A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Bioluminescent Bacterial Imaging In Vivo
In This Article
Summary
This article describes the administration of lux-tagged bacteria to mice and subsequent in vivo analysis using IVIS bioluminescence imaging.
Abstract
This video describes the use of whole body bioluminesce imaging (BLI) for the study of bacterial trafficking in live mice, with an emphasis on the use of bacteria in gene and cell therapy for cancer. Bacteria present an attractive class of vector for cancer therapy, possessing a natural ability to grow preferentially within tumors following systemic administration. Bacteria engineered to express the lux gene cassette permit BLI detection of the bacteria and concurrently tumor sites. The location and levels of bacteria within tumors over time can be readily examined, visualized in two or three dimensions. The method is applicable to a wide range of bacterial species and tumor xenograft types. This article describes the protocol for analysis of bioluminescent bacteria within subcutaneous tumor bearing mice. Visualization of commensal bacteria in the Gastrointestinal tract (GIT) by BLI is also described. This powerful, and cheap, real-time imaging strategy represents an ideal method for the study of bacteria in vivo in the context of cancer research, in particular gene therapy, and infectious disease. This video outlines the procedure for studying lux-tagged E. coli in live mice, demonstrating the spatial and temporal readout achievable utilizing BLI with the IVIS system.
Protocol
1. Tumor Induction
- For routine tumor induction, the minimum tumorigenic dose of cells suspended in 200 μl of serum-free culture medium was injected subcutaneously (s.c.) into the flank of infection free 6-8 week old female Balb/C or athymic MF1-nu/nu mice n=6 (Harlan, Oxfordshire, UK) (1 x 106 4T1 cells) using a 21-gauge syringe needle. The viability of cells used for inoculation was greater than 95 % as determined by visual count using a haemocytometer and Trypan Blue Dye Exclusion (Gibco).
- Following tumor establishment, tumors were allowed to grow and develop and were monitored twice weekly. Tumor volume was calculated according to the formula V=(ab2) Π/6, where a is the longest diameter of the tumor and b is the longest diameter perpendicular to diameter a.
2. Bacterial Preparation
- The bacterial strain used in this protocol was E. coli K-12 MG1655, a non-protein-toxin-expressing strain, harboring a luxABCDE-encoding plasmid that enables the bacteria to be detected by BLI. E. coli MG1655 containing the integrated luxABCDE was grown aerobically at 37 °C in LB medium (Sigma-Aldrich, Ireland) supplemented with 300 μg/ml erythromycin (Em). The bioluminescent derivative of MG1655 was created using the plasmid p16Slux which contains the constitutive PHELPluxABCDE operon 1.
- For preparation for administration to mice, cultures were incubated in LB medium at 37 °C in a shaker at 200 rpm to mid-log phase (optical density at 600 nm). Bacteria were harvested by centrifugation (6,000 × g for 5 min), washed with PBS (Sigma), and diluted in PBS 1 × 107 colony forming units (cfu) /ml for IV administration, or 1 x 1010 for gavage.
3. Bacterial Administration
- Mice were randomly divided into experimental groups when tumors reached approximately 100 mm3 in volume. For intravenous administration, restrained mice each received 106 cells in 100 μl, injected directly into the lateral tail vein using a 28G syringe needle. The viable count of each inoculum was determined by retrospective plating.
- For GIT colonization studies, 109 bacterial cells were orally administered in 100 μl per mouse by gavage, on three consecutive days. Pre-existing commensal bacterial levels were decreased prior to feeding by addition of 5 mg/ml streptomycin in mouse drinking water for 7 days prior to commencement of oral gavage 1.
4. BioLuminescence Imaging
- 2D in vivo BLI imaging was performed using the IVIS100 (Caliper). At defined time points post bacterial administration, mice were anesthetized using Caliper's XGI-8 Gas Anesthesia System with 3% Isofluorane, and whole-body image analysis was performed in the IVIS 100 system for 2-5 min at high sensitivity.
- For 3D imaging, anesthetized mice were placed in a mouse imaging shuttle inside of the optical imaging system for dorsal imaging (IVIS Spectrum, Caliper). To acquire images of the bacterial luciferase signal for 3D optical reconstruction, emission filter wavelengths ranging from 500-580 nm were used with bin 16 acquisition times of 3-4 min per filter to maximize the signal to noise ratio. As part of this image acquisition sequence, a structured light image was obtained to define a height map. This map was input diffuse light imaging tomography (DLIT) reconstructions algorithms that were used to form a 3D optical image using a non-negative least squares optimization 2.
- Image Analysis: Regions of interest were identified and quantified using Living Image software (Caliper).
5. Representative Results
In this study, the non-pathogenic commensal bacteria E.coli K-12 MG1655 expressing the luxABCDE operon was IV administered to mice bearing s.c. 4T1 xenograft tumors. Bacterial lux signal was detected specifically in tumors of mice post IV-administration (Figure 2). Culture recovery of bacteria from sample mice validates the existence of a linear relationship between viable bacterial numbers and the quantity of light detected (Figure 3). In vivo imaging of orally-administered commensal bacteria in the GIT is also achieved using 3D BLI.
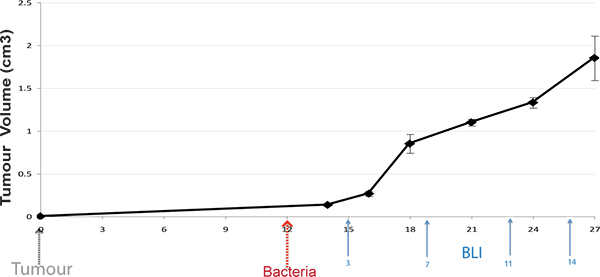
Figure 1. Protocol Timeline. Subcutaneous tumors are induced in mice, and bacteria administered upon tumor development (100 mm3). Live mice are BLI imaged at various time-points post bacterial administration (arrows display typical times).

Figure 2. Administration of E. coli MG1655 luxABCDE to tumor bearing mice. Subcutaneous 4T1 tumors were induced in MF1 nu/nu mice and E. coli MG1655 luxABCDE administered upon tumor development. Each animal received 106 cells injected directly into the lateral tail vein. Mice were imaged at four time points during the study (black dots z-axis and images) with subsequent recovery of viable bacteria (cfu) from tumors of sample sacrificed mice (bar graph). Increase in bacterial numbers and plasmid gene expression specifically in tumors was observed over time (representative mouse illustrated per time point). Click here to view larger figure.
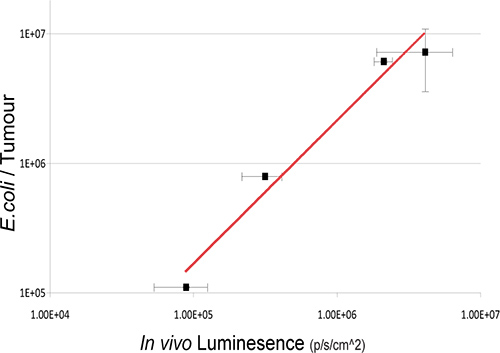
Figure 3. Relationship Between Intratumoral Bacterial Numbers And Bioluminescence. Viable bacteria in tumors were enumerated by ex vivo bacterial culture from tumors subsequent to BLI at various time-points post IV administration. Log values of bacterial numbers (cfu) relative to in vivo bioluminesce units are graphed. A robust correlation between bacterial counts and bacterial bioluminescence signals is observed R2=0.97171.
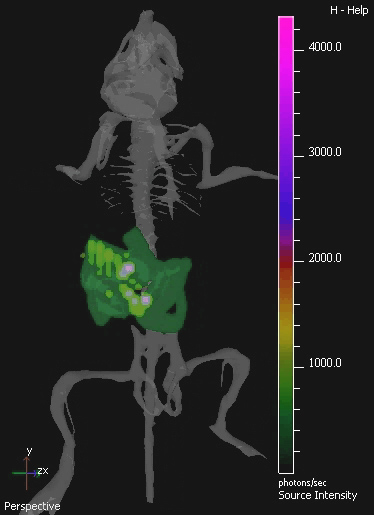
Figure 4. 3D IVIS Image Of Murine Gastrointestinal Tract Colonized By E. coli MG1655. The GIT of mice was colonized by oral administration of 109 cfu of E. coli for three consecutive days. A sample isolated image from 3D tomography of the colonized mouse is shown. 3D images show a digital mouse atlas of the skeleton to provide anatomical registration. E. coli MG1655 bioluminescence is visible in green at lower, and purple at higher levels.
Access restricted. Please log in or start a trial to view this content.
Discussion
In the context of gene therapy, the use of biological agents for delivery of therapeutic genes to patients has shown great promise 3-5. Like viruses, the innate biological properties of bacteria permit efficient DNA delivery to cells or tissues, particularly in the context of cancer. It has been shown that bacteria are naturally capable of homing to tumors when systemically administered resulting in high levels of replication locally, either external to (non-invasive species) or within tumor cells (pathogens)....
Access restricted. Please log in or start a trial to view this content.
Disclosures
No conflicts of interest declared.
Acknowledgements
The authors wish to acknowledge support relevant to this manuscript from the European Commission Seventh Framework Programme (PIOF-GA-2009-255466) and the Irish Health Research Board (HRA_POR/2010/138). Lux-tagged E. coli was a kind gift from Dr. Cormac Gahan, University College Cork.
Access restricted. Please log in or start a trial to view this content.
Materials
| Name | Company | Catalog Number | Comments |
| 4T1 cell line | ATCC | CRL-2539 | Syngeneic breast cancer model derived from a spontaneously arising BALB/c mammary tumor |
| DMEM | Sigma-Aldrich | D6429 | Dulbecco's Modified Eagle's Medium |
| PBS | Sigma-Aldrich | D8537 | Phosphate Buffered Saline |
| Xenogen IVIS | Caliper Life Sciences | IVIS 100 for 2D imaging; IVIS Spectrum for 3D. | |
| Luria Broth Miller (LB) | Sigma-Aldrich | L2542 | Growth medium for E. coli |
| Erythromycin | Sigma-Aldrich | E5389 | Antibiotic |
| Streptomycin | Sigma-Aldrich | S9137 | Antibiotic |
| MF1nu/nu mice | Harlan (UK) | 069(nu)/070(nu/+) | Hsd:Athymic Nude-Foxn1nu |
| Balb/c mice | Harlan (UK) | 066 | Haplotype:H-2d |
| Gavage needle | Vet-tech Solutions (UK) | DE009 | 22G x 38mm straight gavage needle |
| Syringe for IV injection | BD BioSciences | 309309 - 1 ml | Insulin syringe with 28 G x ½ inch micro-fine IV needle. |
| Syringe for tumor inoculation | Braun | 9161376V | Omnifix 26 G x ½ inch needle |
References
- Cronin, M., et al. High resolution in vivo bioluminescent imaging for the study of bacterial tumour targeting. PLoS One. 7, e30940(2012).
- Kuo, C., Coquoz, O., Troy, T. L., Xu, H., Rice, B. W. Three-dimensional reconstruction of in vivo bioluminescent sources based on multispectral imaging. J. Biomed. Opt. 12, 024007(2007).
- Tangney, M., Ahmad, S., Collins, S. A., O'Sullivan, G. C. Gene therapy for prostate cancer. Postgrad Med. 122, 166-180 (2010).
- Morrissey, D., O'Sullivan, G. C., Tangney, M. Tumour targeting with systemically administered bacteria. Curr. Gene Ther. 10, 3-14 (2010).
- Collins, S. A., et al. Viral vectors in cancer immunotherapy: which vector for which strategy. Curr. Gene Ther. 8, 66-78 (2008).
- Yu, Y. A., Zhang, Q., Szalay, A. A. Establishment and characterization of conditions required for tumor colonization by intravenously delivered bacteria. Biotechnol. Bioeng. 100, 567-578 (2008).
- Baban, C. K., Cronin, M., O'Hanlon, D., O'Sullivan, G. C., Tangney, M. Bacteria as vectors for gene therapy of cancer. Bioeng. Bugs. 1, 385-394 (2010).
- Cronin, M., et al. Orally administered bifidobacteria as vehicles for delivery of agents to systemic tumors. Mol. Ther. 18, 1397-1407 (2010).
- van Pijkeren, J. P., et al. A novel Listeria monocytogenes-based DNA delivery system for cancer gene therapy. Hum. Gene Ther. 21, 405-416 (2010).
- Ahmad, S., et al. Induction of effective antitumor response after mucosal bacterial vector mediated DNA vaccination with endogenous prostate cancer specific antigen. J. Urol. 186, 687-693 (2011).
- Riedel, C. U., et al. Improved luciferase tagging system for Listeria monocytogenes allows real-time monitoring in vivo and in vitro. Appl Environ Microbiol. 73, 3091-3094 (2007).
- Cheng, C. M., et al. Tumor-targeting prodrug-activating bacteria for cancer therapy. Cancer Gene Ther. 15, 393-401 (2008).
- Foucault, M. L., Thomas, L., Goussard, S., Branchini, B. R., Grillot-Courvalin, C. In vivo bioluminescence imaging for the study of intestinal colonization by Escherichia coli in mice. Appl. Environ. Microbiol. 76, 264-274 (2010).
- Collins, S. A., Hiraoka, K., Inagaki, A., Kasahara, N., Tangney, M. PET Imaging For Gene & Cell Therapy. Curr. Gene Ther. , (2012).
- Tangney, M., Francis, K. P. In vivo Optical Imaging in Gene & Cell Therapy. Curr. Gene Ther. , (2012).
Access restricted. Please log in or start a trial to view this content.
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved