibition by optical ion flux in a droplet interface bilayer array. Angewandte Chemie International Edition. 51 (13), 3134-3138 (2012).
- Dixit, S. S., Pincus, A., Guo, B., Faris, G. W. Droplet shape analysis and permeability studies in droplet lipid bilayers. Langmuir. 28 (19), 7442-7451 (2012).
- Elani, Y., deMello, A. J., Niu, X., Ces, O. Novel technologies for the formation of 2-D and 3-D droplet interface bilayer networks. Lab on a Chip. 12 (18), 3514-3520 (2012).
- Michalak, Z., Fartash, D., Haque, N., Lee, S. Tunable crystallization via osmosis-driven transport across a droplet interface bilayer. CrystEngComm. 14 (23), 7865-7868 (2012).
- Punnamaraju, S., You, H., Steckl, A. J. Triggered release of molecules across droplet interface bilayer lipid membranes using photopolymerizable lipids. Langmuir. 28 (20), 7657-7664 (2012).
- Boreyko, J. B., Mruetusatorn, P., Sarles, S. A., Retterer, S. T., Collier, C. P. Evaporation-induced buckling and fission of microscale droplet interface bilayers. Journal of the American Chemical Society. 135 (15), 5545-5548 (2013).
- Leptihn, S., et al. Constructing droplet interface bilayers from the contact of aqueous droplets in oil. Nature Protocols. 8 (6), 1048-1057 (2013).
- Villar, G., Graham, A. D., Bayley, H. A Tissue-like printed material. Science. 340 (6128), 48-52 (2013).
- Barriga, H. M. G., et al. Droplet interface bilayer reconstitution and activity measurement of the mechanosensitive channel of large conductance from Escherichia coli. Journal of The Royal Society Interface. 11 (98), (2014).
- Boreyko, J. B., Polizos, G., Datskos, P. G., Sarles, S. A., Collier, C. P. Air-stable droplet interface bilayers on oil-infused surfaces. Proceedings of the National Academy of Sciences. 111 (21), 7588-7593 (2014).
- Mruetusatorn, P., et al. Dynamic morphologies of microscale droplet interface bilayers. Soft Matter. 10 (15), 2530-2538 (2014).
- Najem, J., Dunlap, M., Sukharev, S., Leo, D. J. The gating mechanism of mechanosensitive channels in droplet interface bilayers. MRS Proceedings. , 1755 (2015).
- Taylor, G. J., Venkatesan, G. A., Collier, C. P., Sarles, S. A. Direct in situ measurement of specific capacitance, monolayer tension, and bilayer tension in a droplet interface bilayer. Soft Matter. 11 (38), 7592-7605 (2015).
- Bayley, H., Cazimoglu, I., Hoskin, C. E. G. Synthetic tissues. Emerging Topics in Life Sciences. 3 (5), 615-622 (2019).
- Oliver, A. E., et al. Protecting, patterning, and scaffolding supported lipid membranes using carbohydrate glasses. Lab on a Chip. 8 (6), 892-897 (2008).
- Maglia, G., et al. Droplet networks with incorporated protein diodes show collective properties. Nature Nanotechnology. 4 (7), 437-440 (2009).
- Najem, J. S., et al. Activation of bacterial channel MscL in mechanically stimulated droplet interface bilayers. Scientific Reports. 5, 13726 (2015).
- Freeman, E. C., Najem, J. S., Sukharev, S., Philen, M. K., Leo, D. J. The mechanoelectrical response of droplet interface bilayer membranes. Soft Matter. 12 (12), 3021-3031 (2016).
- Tamaddoni, N., Sarles, S. A. Toward cell-inspired materials that feel: measurements and modeling of mechanotransduction in droplet-based, multi-membrane arrays. Bioinspiration & Biomimetics. 11 (3), 036008 (2016).
- Restrepo Schild, V., et al. Light-patterned current generation in a droplet bilayer array. Scientific Reports. 7, 46585 (2017).
- Milianta, P. J., Muzzio, M., Denver, J., Cawley, G., Lee, S. Water permeability across symmetric and asymmetric droplet interface bilayers: Interaction of cholesterol sulfate with DPhPC. Langmuir. 31 (44), 12187-12196 (2015).
- Mruetusatorn, P., et al. Control of membrane permeability in air-stable droplet interface bilayers. Langmuir. 31 (14), 4224-4231 (2015).
- Wauer, T., et al. Construction and manipulation of functional three-dimensional droplet networks. ACS Nano. 8 (1), 771-779 (2013).
- Bayley, H. Building blocks for cells and tissues: Beyond a game. Emerging Topics in Life Sciences. 3 (5), 433-434 (2019).
- Booth, M., Restrepo Schild, V., Downs, F., Bayley, J. Droplet network, from lipid bilayer to synthetic tissues. Encyclopedia of Biophysics. , (2019).
- Booth, M. J., Cazimoglu, I., Bayley, H. Controlled deprotection and release of a small molecule from a compartmented synthetic tissue module. Communications Chemistry. 2 (1), 142 (2019).
- Gobbo, P., et al. Programmed assembly of synthetic protocells into thermoresponsive prototissues. Nature Materials. 17 (12), 1145-1153 (2018).
- Taylor, G. J., Sarles, S. A. Heating-enabled formation of droplet interface bilayers using escherichia coli total lipid extract. Langmuir. 31 (1), 325-337 (2015).
- Taylor, G. J., et al. Capacitive detection of low-enthalpy, higher-order phase transitions in synthetic and natural composition lipid membranes. Langmuir. 33 (38), 10016-10026 (2017).
- Lee, S., Kim, D. H., Needham, D. Equilibrium and dynamic interfacial tension measurements at microscopic interfaces using a micropipet technique. 2. Dynamics of phospholipid monolayer formation and equilibrium tensions at the water-air interface. Langmuir. 17 (18), 5544-5550 (2001).
- Najem, J. S., et al. Assembly and characterization of biomolecular memristors consisting of ion channel-doped lipid membranes. Journal of Visualized Experiments. (145), e58998 (2019).
- Wang, Y. G., Shao, H. H. Optimal tuning for PI controller. Automatica. 36 (1), 147-152 (2000).
- Needham, D., Haydon, D. A. Tensions and free energies of formation of "solventless" lipid bilayers. Measurement of high contact angles. Biophysical Journal. 41 (3), 251-257 (1983).
- Sarles, S. A., Leo, D. J. Physical Encapsulation of Interface Bilayers for durable portable biolayer network. Lab on a Chip. 10 (6), 710-717 (2010).
- Muller, R. U., Peskin, C. S. The kinetics of monazomycin-induced voltage-dependent conductance. II. Theory and a demonstration of a form of memory. The Journal of General Physiology. 78 (2), 201-229 (1981).
- Nenninger, A., et al. Independent mobility of proteins and lipids in the plasma membrane of Escherichia coli. Molecular Microbiology. 92 (5), 1142-1153 (2014).
- Venkatesan, G. A., et al. Adsorption kinetics dictate monolayer self-assembly for both lipid-in and lipid-out approaches to droplet interface bilayer formation. Langmuir. 31 (47), 12883-12893 (2015).
- Najem, J. S., et al. Memristive ion channel-doped biomembranes as synaptic mimics. ACS Nano. 12 (5), 4702-4711 (2018).
- Tamaddoni, N., Taylor, G., Hepburn, T., Michael Kilbey, S., Sarles, S. A. Reversible, voltage-activated formation of biomimetic membranes between triblock copolymer-coated aqueous droplets in good solvents. Soft Matter. 12, 5096-5109 (2016).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved