A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Positron Emission Tomography-based Dose Painting Radiation Therapy in a Glioblastoma Rat Model using the Small Animal Radiation Research Platform
In This Article
Summary
Here we present a protocol to perform preclinical positron emission tomography-based radiotherapy in a rat glioblastoma model using algorithms developed in-house to optimize the accuracy and efficiency.
Abstract
A rat glioblastoma model to mimic chemo-radiation treatment of human glioblastoma in the clinic was previously established. Similar to the clinical treatment, computed tomography (CT) and magnetic resonance imaging (MRI) were combined during the treatment-planning process. Positron emission tomography (PET) imaging was subsequently added to implement sub-volume boosting using a micro-irradiation system. However, combining three imaging modalities (CT, MRI, and PET) using a micro-irradiation system proved to be labor-intensive because multimodal imaging, treatment planning, and dose delivery have to be completed sequentially in the preclinical setting. This also results in a workflow that is more prone to human error. Therefore, a user-friendly algorithm to further optimize preclinical multimodal imaging-based radiation treatment planning was implemented. This software tool was used to evaluate the accuracy and efficiency of dose painting radiation therapy with micro-irradiation by using an in silico study design. The new methodology for dose painting radiation therapy is superior to the previously described method in terms of accuracy, time efficiency, and intra- and inter-user variability. It is also an important step towards the implementation of inverse treatment planning on micro-irradiators, where forward planning is still commonly used, in contrast to clinical systems.
Introduction
Glioblastoma (GB) is a malignant and very aggressive primary brain tumor. GB is a solid heterogeneous tumor typically characterized by infiltrative boundaries, nuclear atypia, and necrosis1. The presence of the blood-brain-barrier and the brain's status as an immune-privileged site makes the discovery of novel targets for chemo- and immunotherapy a daunting task2,3,4. It is noteworthy that the treatment of GB patients has barely changed since the introduction, in 2005, of the Stupp protocol that combines external beam radiation therapy (RT) with concomitant temozolomide, usually followed by adjuvant temozolomide5. Typically, the Stupp protocol is preceded by maximal surgical resection. Therefore, alternative treatment approaches are of pivotal importance.
Current radiation therapy for glioblastoma patients delivers a uniform radiation dose to the defined tumor volume. In radiation oncology, there is an important dose-response correlation for glioblastoma with increasing dose, which seems to cap around 60 Gy, due to increased toxicity to the normal brain6,7. However, tumors can be very (radiobiologically) heterogeneous, with gradients of oxygen level and/or large differences in cellular density. Metabolic imaging techniques, such as PET, can visualize these biological features and can be utilized to customize the dose prescription. This approach is known as dose painting RT. This term was introduced by Ling et al. in 2000. The authors defined dose painting RT as producing "exquisitely conformal dose distributions within the constraints of radiation propagation and scatter"8.
There are two types of dose painting RT, dose painting by contours (DPBC), by which a dose is prescribed to a set of nested sub-volumes, and dose painting by numbers (DPBN), whereby a dose is prescribed at the voxel level. The dose distribution for DPBN RT can be extracted from functional images. The dose in each voxel is determined by the intensity I of the corresponding voxel in the image, with a lower and upper limit, to make sure that, on the one hand, a sufficient dose is delivered to every part of the tumor. On the other hand, doses do not exceed an upper limit to protect organs at risk and avoid toxicity. The most straightforward method is a linear interpolation (see Eq. 1) between minimum dose Dmin and maximum dose Dmax, proportionally varying between minimum intensity Imax and maximum intensity within the target volume9,10
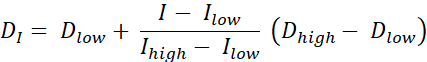 Eq. 1
Eq. 1
Because there is some skepticism about the quality assurance of DPBN RT, the deposition of the dose should be verified through preclinical and clinical research10. However, only limited data can be acquired from clinical trials, and it has been hypothesized that more insights can be obtained by downscaling to laboratory animals11,12. Hence, preclinical studies utilizing precision image-guided radiation research platforms that allow coupling with some very specific techniques, such as autoradiography, are suited for examining open issues and paving the way towards personalized medicine and novel treatment strategies, such as dose painting RT13,14. However, the interpretation of preclinical data must be performed with caution, and drawbacks of these preclinical setups have to be considered14.
Micro-irradiation systems, such as the Small Animal Radiation Research Platform (SARRP), are equipped with similar technologies as their clinical counterpart. They include on-board cone-beam CT (CBCT) imaging, a preclinical treatment-planning system (PCTPS), and provide sub-millimeter precision. Clinical dose calculations are performed by inverse treatment planning, whereby one initiates from the desired dose distribution to determine the beams via an iterative algorithm. Preclinical irradiators often use forward planning. In forward planning, the required amount and angle of the beams are selected, and the PCTPS then calculates the dose distribution. The optimization of the plans is performed by manual iteration, which is labor-intensive15.
After 2009, novel developments have made the implementation of inverse planning on these research platforms possible16,17,18. To increase the similarity with the clinical method, a motorized variable rectangular collimator (MVC) was developed as a preclinical counterpart of the multi-leaf collimator. A two-dimensional dose painting method utilizing a variable collimator was published by Cho et al.19. This research group implemented a three-dimensional (3D) inverse treatment-planning protocol on a micro-irradiator and determined minimum and maximum doses for the target volume and a maximum dose for the organs at risk. These techniques have mainly been evaluated in silico, and their preclinical applications need to be explored.
This paper presents an in silico study to compare two methodologies for [18F]-fluoro-ethyl-L-tyrosine ([18F]FET) PET-based dose painting in a GB rat model20,21,22 using a small animal radiation research platform. These two methodologies are (1) sub-volume boosting using predefined beam sizes and (2) dose painting using a motorized variable collimator where jaw dimensions are modified based on the PET tracer uptake in the tumor volume. [18F]FET is a PET tracer often used in neuro-oncology because of its ability to detect brain tumors23. [18F]FET is an artificial amino acid that is internalized into tumoral cells but not incorporated into cell proteins. [18F]FET uptake corresponds with cell proliferation rate, tumor cell density, and angiogenesis24. As this is the most commonly used oncologic brain PET tracer in these authors' institute, this radiotracer was chosen to evaluate the new workflow.
Protocol
The study was approved by the local ethics committee for animal experiments (ECD 18/21). Anesthesia monitoring is performed by acquiring the respiratory rate of the animals using a sensor.
1. F98 GB rat cell model
- Culture the F98 GB cells in a monolayer using Dulbecco's Modified Eagle Medium, supplemented with 10% calf serum, 1% penicillin, 1% streptomycin, and 1% L-glutamine, and place them in a CO2 incubator (5% CO2 and 37 °C).
- Inoculate the glioma cells into the brain of female Fischer F344 rats (body weight 170 g).
NOTE: Use sterile instruments and wear sterile gloves at all times.- Anesthetize the rats through the inhalation of isoflurane (5% induction, 2% maintenance) mixed with oxygen (0.3 mL/min) through a nose cone. Confirm the anesthetization by the absence of withdrawal reflex of the limb, and immobilize the rats in a stereotactic device using fixation points for the nose and ears. Apply a carbomer eye gel to prevent dry eyes under anesthesia. Maintain the body temperature by a thermoregulated heating pad and rectal probe at 37 °C.
- Shave the rat from the eye level to the back of the skull, and disinfect the skin with isobetadine. Inject xylocaine (with adrenaline 1:200000, 0.1 mL) subcutaneously for local anesthesia.
- Expose the skull through a midline scalp incision and make a small hole with a drill tool 3 mm posterior and 3 mm lateral to the bregma in the right hemisphere.
- Insert a stereotactically guided insulin needle (29 G) and inject 5 µL of cell suspension (20,000 F98 GB cells) 3 mm deep using a microsyringe pump controller. Leave the needle in place for 5 min, giving the cell suspension time to diffuse into the tissue.
- Withdraw the syringe slowly and close the hole in the skull with bone wax. Suture the skin (polyamide 6, thickness 4-0) and inject meloxicam subcutaneously (1 mg/kg, 2 mg/mL). Apply xylocaine gel.
- Stabilize the body temperature of the animal post-surgery using a red lamp. Monitor the awakening of the rat until it has regained sufficient consciousness. Do not return the animal to the company of other animals until fully recovered. House all animals under environmentally controlled conditions (12 h light/dark cycle, 20-24 °C, and 40-70% relative humidity) with food and water ad libitum.
- Be sure to monitor the animals daily and maintain a daily health status log by checking their body weight, food, water intake, and their activity and behavior. Use a lethal dose of pentobarbital sodium to euthanize the animals (160 mg/kg) if a decline of 20% body weight is observed or when the normal behavior severely deteriorates (e.g., lack of grooming).
2. Confirmation of tumor growth
- Evaluate tumor growth 8 days post-inoculation. Anesthetize the rats through the inhalation of isoflurane (5% induction, 2% maintenance) mixed with oxygen (0.3 mL/min) through a nose cone. Confirm the anesthetization by the absence of withdrawal reflex of the limb.
- Inject a gadolinium-containing contrast agent (0.4 mL/kg) through an intravenously placed tubing in the lateral tail vein. Cover the animal with a warm water circulating heating blanket and place them in the MRI bed. Apply a carbomer eye gel to prevent dry eyes under anesthesia. Place the MRI bed in the holder with a Tx/Rx Rat brain volume coil.
- Perform a localizer scan followed by a T2-weighted spin-echo scan to assess tumor growth. Use these T2-MRI sequence settings: repetition time (TR)/echo time (TE) 3661/37.1 ms, 109 µm isotropic in-plane resolution, slice thickness 600 µm, 4 averages, 30 slices, total acquisition time (TA) 9 min 45 s.
- If a tumor is confirmed on the T2-weighted acquisition, perform a T1-weighted contrast-enhanced spin echo scan. Use these T1-MRI sequence settings: TR/TE 1539/9.7 ms, 0.117 mm isotropic in-plane resolution, slice thickness 600 µm, 3 averages, 30 slices, TA 4 min 15 s.
- After the MRI, continuously supervise the animal until it regains full consciousness.
- When the tumor reaches a diameter of 7 to 8 mm, usually observed 12 days after inoculation, select the animal for therapy.
3. Multimodality imaging of target volume selection
NOTE: PET/MRI-guided irradiation requires the sequential acquisition of a multimodal dataset. After intravenous administration of the radiotracer, PET imaging is started, followed by contrast-enhanced T1-weighted MRI and finally a treatment-planning CT.
- Anesthetize the animal with isoflurane (5% induction, 2% maintenance) mixed with oxygen (0.3 L/min) using a nose cone. Confirm anesthetization when the rats do not exhibit any withdrawal reflex of the limb. Apply carbomer eye gel to prevent dry eyes under anesthesia.
- Insert the tubing intravenously in the lateral tail vein, enabling the injection of 10-12 MBq of PET radioactive tracer dissolved in 200 µL of saline. Inject [18F]-FET, 1 h before PET acquisition. Let the animal regain consciousness while the tracer is distributed through the body.
- Anesthetize the animal again, as described in step 3.1. Place the animal on a multimodality bed (here, made in-house) and secure it using hook-and-loop fasteners, maintaining a fixed position during the imaging and micro-irradiation. Fix a capillary filled with the MRI/PET agent (see the Table of Materials) for easier co-registration. Wrap the animal in bubble wrap to preserve its body temperature during the multimodality imaging and therapy.
- Perform a PET scan 1 h after the injection of the PET tracer. Reconstruct the PET scan into a 3D volume (192 x 192 x 384 matrix) with 0.4 mm voxel size by applying a Maximum-Likelihood Expectation-Maximization (MLEM)-algorithm using 30 iterations.
NOTE: A dedicated PET scanner for laboratory animal imaging was used with an axial field of view of 130 mm and a bore diameter of 72 mm. This system provides sub-mm (0.85 mm) spatial resolution. - Inject an MRI contrast agent (0.4 mL/kg) intravenously into the tail vein. Place the rat, still fixed on the multimodality bed, in the animal holder of the MRI scanner (Table of Materials). Perform a localizer scan followed by a contrast-enhanced T1-weighted spin-echo sequence, analogous to step 2.4.
- Place the animal, still fixed on the multimodality bed, on a plastic holder secured onto the four-axis robotic positioning table on the micro-irradiator. Perform a high-resolution treatment-planning cone-beam CT using a tube voltage of 70 kV, a tube current of 0.4 mA, a 1 mm aluminum filter, and a 20 x 20 cm (1024 x 1024 pixel) amorphous Si flat panel detector. Acquire a total of 360 projections over 360°. Reconstruct the CT images with an isotropic voxel size of 0.275 mm (411 x 411 x 251 matrix).
4. Image co-registration
NOTE: The co-registration is performed with a semi-automatic MATLAB code developed in-house. The code can be found on Github at https://github.com/sdonche/DosePainting. The different steps are described below.
- Place the three image modalities ([18F]FET PET, contrast-enhanced T1-weighted MRI, and cone-beam CT) into one folder. Convert DICOM images to the NIfTI format using the dcm2niix function from the mricron image viewer24.
- Import the converted images into MATLAB and filter the PET image with a Gaussian filter using a Full-Width Half-Max (FWHM) of 1 mm.
- Reorient the images so that the cartesian axes from all imaging modalities correspond with each other.
NOTE: For this setup, the CT image was flipped around the Y-axis; the MRI was flipped around the X-axis, and the PET was flipped around the Y-axis. - Crop the PET image to simplify automatic co-registration.
NOTE: For this setup, 40 pixels were set to zero from both sides of the X-axis (left and right of the animal); on the dorsal and ventral side of the animal (Y-axis), 60 and 40 pixels were set to zero, respectively; along the longitudinal axis (or Z-axis), 170 and 30 pixels are set to zero for inferior and superior side, respectively. - Move the image centers close to each other to simplify automatic co-registration.
- Perform the actual rigid-body co-registration using Statistical Parametric Mapping (SPM) in MATLAB26. Use the following registration parameters (others on default): objective function: mutual information; separation: [4 1 0.2]; tolerances: [0.02 0.02 0.02 0.001 0.001 0.001 0.01 0.01 0.01 0.001 0.001 0.001]; histogram smoothing: [1 1]; interpolation: trilinear.
5. Radiation treatment planning
NOTE: A MATLAB app and multiple MATLAB scripts were written for the radiation treatment planning. The code can be found on Github at https://github.com/sdonche/DosePainting. The different steps are explained below.
- Method 1
- Load the three different imaging modalities into the MATLAB app. Place a generous bounding box around the contrast enhancement on the T1-weighted MRI scan (Figure 1). Determine the contrast-enhanced volume using a threshold (Figure 2). If multiple regions have been selected, select only the largest volume, the center of which is considered as the first isocenter to deliver a prescribed dose for RT (Figure 3).
- Expand the previously determined MRI contrast enhancement by 10 pixels in each direction. If multiple regions are detected, retain only the largest PET volume, the center of which is considered the second isocenter to deliver a prescribed dose for RT.
NOTE: In this PET volume, the PET boost volume is defined by the pixels with a higher signal intensity than 0.90 × maximal signal intensity (in the bounding box) in this volume. - Use the following irradiation settings for the calculated isocenters (Figure 4 and Table 1).
- For the first isocenter (MRI), give a prescribed dose of 2000 cGy using 3 non-coplanar arcs at couch positions 0°, -45°, and -90° with a gantry rotation of 120°, 120°, and 60°, respectively. Use a fixed collimator size of 10 x 10 mm, but use an appropriate collimator (e.g., a 5 x 5 mm collimator) when smaller tumor sizes need to be irradiated. Be careful in considering the animal's welfare when the tumor volumes are larger than 10 mm.
- For the second isocenter (PET), give a prescribed dose of 800 cGy using 3 non-coplanar arcs at couch positions 0°, -45°, and -90° with a gantry rotation of 120°, 120°, and 60°, respectively. Use a fixed collimator size of 3 x 3 mm.
- Calculate the dose distribution within the animal and the beam delivery parameters.
- Method 2
- Load the three different imaging modalities into the MATLAB app. Place a generous bounding box around the contrast-enhancement on the [18F]FET PET image, analogous to step 5.1.1.
- Determine the volumes defined by the pixels with a signal intensity higher than A × maximal signal intensity (in the aforementioned bounding box), with A equal to 0.50, 0.60, 0.70, 0.80, and 0.90. Name these volumes V50, V60, V70, V80, and V90, respectively.
- Determine the isocenters and the jaw dimensions for each beam required to guide the motorized variable collimator using the MATLAB script (see Figure 5).
- Use the following settings for the calculated isocenters and jaw dimensions:
- For V50, give a prescribed dose of 2000 cGy distributed over 16 beams (each 125 cGy; couch and gantry positions in Table 2). Use the calculated jaw dimensions for the MVC.
NOTE: Here, an additional margin of 1 mm has been included to account for microscopic tumor infiltration. - For V60-V90, give a prescribed dose of 800 cGy distributed over 40 beams (each 20 cGy; couch and gantry positions in Table 2). Use the calculated jaw dimensions for the MVC.
- For V50, give a prescribed dose of 2000 cGy distributed over 16 beams (each 125 cGy; couch and gantry positions in Table 2). Use the calculated jaw dimensions for the MVC.
- Calculate the dose distribution within the animal and the beam delivery parameters.
6. Plan evaluation
NOTE: To compare the two methods, calculate the dose-volume histograms (DVH) and Q-volume histogram (QVH) in the V50 PET volume. Here, a MATLAB script, developed in-house, was used. The code can be found on Github at https://github.com/sdonche/DosePainting.
- Dose-volume histogram
- Generate DVH from the dose distribution that was obtained from the SARRP.
- Determine the maximum, mean, and minimum doses from the DVH by calculating the D10, D50, and D90, where Dx stands for the dose received by x% of the volume.
- Q-volume histogram
- Calculate an ideal dose for every pixel using Eq. 1, which is a linear interpolation between the minimum and maximum doses, proportionally varying between the minimum PET intensity and maximum PET intensity within the target volume to give an ideal dose map.
- Calculate the Q-value Qp for every pixel using the following equation (Eq. 2):
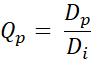 Eq. 2
Eq. 2
With Dp being the dose obtained by planning and Di, the dose objective for planning. - Generate QVH from the obtained Q-values.
- Calculate the quality factor (Q-factor, QF) to evaluate the difference between the planned and intended doses using Eq. 3:
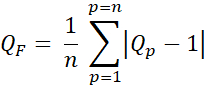 Eq. 3
Eq. 3
Results
The feasibility of PET- and MRI-guided irradiation in a glioblastoma rat model using the SARRP to mimic the human treatment strategy has been previously described20,21,22. While the animal was fixed on a multimodality bed made in-house, it was possible to create an acceptable radiation treatment plan combining three imaging modalities: PET, MRI, and CT. In these methods, an external software package (see the Table of Mat...
Discussion
A rat GB model to mimic the chemo-radiation treatment in the clinic for glioblastoma patients was previously described20. Similar to the clinical method, CT and MRI were combined during the treatment-planning process to obtain more precise irradiation. A multimodality bed to minimize (head) movement was used when the animal was moved from one imaging system to another. Subsequently, PET imaging was added to the treatment-planning process, and PET-based sub-volume boosting could be successfully imp...
Disclosures
The authors have no conflicts of interest to disclose.
Acknowledgements
The authors would like to thank Lux Luka Foundation for supporting this work.
Materials
| Name | Company | Catalog Number | Comments |
| Cell culture | |||
| F98 Glioblastoma Cell Line | ATCC | CRL-2397 | https://www.lgcstandards-atcc.org/products/all/CRL-2397 |
| Dulbeco's Modified Eagle Medium | Thermo Fisher Scientific | 22320-030 | |
| Cell culture flasks | Thermo Fisher Scientific | 178883 | 75 cm² |
| FBS | Thermo Fisher Scientific | 10270106 | |
| L-Glutamine | Thermo Fisher Scientific | 25030-032 | 200 mM |
| Penicilline-Streptomycin | Thermo Fisher Scientific | 15140-148 | 10,000 U/mL |
| Phosphate-Buffered Saline (PBS) | Thermo Fisher Scientific | 14040-224 | |
| Trypsin-EDTA | Thermo Fisher Scientific | 25300-062 | 0.05% |
| GB Rat Model | |||
| Ball-shaped burr | Foredom | A-228 | 1.8 mm |
| Bone Wax | Aesculap | 1029754 | https://www.aesculapusa.com/en/healthcare-professionals/or-solutions/or-solutions-cranial-closure/hemostatic-bone-wax.html |
| Ethilon | Ethicon | 662G/662H | FS-2, 4-0, 3/8, 19 mm |
| Fischer F344/Ico crl Rats | Charles River | - | |
| Insulin Syringe Microfine | Beckton-Dickinson | 320924 | 1 mL, 29 G |
| IR Lamp | Philips | HP3616/01 | |
| Meloxicam (Metacam) | Boehringer Ingelheim | - | 2 mg/mL |
| Micromotor rotary tool | Foredom | K.1090-22 | |
| Micropump system | Stoelting Co. | 53312 | Stoelting Stereotaxic Injector |
| Stereotactic frame | Stoelting Co. | 51600 | |
| Xylocaine (1%, with adrenaline 1:200,000) | Aspen | - | 1%, with adrenaline 1:200,000 |
| Xylocaine gel (2%) | Aspen | - | 2% |
| Animal Irradiation | |||
| Micro-irradiator | X-Strahl | SARRP | Version 4.2.0 |
| Software | X-Strahl | Muriplan | Preclinical treatment planning system (PCTPC), version 2.2.2 |
| Small Animal PET | |||
| [18F]FET | Inhouse made | - | PET tracer; along with Prohance: MRI/PET agent |
| Micro-PET | Molecubes | Beta-Cube | https://www.molecubes.com/b-cube/ |
| Small Animal MRI | |||
| Micro-MRI | Bruker Biospin | Pharmascan 70/16 | https://www.bruker.com/products/mr/preclinical-mri/pharmascan.html |
| 30 G Needle for IV injection | Beckton-Dickinson | 305128 | |
| PE 10 Tubing | Instech Laboratories Inc | BTPE-10 | BTPE-10, polyethylene tubing 0.011 x 0.024 in (0.28 x 60 mm), non sterile, 30 m (98 ft) spool, Instech laboratories, Inc Plymouth meeting PA USA- (800) 443-4227- http://www.instechlabs.com |
| Prohance contrast agent | Bracco Imaging | - | 279.3 mg/mL, gadolinium-contrast agent (along with [18F]FET: MRI/PET agent) |
| Tx/Rx Rat Brain - Mouse Whole Body Volumecoil | Bruker Biospin | - | 40 mm diameter |
| Water-based Heating Unit | Bruker Biospin | MT0125 | |
| Consumables | |||
| Isoflurane | Zoetis | B506 | Anesthesia |
| Insulin Syringe Microfine | Beckton-Dickinson | 320924 | 1 mL, 29 G |
| Image Analysis | |||
| MATLAB | Mathworks | - | Version R2019b |
| PMOD | PMOD technologies LLC | Preclinical and molecular imaging software |
References
- Louis, D. N., et al. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathologica. 131 (6), 803-820 (2016).
- Wadajkar, A. S., et al. Tumor-targeted nanotherapeutics: Overcoming treatment barriers for glioblastoma. Wiley Interdisciplinary Reviews. Nanomedicine & Nanobiotechnology. 9 (4), (2016).
- Lim, M., Xia, Y., Bettegowda, C., Weller, M. Current state of immunotherapy for glioblastoma. Nature Reviews. Clinical Oncology. 15 (7), 422 (2018).
- McGranahan, T., Li, G., Nagpal, S. History and current state of immunotherapy in glioma and brain metastasis. Therapeutic Advances in Medical Oncology. 9 (5), 347-368 (2017).
- Stupp, R., et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. The New England Journal of Medicine. 352 (10), 987-996 (2005).
- Von Neubeck, C., Seidlitz, A., Kitzler, H. H., Beuthien-Baumann, B., Krause, M. Glioblastoma multiforme: Emerging treatments and stratification markers beyond new drugs. The British Journal of Radiology. 88 (1053), 20150354 (2015).
- Mann, J., Ramakrishna, R., Magge, R., Wernicke, A. G. Advances in radiotherapy for glioblastoma. Frontiers in Neurology. 8, 748 (2018).
- Ling, C. C., et al. Towards multidimensional radiotherapy (MD-CRT): Biological imaging and biological conformality. International Journal of Radiation Oncolology, Biology, Physics. 47 (3), 551-560 (2000).
- Bentzen, S. M., Gregoire, V. Molecular imaging-based dose painting: a novel paradigm for radiation therapy prescription. Seminars in Radiation Oncology. 21 (2), 101-110 (2011).
- Bentzen, S. M. Theragnostic imaging for radiation oncology: Dose-painting by numbers. The Lancet. Oncology. 6 (2), 112-117 (2005).
- Wong, J., et al. High-resolution, small animal radiation research platform with X-ray tomographic guidance capabilities. International Journal of Radiation Oncolology, Biology, Physics. 71 (5), 1591-1599 (2008).
- Van Hoof, S. J., Granton, P. V., Verhaegen, F. Development and validation of a treatment planning system for small animal radiotherapy: SmART-Plan. Radiotherapy and Oncology. 109 (3), 361-366 (2013).
- Verhaegen, F., Granton, P., Tryggestad, E. Small animal radiotherapy research platforms. Physics in Medicine & Biology. 56 (12), 55-83 (2011).
- Butterworth, K. T., Prise, K. M., Verhaegen, F. Small animal image-guided radiotherapy: Status, considerations and potential for translational impact. The British Journal of Radiology. 88 (1045), 20140634 (2015).
- Nasr, A., Habash, A. Dosimetric analytic comparison of inverse and forward planned IMRT techniques in the treatment of head and neck cancer. Journal of the Egyptian National Cancer Institute. 26 (3), 119-125 (2014).
- Matinfar, M., Iyer, S., Ford, E., Wong, J., Kazanzides, P. Image guided complex dose delivery for small animal radiotherapy. IEEE International Symposium on Biomedical Imaging: From Nano to Macro. , 1243-1246 (2009).
- Matinfar, M., Iordachita, I., Wong, J., Kazanzides, P. Robotic delivery of complex radiation volumes for small animal research. IEEE International Conference on Robotics and Automation. , 2056-2061 (2010).
- Balvert, M., et al. A framework for inverse planning of beam-on times for 3D small animal radiotherapy using interactive multi-objective optimisation. Physics in Medicine & Biology. 60 (14), 5681-5698 (2015).
- Cho, N. B., Wong, J., Kazanzides, P. Dose Painting with a Variable Collimator for the Small Animal Radiation Research Platform (SARRP). The Midas Journal. , 1-8 (2014).
- Bolcaen, J., et al. MRI-guided 3D conformal arc micro-irradiation of a F98 glioblastoma rat model using the Small Animal Radiation Research Platform (SARRP). Journal of Neuro-oncology. 120 (2), 257-266 (2014).
- Bolcaen, J., Descamps, B., Boterberg, T., Vanhove, C., Goethals, I. PET and MRI guided irradiation of a glioblastoma rat model using a micro-irradiator. Journal of Visualized Experiments: JoVE. (130), e56601 (2017).
- Verhoeven, J., et al. Technical feasibility of [18F]FET and [18F]FAZA PET guided radiotherapy in a F98 glioblastoma rat model. Radiation Oncology. 14 (1), 89 (2019).
- Hutterer, M., et al. FET PET: a valuable diagnostic tool in neuro-oncology, but not all that glitters is glioma. Neuro-oncology. 15 (3), 341-351 (2013).
- Stockhammer, F., Plotkin, M., Amthauer, H., Landeghem, F. K. H., Woiciechowsky, C. Correlation of F-18-fluoro-ethyl-tyrosin uptake with vascular and cell density in non-contrast-enhancing gliomas. Journal of Neuro-oncology. 88 (2), 205-210 (2008).
- . Mricron dicom to nifti converter. neuroimaging informatics tools and resources clearinghouse (nitrc) Available from: https://www.nitrc.org/projects/mricron (2015)
- . SPM12 Manual Available from: https://www.fil.ion.ucl.ac.uk/spm/doc/spm12_manual.pdf (2014)
- España, S., Marcinkowski, R., Keereman, V., Vandenberghe, S., Van Holen, R. DigiPET: Sub-millimeter spatial resolution small-animal PET imaging using thin monolithic scintillators. Physics in Medicine & Biology. 59 (13), 3405-3420 (2014).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved