Interne Standards
Überblick
Quelle: Labor von Dr. B. Jill Venton - University of Virginia
Das Ziel vieler chemischer Analysen ist eine Quantitative Analyse, wo die Menge einer Substanz in einer Probe bestimmt. Um die Konzentration eines unbekannten aus einer Stichprobe genau zu berechnen, ist die sorgfältige Probenvorbereitung Schlüssel. Jedes Mal, wenn eine Probe verarbeitet oder übertragen wird, können einige der Probe verloren gehen. Allerdings gibt es Strategien zur Minimierung der Probenverlust. Es gibt auch Strategien zur Bewältigung der Probenverlust und noch genaue Messungen der Konzentration.
Zur Minimierung von Probenverlust ist Ideal um die Anzahl der Beispielschritte Handhabung und Übertragung zu minimieren. Beispielsweise reduziert die Massierung von einer festen Probe direkt in eine Flasche, der eine Lösung erzielt werden einen Transfer-Schritt. Wenn es notwendig, aus einer Flasche auf einen anderen übertragen und eine Verdünnung erfolgt, hilft dreifach spülen der Gläser, sicherzustellen, dass die Probe übertragen wird. Andere Strategien sind spezifischer auf die Probe. Proben, die auf Glas, wie zum Beispiel Proteine, adsorbieren könnte beispielsweise besser in Einweg-Polypropylenröhrchen gehandhabt werden. Die Rohre sind nicht hydrophil, also wenn eine kleine Menge der Probe in Wasser pipettiert werden wird, es empfiehlt sich, bereits hinzugefügt haben, das Wasser mit dem Schlauch direkt in das Lösungsmittel die Probe pipettiert werden kann. Es möglicherweise besser zu konzentrieren, anstatt eine Probe durch Verluste aus Insolubilities nach Rehydratation vollständig trocknen.
Eine weitere Quelle von Probenverlust ist durch unvollständige Probe Manipulationen. Beispielsweise wird wenn eine Derivatisierung Verfahren verwendet wird und die Derivatisierung unvollständig ist, dann der volle Betrag der Probe nicht beobachtet. Fehler wie diese sind systematische Fehler und Behebung des Problems, wie das Ändern der Derivatisierung Verfahren gelöst werden können. Eine weitere Ursache für systematische Fehler bei Messungen ist Matrix-Effekte. Diese können Messung bestimmter Stoffe und darstellende Kalibrierungen in der gleichen Matrix stören, da die Probe kann diesen Effekt reduzieren.
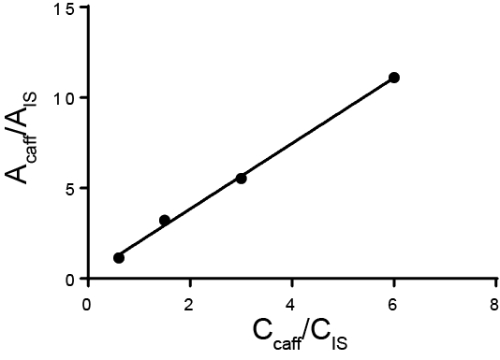
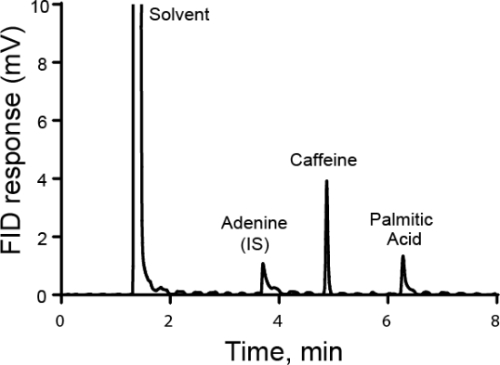
Quantitative Analyse wird in der Regel mit entweder extern oder intern Standards durchgeführt. Für externe Standards erfolgt eine Kalibrierkurve durch Messung unterschiedliche bekannteste Konzentration des Analyten von Interesse. Dann ist das Beispiel separat vom Standard ausgeführt. Für interne Standards ist der Standard in der gleichen Probe als der Analyten von Interesse, so dass die Messung gleichzeitig entnommen werden. In der Regel einer anderen Spezies wird hinzugefügt, dass interne Standard des internen Standards und das Verhältnis der Antwort gefordert und der Analyt wird berechnet. Die Idee ist, dass das Verhältnis der Reaktion, genannt der Responsefaktor proportional zu ihrer Konzentration. Während die Methode der Analyten von Interesse und dem internen Standard unterscheiden kann muss, sollte jede Probe-Verluste, die auftreten, nachdem der interne Standard hinzugefügt wird für beide Substanzen ähnlich und somit das Verhältnis der Antwort bleibt die gleiche. Ein besonderer Fall der Verwendung von internen Standards ist die Methode der Standardzusätzen, wo immer größere Mengen des Analyten wird der Projektmappe hinzugefügt und der Ursprungsbetrag des Analyten zurück berechnet. Interne Standards können in der Chromatographie, Elektrochemie und Spektroskopie verwendet werden.
Verfahren
1. richtige Probenbehandlung: Bildet eine Lösung
- Nehmen Sie ein sauberes Becherglas und Masse die richtige Menge der Probe hinein. Notieren Sie die tatsächliche Masse verwendet. In diesem Beispiel wird eine Lösung von Adenin in einem volumetrischen Kolben für den Einsatz als interner Standard für die nächste Analyse gemacht. Die Masse von Adenin ist 100 mg. Tun nicht direkt in einem volumetrischen Kolben Masse, denn es einen langen Hals hat und die Adenin kann nicht leicht hinzugefügt oder entfernt.
Anwendung und Zusammenfassung
Interne Standards dienen in vielen Bereichen, einschließlich Spektroskopie und Chromatographie. Spektroskopie hilfst internen Standards korrekt für zufällige Fehler aufgrund von Änderungen in der Intensität der Lichtquelle. Wenn eine Lampe oder andere Lichtquelle Variablen Leistung hat, wirkt sich die Absorption und infolgedessen Emission einer Probe. Jedoch wird das Verhältnis von internem Standard, Analyt bleiben konstant, auch wenn die Lichtquelle nicht der Fall ist. Ein Beispiel hierfür ist Lithium (Li) als in...
Tags
pringen zu...
Videos aus dieser Sammlung:

Now Playing
Interne Standards
Analytical Chemistry
204.8K Ansichten

Probenvorbereitung für die analytische Charakterisierung
Analytical Chemistry
84.7K Ansichten

Standard-Additionsverfahren
Analytical Chemistry
320.0K Ansichten

Kalibrierkurven
Analytical Chemistry
796.6K Ansichten

UV/Vis-Spektroskopie
Analytical Chemistry
623.3K Ansichten

Raman-Spektroskopie für die chemische Analyse
Analytical Chemistry
51.2K Ansichten

Röntgen-Fluoreszenz (XRF)
Analytical Chemistry
25.4K Ansichten

Gaschromatographie (GC) mit Flammen-Ionisations-Detektion
Analytical Chemistry
282.0K Ansichten

Hochleistungs-Flüssigkeitschromatographie (HPLC)
Analytical Chemistry
384.4K Ansichten

Ionenaustausch-Chromatographie
Analytical Chemistry
264.5K Ansichten

Kapillarelektrophorese (CE)
Analytical Chemistry
93.9K Ansichten

Einführung in die Massenspektrometrie
Analytical Chemistry
112.2K Ansichten

Rasterelektronenmikroskopie (SEM)
Analytical Chemistry
87.2K Ansichten

Elektrochemische Messungen von Trägerkatalysatoren mit einem Potentiostat / Galvanostat
Analytical Chemistry
51.4K Ansichten

Zyklische Voltammetrie (CV)
Analytical Chemistry
125.1K Ansichten
Copyright © 2025 MyJoVE Corporation. Alle Rechte vorbehalten