A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Determination of the Absorption, Translocation, and Distribution of Imidacloprid in Wheat
In This Article
Summary
Presented here is a protocol for the determination of the absorption, translocation, and distribution of imidacloprid in wheat under hydroponic conditions using liquid chromatography-tandem mass spectrometry (LC-MS-MS). The results showed that imidacloprid can be absorbed by wheat, and imidacloprid was detected in both the wheat roots and leaves.
Abstract
Neonicotinoids, a class of insecticides, are widely used because of their novel modes of action, high insecticidal activity, and strong root uptake. Imidacloprid, the most widely used insecticide worldwide, is a representative first-generation neonicotinoid and is used in pest control for crops, vegetables, and fruit trees. With such a broad application of imidacloprid, its residue in crops has attracted increasing scrutiny. In the present study, 15 wheat seedlings were placed in a culture medium containing 0.5 mg/L or 5 mg/L imidacloprid for hydroculture. The content of imidacloprid in the wheat roots and leaves was determined after 1 day, 2 days, and 3 days of hydroculture to explore the migration and distribution of imidacloprid in wheat. The results showed that imidacloprid was detected both in the roots and leaves of the wheat plant, and the content of imidacloprid in the roots was higher than that in the leaves. Furthermore, the imidacloprid concentration in the wheat increased with increasing exposure time. After 3 days of exposure, the roots and leaves of the wheat in the 0.5 mg/L treatment group contained 4.55 mg/kg ± 1.45 mg/kg and 1.30 mg/kg ± 0.08 mg/kg imidacloprid, respectively, while the roots and leaves of the 5 mg/L treatment group contained 42.5 mg/kg ± 0.62 mg/kg and 8.71 mg/kg ± 0.14 mg/kg imidacloprid, respectively. The results from the present study allow for a better understanding of pesticide residues in crops and provide a data reference for the environmental risk assessment of pesticides.
Introduction
In present day agronomy, the use of pesticides is essential to increase crop yield. Neonicotinoid insecticides alter the membrane potential balance by controlling nicotinic acetylcholine receptors in the insect nervous system, thereby inhibiting the normal conduction of the insect central nervous system, leading to the paralysis and death of the insects1. Compared with traditional insecticides, neonicotinoids have advantages such as novel modes of action, high insecticidal activity, and strong root absorption, making them highly successful in the pesticide market2,3. The sales volume of neonicotinoids was reported to account for 27% of the world pesticide market in 2014. The average annual growth rate of neonicotinoids was 11.4% from 2005 to 2010, of which about 7% was registered in China4,5,6. From the end of 2016 to the first half of 2017, the sales of pesticides in China began to rebound after falling, and the prices of pesticides continued to rise, among which neonicotinoid insecticides showed a significant price increase7. So far, three generations of neonicotinoid insecticides have been developed, each containing pyridine chloride, thiazolyl, and tetrahydrofuran groups of nicotine, respectively8.
Imidacloprid represents the first generation of neonicotinoid insecticides, whose molecular formula is C9H10ClN5O2, and is a colorless crystal. Imidacloprid is used mainly to control pests, such as aphids, planthoppers, mealworms, and thrips9 and can be applied to crops such as rice, wheat, corn, cotton, and vegetables such as potatoes, as well as fruit trees. Due to the long-term, substantial, and continual application of pesticides, both beneficial insects and the natural enemies of pests have been rapidly reduced, and some agricultural pests have become resistant to pesticides, resulting in a vicious circle of applying continual and increasing amounts of pesticides10. In addition, the extensive application of pesticides has led to the deterioration of soil quality, persistent pesticide residues in agricultural products, and other ecological problems, which not only cause significant damage to the agricultural ecological environment11 but also pose a serious threat to human health12. Pesticide spraying severely impacts the growth and quality of soil microbes and soil animals13. The unreasonable or excessive use of pesticides has caused significant security risks to the soil and water environment, animals and plants, and even human life14. In recent years, the problem of excessive pesticide residues in crops has become more severe with the extensive application of pesticides. When imidacloprid was used to increase vegetable yield, the absorption rate of imidacloprid in the vegetables increased with the increase in the amount and residue of imidacloprid15. As a major food crop, both the production and safety of wheat are critical. Therefore, the residue and distribution policies of pesticides used for wheat need to be clarified.
In recent years, many methods have been developed to extract imidacloprid residues from water, soil, and plants. The QuEChERS method (quick, easy, cheap, effective, rugged, and safe) is a new method that combines solid-phase microextraction technology and dispersed solid-phase extraction technology and involves the use of acetonitrile as the extraction solvent and the removal of mixed impurities and water in the sample using NaCl and anhydrous MgSO4, respectively16. The QuEChERS method requires minimal glassware and has simple experimental steps, making it one of the most popular pesticide extraction methods17. For the detection of imidacloprid, a detection limit as low as 1 × 10−9 g18 has been achieved with liquid chromatography (LC), and 1 × 10−11 g19 has been achieved with gas chromatography (GC). Due to their high resolution and sensitivity, LC-MS and GC-MS have shown even lower imidacloprid detection limits of 1 × 10-13 to 1 × 10-14 g20,21; these techniques are, therefore, well suited for the analysis of trace imidacloprid residues.
In the present study, imidacloprid was chosen as the target pollutant, and wheat was selected as the test crop to study the distribution of imidacloprid residues in wheat. This protocol details a method for the comprehensive analysis of the enrichment and transfer of the pesticide imidacloprid in wheat by exploring the absorption and storage of imidacloprid in different parts of wheat plants grown under hydroponic conditions. The present study aims to provide a theoretical basis for the risk assessment of pesticide residues in wheat, guide the rational application of pesticides in agricultural production activities to reduce pesticide residues, and improve the safety of crop production.
Protocol
1. Germination of wheat seeds
- Select 1,000 wheat seeds (Jimai 20) with complete granules, intact embryos, and uniform size (length: 6 mm ± 0.5 mm).
- Transfer 333.3 mL of 30% H2O2 solution to a 1 L volumetric flask and dilute with deionized water to prepare 1 L of 10% H2O2 solution. Immerse the wheat seeds in 10% H2O2 solution for 15 min to disinfect the seed surface (Figure 1).
- Rinse the wheat seeds 5x with running sterile water for 10 s each time.
- Spread the wheat seeds evenly with the embryos pointing up in a glass Petri dish containing moist sterile filter paper (Figure 2). Place the Petri dish in an artificial climate incubator at 30 °C and 80% relative humidity. Culture the wheat seeds in the dark for 3 days until they germinate and take root.

Figure 1: Disinfection of the wheat seeds. The wheat seeds were soaked in 10% H2O2 solution (in a beaker) for 15 min to disinfect the seed surface. Please click here to view a larger version of this figure.
Figure 2: Germinating the wheat seeds. The wheat seeds were spread evenly in a glass Petri dish containing moist sterile filter paper. The Petri dish was placed in an artificial climate incubator to germinate the wheat seeds. Please click here to view a larger version of this figure.
2. Cultivation of wheat seedlings
- Dissolve 551 mg of Hoagland's basal salt mixture in 1 L of deionized water to prepare 1/2 Hoagland nutrient solution (containing 0.75 mmol/L K2SO4, 0.1 mmol/L KCl, 0.6 mmol/L MgSO4, 4.0 × 10−2 mmol/L FeEDTA, 1.0 × 10−3 mmol/L H3BO3, 1.0 × 10−3 mmol/L MnSO4, 1.0 × 10−3 mmol/L ZnSO4, 1.0 × 10−4 mmol/L CuSO4, and 5.0 × 10−6 mmol/L Na2 MoO4).
- After the wheat seeds (step 1.4) have germinated, place 15 wheat seedlings in hydroponic equipment (see Table of Materials) containing 100 mL of 1/2 Hoagland nutrient solution for hydroponics (Figure 3). Place the entire hydroponic apparatus in an artificial climate incubator (see Table of Materials) and incubate for 7 days at 25 °C and 80% relative humidity with a 16 h light/8 h dark photoperiod.

Figure 3: Hydroponic cultivation of the wheat seedlings. The wheat seedlings were hydroponically cultivated for 0 days, 3 days, and 7 days in 100 mL of 1/2 Hoagland nutrient solution. Please click here to view a larger version of this figure.
3. Experiment exposing the wheat plants to imidacloprid solution
- After a 7 day hydroponic period, transplant the wheat plants into 1/2 Hoagland nutrient solution containing 0.5 mg/L or 5 mg/L imidacloprid to conduct the imidacloprid exposure experiments. Grow 15 wheat plants in each hydroponic device. Set up 15 hydroponic devices for each imidacloprid concentration group to ensure that adequate samples are taken during sampling.
- Place the entire hydroponic equipment in an artificial climate incubator for 3 days at 25 °C and 80% relative humidity with a 16 h light/8 h dark photoperiod.
- Throughout the exposure period, collect wheat roots (0.2 g per wheat plant) and leaves (0.5 g per wheat plant) daily. Integrate the wheat samples from every fifth hydroponic device as a parallel group and determine the imidacloprid content of the samples.
4. Procedure for extracting imidacloprid from wheat
- Extraction of imidacloprid from wheat roots
- To avoid experimental errors, wash the wheat roots 4x with running sterile water for 10 s each time to remove any imidacloprid adsorbed on the root surface.
- Shred the wheat roots into approximately 1 cm pieces with scissors (Figure 4). Weigh 10.00 g of the shredded wheat roots and place in a 50 mL centrifuge tube.
- Add 10 mL of acetonitrile to the centrifuge tube and vortex the tube on a vortexer for 1 min. Then, add 4 g of anhydrous MgSO4 and 1.5 g of NaCl to the centrifuge tube and vortex the tube immediately for 30 s. Centrifuge the tube for 5 min at 6,000 x g.
- Aspirate the supernatant with a disposable syringe and pass it through a syringe filter (0.22 µm pore size) to obtain the sample.
- Extraction of imidacloprid from wheat leaves (Figure 5)
- Shred the fresh wheat leaves into approximately 1 cm pieces with scissors (Figure 4). Weigh 10.00 g of the shredded wheat leaves and place in a 50 mL centrifuge tube.
- Add 10 mL acetonitrile to the centrifuge tube and vortex the tube on a vortexer for 1 min.
- Add 4 g of anhydrous MgSO4 and 1.5 g of NaCl to the centrifuge tube and vortex the tube immediately for 30 s.
- Centrifuge the tube for 5 min at 6,000 x g.
- After centrifugation, add 2 mL of the supernatant to a 5 mL centrifuge tube containing 50 mg of graphitized carbon black (GCB) and 150 mg of anhydrous MgSO4 (to remove pigment and moisture from the sample), and vortex the centrifuge tube for 30 s (Figure 6). Centrifuge the tube for 5 min at 6,000 x g.
- Aspirate the supernatant with a disposable syringe and pass it through a syringe filter (0.22 µm pore size) to obtain the sample.

Figure 4: Shredded wheat roots and leaves. Fresh wheat roots and leaves were shredded using scissors into approximately 1 cm pieces. Please click here to view a larger version of this figure.

Figure 5: Extraction of imidacloprid in the wheat leaves. Imidacloprid in the samples was extracted using the QuEChERS method (steps 4.2.1-4.2.4 of the protocol). Please click here to view a larger version of this figure.

Figure 6: Purification of imidacloprid in the wheat leaves. The decontaminant was 50 mg GCB + 150 mg MgSO4. Please click here to view a larger version of this figure.
5. Quantification of imidacloprid
- Quantify the imidacloprid in the sample using liquid chromatography-tandem mass spectrometry (LC-MS-MS),based on a standard curve (y = 696.61x + 56.411, R=1) obtained from 0.2-250 µg/L imidacloprid concentrations. (Figure 7). The mass spectrometer was equipped with a C18 column (100 mm x 2.1 mm, 3 µm) and an electrospray ionization source (ESI+). The elution program and ion source parameters are shown in Table 1.
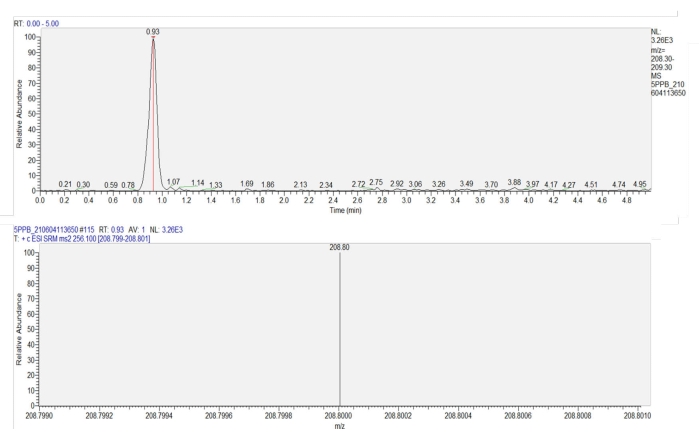
Figure 7: Chromatogram and mass spectrogram of imidacloprid in the wheat leaves. The upper panel shows a chromatogram of imidacloprid (retention time = 0.93 min). The lower panel shows the mass spectrogram of imidacloprid at 0.93 min, showing the response intensity of the production (m/z = 208.8) of imidacloprid. Please click here to view a larger version of this figure.
| Column temperature | 40 °C |
| Solvent A | 99.9% water/0.1% formic acid (v/v) |
| Solvent B | acetonitrile |
| Elution program | 0–0.5 min, A = 20% |
| 0.5–2 min, A = 20%–50% | |
| 2–3 min, A = 50% | |
| 3–3.1 min, A = 50%–20% | |
| 3.1–5 min, A=20% | |
| Flow rate (mL/min) | 0.3 |
| Injection volume (μL) | 5 |
| Capillary temperature (°C) | 330 |
| Vaporizer temperature (°C) | 350 |
| Sheath gas flow rate (Arb) | 40 |
| Aux gas flow rate (Arb) | 20 |
| Spray voltage (V) | 3900 |
| Collision gas pressure (mTorr) | 1.5 |
| Precursor ion | 256.1 |
| Product ion/Collision energy (eV) | 208.8/16 |
Table 1: Elution program and ion source parameters of the liquid chromatography-mass spectrometry method.
Results
The instrument limit of detection (LOD) of imidacloprid was 5.76 × 10−14 g, and the method's LOD of imidacloprid in the wheat root or leaf was 0.01 µg/kg; no matrix effect was observed. The recovery yields of imidacloprid in wheat are shown in Table 2. The recovery yields of imidacloprid from the wheat roots exposed to imidacloprid concentrations of 0.5 mg/L and 5 mg/L were 94.0%-97.6% and 98.8%-99.2%, respectively; the coefficients of variation were 1.92% and 0.20%, respec...
Discussion
In recent years, methods for the pretreatment and detection of residues of the pesticide imidacloprid have been frequently reported. Badawy et al.23 used high-performance liquid chromatography to determine the content of imidacloprid in tomato fruit grown under greenhouse conditions and reported good linearity for imidacloprid in the range 0.0125-0.15 µg/mL. Zhai et al.24 used LC-MS-MS to study the residue of imidacloprid in Chinese chives. In the present study, the Qu...
Disclosures
The authors declare that they have no conflicts of interest. All the authors have read and approved the manuscript. This work has not been published previously, nor is it being considered by any other peer-reviewed journal.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 42277039).
Materials
| Name | Company | Catalog Number | Comments |
| Acetonitrile | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | 01-06-1995 | Suitable for HPLC, gradient grade, >99.9% |
| Analytical balance | Sartorius Lab Instruments Co.Ltd. | GL124-1SCN | |
| Artificial climate incubator | Shanghai Badian Instrument Equipment Co. Ltd. | HK320 | |
| Centrifuge | Eppendorf China Co. Ltd. | Centrifuge5804 | |
| Disposable syringe | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | Z116866 | Capacity 5 mL, graduated 0.2 mL, non-sterile |
| Formic acid | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | Y0001970 | European pharmacopoeia reference standard |
| Graphitized carbon black (GCB) | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | V900058 | 45 μm |
| H2O2 | Sigma-Aldrich (Shanghai) Trading Co.Ltd. | 31642 | 30% (w/w) |
| Hoagland’s Basal Salt Mixture | Shanghai Yu Bo Biotech Co. Ltd. | NS1011 | Anhydrous, reagent grade |
| Hydroponic equipment | Jiangsu Rongcheng Agricultural Science and Technology Development Co.Ltd. | SDZ04BD | |
| Hypersil BDS C18 column | Thermo Fisher Scientific (China) Co. Ltd. | 28103-102130 | |
| Imidacloprid | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | Y0002028 | European pharmacopoeia reference standard |
| MgSO4 | Sigma-Aldrich (Shanghai) Trading Co. Ltd. | 208094 | Anhydrous, reagent grade, >97% |
| NaCl | Sigma-Aldrich (Shanghai) Trading Co.Ltd. | S9888 | Reagent grade, 99% |
| pH meter | Shanghai Thunder Magnetic Instrument Factory | PHSJ-3F | |
| Phytotron box | Harbin Donglian Electronic Technology Co. Ltd. | HPG-280B | |
| Pipettes | Eppendorf China Co. Ltd. | Research plus | |
| Syringe filter | Sigma-Aldrich (Shanghai) Trading Co.Ltd. | SLGV033N | Nylon, 0.22 µm pore size, 33 mm, non-sterile |
| Ultra performance liquid chromatography tandem triple quadrupole mass spectrometry | Thermo Fisher Scientific (China) Co. Ltd. | UltiMate 3000 | |
| TSQ Quantum Access MAX | |||
| Vortex mixer | Shanghai Yetuo Technology Co. Ltd. | Vortex-2 | |
| Wheat seed | LuKe seed industry | Jimai 20 |
References
- Lin, P. C., Lin, H. J., Liao, Y. Y., Guo, H. R., Chen, K. T. Acute poisoning with neonicotinoid insecticides: A case report and literature review. Basic & Clinical Pharmacology & Toxicology. 112 (4), 282-286 (2013).
- North, J. H., et al. Value of neonicotinoid insecticide seed treatments in Mid-South corn (Zea mays) production systems. Journal of Economic Entomology. 111 (1), 187-192 (2018).
- Simon-Delso, N., et al. Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environmental Science and Pollution Research. 22 (1), 5-34 (2015).
- Bass, C., Denholm, I., Williamson, M. S., Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pesticide Biochemistry and Physiology. 121, 78-87 (2015).
- Craddock, H. A., Huang, D., Turner, P. C., Quiros-Alcala, L., Payne-Sturges, D. C. Trends in neonicotinoid pesticide residues in food and water in the United States, 1999-2015. Environmental Health. 18 (1), 7 (2019).
- Shao, X. S., Liu, Z. W., Xu, X. Y., Li, Z., Qian, X. H. Overall status of neonicotinoid insecticides in China: Production, application and innovation. Journal of Pesticide Science. 38 (1-2), 1-9 (2013).
- Zhao, Y., et al. Urinary neonicotinoid insecticides in children from South China: Concentrations, profiles and influencing factors. Chemosphere. 291, 132937 (2022).
- Kurwadkar, S., Evans, A. Neonicotinoids: Systemic insecticides and systematic failure. Bulletin of Environmental Contamination and Toxicology. 97 (6), 745-748 (2016).
- Sadaria, A. M., et al. Passage of fiproles and imidacloprid from urban pest control uses through wastewater treatment plants in northern California, USA. Environmental Toxicology and Chemistry. 36 (6), 1473-1482 (2017).
- Damalas, C. A., Eleftherohorinos, I. G. Pesticide exposure, safety issues, and risk assessment indicators. International Journal of Environmental Research and Public Health. 8 (5), 1402-1419 (2011).
- Hayes, T. B., et al. Demasculinization and feminization of male gonads by atrazine: Consistent effects across vertebrate classes. Journal of Steroid Biochemistry and Molecular Biology. 127 (1-2), 64-73 (2011).
- Rani, L., et al. An extensive review on the consequences of chemical pesticides on human health and environment. Journal of Cleaner Production. 283, 124657 (2021).
- Xu, Y. Q., et al. Ecotoxicity evaluation of azoxystrobin on Eisenia fetida in different soils. Environmental Research. 194, 110705 (2021).
- Yavari, S., Malakahmad, A., Sapari, N. B. Biochar efficiency in pesticides sorption as a function of production variables-A review. Environmental Science and Pollution Research. 22 (18), 13824-13841 (2015).
- Delcour, I., Spanoghe, P., Uyttendaele, M. Literature review: Impact of climate change on pesticide use. Food Research International. 68, 7-15 (2015).
- Zhang, C. Y., et al. The application of the QuEChERS methodology in the determination of antibiotics in food: A review. TrAC-Trends in Analytical Chemistry. 118, 517-537 (2019).
- Wiilkowska, A., Biziuk, M. Determination of pesticide residues in food matrices using the QuEChERS methodology. Food Chemistry. 125 (3), 803-812 (2011).
- Ishii, Y., et al. HPLC determination of the new insecticide imidacloprid and its behavior in rice and cucumber. Journal of Agricultural and Food Chemistry. 42 (12), 2917-2921 (1994).
- Ko, A. Y., et al. Development of a simple extraction and oxidation procedure for the residue analysis of imidacloprid and its metabolites in lettuce using gas chromatography. Food Chemistry. 148, 402-409 (2014).
- Yuan, W. L., et al. Application of imidacloprid controlled-release granules to enhance the utilization rate and control wheat aphid on winter wheat. Journal of Integrative Agriculture. 19 (12), 3045-3053 (2020).
- Phugare, S. S., Kalyani, D. C., Gaikwad, Y. B., Jadhav, J. P. Microbial degradation of imidacloprid and toxicological analysis of its biodegradation metabolites in silkworm (Bombyx mori). Chemical Engineering Journal. 230, 27-35 (2013).
- Li, Y., et al. Uptake, translocation and accumulation of imidacloprid in six leafy vegetables at three growth stages. Ecotoxicology and Environmental Safety. 164, 690-695 (2018).
- Badawy, M. E. I., Ismail, A. M. E., Ibrahim, A. I. H. Quantitative analysis of acetamiprid and imidacloprid residues in tomato fruits under greenhouse conditions. Journal of Environmental Science and Health Part B-Pesticides Food Contaminants and Agricultural Wastes. 54 (11), 898-905 (2019).
- Zhai, R. Q., et al. Residue, dissipation pattern, and dietary risk assessment of imidacloprid in Chinese chives. Frontiers in Nutrition. 9, 846333 (2022).
- Aria, M. M., et al. Uptake and translocation monitoring of imidacloprid to chili and tomato plants by molecularly imprinting extraction - ion mobility spectrometry. Microchemical Journal. 144, 195-202 (2019).
- Chen, Y., et al. Translocation and metabolism of imidacloprid in cabbage: Application of C-14-labelling and LC-QTOF-MS. Chemosphere. 263, 127928 (2021).
- Wild, S., Jones, K. Organic chemicals entering agricultural soils in sewage sludges: Screening for their potential to transfer to crop plants and livestock. Science of the Total Environment. 119, 85-119 (1992).
- Gong, W. W., et al. Uptake and dissipation of metalaxyl-M, fludioxonil, cyantraniliprole and thiamethoxam in greenhouse chrysanthemum. Environmental Pollution. 257, 113499 (2020).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved