Method Article
Isolating Mesangiogenic Progenitor Cells (MPCs) from Human Bone Marrow
* These authors contributed equally
In This Article
Summary
Here we describe an optimized, highly reproducible protocol to isolate Mesodermal Progenitor Cells (MPCs) from human bone marrow (hBM). MPCs were characterized by flow cytometry and nestin expression. They showed the ability to give rise to exponentially growing MSC-like cell cultures while retaining their angiogenic potential.
Abstract
In a research study aimed to isolate human bone marrow (hBM)-derived Mesenchymal Stromal Cells (MSCs) for clinical applications, we identified a novel cell population specifically selected for growth in human serum supplemented medium. These cells are characterized by morphological, phenotypic, and molecular features distinct from MSCs and we named them Mesodermal Progenitor Cells (MPCs). MPCs are round, with a thick highly refringent core region; they show strong, trypsin resistant adherence to plastic. Failure to expand MPCs directly revealed that they are slow in cycling. This is as also suggested by Ki-67 negativity. On the other hand, culturing MPCs in standard medium designed for MSC expansion, gave rise to a population of exponentially growing MSC-like cells. Besides showing mesenchymal differentiation capacity MPCs retained angiogenic potential, confirming their multiple lineage progenitor nature. Here we describe an optimized highly reproducible protocol to isolate and characterize hBM-MPCs by flow cytometry (CD73, CD90, CD31, and CD45), nestin expression, and F-actin organization. Protocols for mesengenic and angiogenic differentiation of MPCs are also provided. Here we also suggest a more appropriate nomenclature for these cells, which has been re-named as "Mesangiogenic Progenitor Cells".
Introduction
Mesenchymal Stromal Cells (MSCs) are of relevant clinical value for their multi-lineage differentiation capacity and their ability to support hemopoiesis, to secrete growth factors/cytokines as well as to play a role in immunoregulation1. In the definition of MSC-based therapies cell production and application have been the object of extensive clinical and pre-clinical research2, with particular attention to specific international regulation for the safety and efficacy of cell based medicinal product (CBMP) treatments3. Human MSCs are extensively cultured in media containing supplements and reagents of animal origin, such as fetal bovine serum (FBS) and bovine trypsin. Therefore, alongside with infectious risks associated with cell manipulation, patients also face prion exposure as well as immunological risks linked to proteins, peptides or other biomolecules of animal origin that could persist after cell harvesting and transplantation4.
To circumvent the problem, we cultured human bone marrow (hBM)-derived MSCs in animal-free medium, replacing FBS with pooled human AB type serum (PhABS). Under these conditions, alongside growing MSCs we identified a novel cell population. These cells were morphologically and phenotypically different from MSCs and showed a distinctive gene expression profile as well as characteristic growing/adhesion properties. They retained both mesengenic and angiogenic potential and therefore were named Mesodermal Progenitor Cells (MPCs)5. Subsequently, we were able to define selective and reproducible culture conditions to generate MPCs at high grade of purity6.
We further investigated the morphological and biological properties of MPCs. The MPCs showed to be nestin-positive, slow in cycling, Ki-67-negative, and with chromosomes characterized by long telomeres5. They expressed pluripotency-associated transcription factors Oct-4 and Nanog rather than MSC master regulators Runx2 and Sox97. Phenotypically, MPCs expressed endoglin (CD105) at lower level than MSCs while lacking mesenchymal markers CD73, CD90, CD166. MPCs also showed a distinctive pattern of adhesion molecules characterized by consistent expression of PECAM (CD31), integrins αL (CD11a), αM (CD11b), αX (CD11c) as well as integrin β2 (CD18) that specifically sustains podosome-like structures8. In standard MSC expansion media, MPCs promptly differentiated into MSCs through an intermediate stage featuring the activation of Wnt5/Calmodulin cell signaling9. MPCs also retained angiogenic properties, as demonstrated by their ability to sprout from spheroids in murine extracellular matrix (ECM) protein 3D cultures. The angiogenic potential was rapidly lost after MPC differentiation along the mesengenic lineage.
Here we present protocols optimized to isolate and to characterize highly purified MPCs from hBM blood samples. Reproducible protocols for MPC mesengenic and angiogenic differentiation are also described.
Protocol
NOTE: After written consent, hBM samples were obtained during orthopedic surgery for hip replacement. Immediately after femoral neck osteotomy and before femoral reaming a 20 ml syringe containing 500 U.I. of heparin, was used to aspirate fresh BM. The protocol is to be considered widely applicable to any BM source.
1. Isolation of Human Bone Marrow Mononuclear Cells (hBM-MNCs)
- Dilute 5 - 10 ml of fresh BM to 50 ml, applying Dulbecco's phosphate buffered saline solution (D-PBS) and mix by inversion. Equally distribute 25 ml in two new 50 ml conical tubes, add 25 ml of D-PBS to each tube and mix by inversion.
- Allow the tubes to stand for 10 min at room temperature, for separation of mineral bone fragments and fat from solution.
- Carefully remove floating fat with a sterile Pasteur pipette and filter cell suspension through 70 μm filters without disturbing the mineral bone fragment pellet.
- Set four 50 ml tubes with 15 ml of medium for discontinuous density gradient centrifugation (1.077 g/ml). Make sure that this medium is at room temperature.
- Gently lay 20 - 25 ml of diluted BM on top of density gradient medium. Perform this operation carefully, letting cell suspension trickle on the tube walls to prevent mixing layers.
- Carry out density gradient centrifugation at 400 x g for 30 min at room temperature with brake disabled.
- Collect the whitish ring of cells located between the two phases using a sterile Pasteur pipette and transfer it to a fresh 50 ml tube.
- Wash cells with fresh culture medium: phenol red-free, low glucose (1,000 mg/l) Dulbecco's Modified Eagle Medium (DMEM), 10% (v/v) pooled human AB type serum (PhABS), 2 mM L-glutamine and antibiotics (DMEM/10% PhABS). Centrifuge at 400 x g for 5 min.
- Aspirate supernatant and re-suspend pellet in 5 - 10 ml of fresh DMEM/10% PhABS.
- Proceed to cell count. Determine the number of white blood cells by 1:1 dilution in trypan. Apply this to the hemocytometer, and observe under a phase contrast microscope. Exclude small and perfectly rounded erythrocytes and blue-stained dead cells from the cell count.
NOTE: It is highly recommended to screen PhABS batches for their performance in MPC recovery. PhABS from USA sources have given best results while most sera of different origin resulted in MPC cultures with higher percentages of MSC-like cells.
2. Isolation of MPCs from hBM-MNCs
- Set hydrophobic T-75 flasks with 15 ml of fresh DMEM/10% PhABS and let pH and temperature equilibrate by pre-incubation at 37 °C in 5% CO2 for 30 min.
- Seed 4 - 6 x 107 hBM-MNCs per flask and incubate at 37 °C in 5% CO2 for 48 hr.
- Aspirate and discard medium and non-adherent cells from flasks. Add 15 ml of fresh DMEM/10% PhABS and incubate at 37 °C in 5% CO2. Maintain cultures for 6 - 8 days, changing medium every 48 hr.
OPTIONAL: To increase MPC yield, non-adherent cells from 2.3 could be re-plated in a new culture flask and maintained as described for the primary cultures. - Aspirate and discard medium from flasks, wash with fresh DMEM and add 2 ml of animal free protease detaching solution. Incubate at 37 °C for 5 - 15 min (avoid prolonged incubation).
- Add 10 ml of fresh DMEM/10% PhABS, aspirate the cell suspension and centrifuge at 400 x g for 5 min
- Aspirate and discard supernatant and re-suspend pellet in 1 - 2 ml of fresh DMEM/10% PhABS. Proceed to cell count as described in step 1.10.
NOTE: Do not use trypsin/EDTA as detaching reagent. MPCs are trypsin resistant. Morphological screening of the cultures, before cell harvesting, is highly recommended in order to evaluate the presence of spindle-shaped MSC-like cells. In case considerable amount of MSC-like cells are detected, increase the purity of the cell product by selectively removing the contaminated cells. To do so, trypsin digestion can be carried out prior to MPC harvesting, by adding 2 ml of trypsin/EDTA 0.05% for 2 min. Wash cultures twice with 5 ml of DMEM/10% PhABS, then proceed to protease treatment as above.
3. Cell Characterization
- Flow Cytometry
- Set up duplicate samples of 105 freshly detached cells in washing solution: D-PBS supplemented with 0.5% (v/v) bovine serum albumin (BSA) and 0.02% (w/v) sodium azide. Centrifuge at 400 x g for 5 min.
CAUTION: Sodium azide is toxic. - Re-suspend pellets in 200 µl of washing solution and add anti-CD90, anti-CD45, anti-CD73 and anti-CD31 antibodies conjugated with florescent dyes ("TEST"); in parallel set up isotype controls ("CTRL").
NOTE: Amounts of staining antibodies should be determined by titration or according to manufacturer's instructions. - Incubate samples at 4 °C for 30 min.
- Centrifuge at 400 x g for 5 min. Re-suspend cells in 500 µl of washing solution and acquire at least 5 x 104 events on multicolor flow cytometer 7 9 10.
- Analyze results by dot-plots and use "CTRL" recorded events to set quadrants.
NOTE: In order to define the culture as MPC culture the percentage of CD73negCD90negCD45+CD31+ cells should be over 95%. For some specific applications, i.e., gene expression analysis, the recovery cut-off has to be increased to 97 - 98%.
- Set up duplicate samples of 105 freshly detached cells in washing solution: D-PBS supplemented with 0.5% (v/v) bovine serum albumin (BSA) and 0.02% (w/v) sodium azide. Centrifuge at 400 x g for 5 min.
- Nestin detection and F-actin organization analysis
- Plate freshly isolated MPCs on culture chamber slides (20,000/cm2). Allow cells to adhere by overnight incubation at 37 °C in 5% CO2.
- Wash cells in washing solution and fix in 4% (w/v) para-formaldehyde at room temperature for 15 min. To remove fixative add washing solution, incubate for 2 min and pour.
- Repeat washing twice.
- Permeabilize cells in D-PBS supplemented with 0.05% (v/v) Triton X-100 for 15 min at RT.
- Stop reaction by protein free signal enhancer (30 min at RT) or standard blocking solution (D-PBS supplemented with 3% (w/v) BSA).
- Remove signal enhancer/blocking solution.
- Add 7 µg/ml (w/v) of anti-human nestin primary antibody and incubate at 4 °C overnight in a humidified chamber. In parallel, use isotypic control antibodies to evaluate not specific fluorescence signals.
- Wash slides by adding D-PBS, leave for 2 min and pour. Repeat twice.
- Add 2 µg/ml (w/v) of fluorescent dye conjugated secondary antibody and incubate at 4 °C for 1 hr in the dark.
- Wash slides as above.
- Add fluorescent Phalloidin (5 UI/ml), leave at RT for 30 min in the dark and wash 3 times in D-PBS.
- Remove the chamber walls and mount slides in aqueous mounting medium supplemented with antifade reagent and 4',6-diamidino-2-phenylindole (DAPI) for nuclei detection. Proceed to imaging 7 9 10.
4. Mesengenic Differentiation of MPCs
- Plate 2 x 104/cm2 freshly isolated MPCs in TC-treated T75 culture flasks and let cells adhere overnight in DMEM/10% PhABS at 37 °C in 5% CO2.
- Replace DMEM/10% PhABS with standard reduced serum medium, about 200 µl/cm2, designed for mesenchymal stromal cell expansion (MSC-RS medium). Grow cells to confluence (P1-MSCs), typically from 7 to 10 days from the induction. Refresh medium every 2 days.
- Aspirate and discard medium from flasks, wash with fresh MSC-RS medium and add 2 ml of animal free protease detaching solution. Incubate at 37 °C for 5 - 15 min (avoid prolonged incubation).
- Add 10 ml of fresh MSC-RS medium, aspirate the cell suspension and centrifuge at 400 x g for 5 min
- Aspirate supernatant and re-suspend pellet in 1 - 2 ml of fresh MSC-RS medium. Proceed to cell count as described in step 1.10
- Proceed to sub-culture them by seeding 3 - 5 x 103 cells/cm2. Grow cells to confluence (P2-MSCs).
- Harvest cells by protease digestion as described from step 4.3 to step 4.5 and proceed to characterization as described in section 3.
NOTE: A percentage of CD73+CD90+CD45negCD31neg cells lower than 95% would indicate only partial differentiation and require a further culture passage. - Plate P2-MSCs at 2 x 104/cm2 in six well TC-treated plates and grow to confluence in MSC-RS medium.
- Mark two wells as "No Diff" and refresh MSC-RS medium.
- Mark two wells as "Osteo" and replace medium with 200 µl/cm2 of standard osteogenic medium, specifically designed for MSC differentiation.
- Mark two wells as "Adipo" and replace medium with 200 µl/cm2 of standard adipogenic medium, specifically designed for MSC differentiation.
- Maintain cultures at 37 °C in 5% CO2 by changing the whole media each 48 hr.
NOTE: It is highly recommended to use standard commercially available differentiating media for test reproducibility. After 2/3 weeks under differentiating conditions, calcium deposits appear in osteogenic induced cultures, while intracellular lipid droplets accumulation is evident in adipogenic induced cells. - Aspirate and discard culture media then wash with D-PBS.
- Fix the cultures by adding 1 ml of 4% (w/v) para-formaldehyde for 15 min at room temperature.
- To remove fixative add D-PBS, incubate for 2 min and pour. Repeat washing twice.
- Stain one "No Diff" together with the two "Osteo" marked wells in hydroxyapatite specific fluorescent solution and one "No Diff" together with the two "Adipo" marked wells in 200 nM Nile Red solution. Incubate for 30 min at room temperature11,12.
- Remove staining solutions and wash in D-PBS twice.
- Remove D-PBS, add D-PBS supplemented with 50% (v/v) glycerin and proceed to imaging7 9 10.
5. MPC Spheroid Sprouting Assay
- To produce 3D spheroids, lay 20 µl drops of freshly isolated MPC suspension (1.5 x 104 cells/drop) on the inner surface of a Petri dish lid.
NOTE: As handling drops could lead to their rupture, it is highly recommendable to lay them in excess. - Carefully use the lid to recap a Petri dish containing D-PBS to prevent hanging drops evaporation. Incubate at 37 °C in 5% CO2 overnight to allow cells to aggregate in 3D spheroids.
- Set a thick gel of murine extracellular matrix (ECM) proteins by adding 300 µl aliquots of standard ECM proteins in a pre-refrigerated 24-well culture plate, and incubate at 37 °C for 30 min.
- Carefully tip over the Petri dish lid and gently pick up the spheroids using a sterile Pasteur pipette.
- Lay spheroids onto the ECM protein gel, add 700 µl aliquots of standard VEGF-rich endothelial cell growth medium and incubate at 37 °C in 5% CO2.
- After 24 hr and at 7 days of culture, take pictures of the 3D cultures at 4X magnification power. Evaluate sprouting from spheroids applying image analysis software by measuring the radial distance between last invading cell and the spheroid edge. Repeat measures along at least 20 different directions. Mean distance is deemed as positive when 50 µm or over.
Results
The selective culture conditions described here have allowed the isolation of a novel adherent and almost monomorphic cell population as 1.0% of the hBM-MNCs (0.5 - 2.0 x 106 hBM-MNCs from 5 - 10 ml of fresh BM samples)5,6. We identified these large (40 - 60 µm in diameter), rounded, quiescent, Ki-67-negative cells as MPCs5. Morphologically, they are characterized by a distinctive fried egg-shape with a thick core region surrounded by a flat thin periphery showing lots of filopodia at higher magnification power (white arrows in Figure 1 A.1). Polar elongation of the outer cell boundary is often observed (black arrows in Figure 1 A.1). Such morphology is clearly different from the typical spindle-shaped mesenchymal stromal cell appearance reported in standard MSC cultures. Flow cytometry showed over 95% of freshly isolated MPCs to express CD31 and CD45 while mesenchymal associated markers CD90 and CD7313 were undetectable (Figure 1 A.2). We regard this restricted set of four antigens as indicative for MPCs. Further distinguishing features of MPCs are dotted F-actin distribution revealing a number of podosome-like structures (red in Figure 1 A.3) and intense expression of nestin (green in Figure 1 A.3), which is not detected in the cells stained with isotypic control antibody (data not shown).
Culturing MPCs in standard RS medium designed for MSC expansion results in rapid differentiation into exponentially growing MSC-like cells (Figure 1 B.1). After two passages cells finally switch their phenotype from CD73negCD90negCD45+CD31+ to CD73+CD90+CD45negCD31neg (Figure 1 B.2). In the process, MPCs re-organize F-actin into stress fibers while nestin expression becomes confined to few rare cells (Figure 1 B.3). MPC mesengenic differentiation into a definite MSC-like phenotype occurs through two distinct steps revealed by different cell morphologies. After one week in MSC RS medium a residual population of MPC-like cells is still detectable within a confluent layer of flat, polygonal multi-branched cells (P1-MSCs in Figure 2 A). A further passage is required to obtain an almost monomorphic culture of spindle-shaped MSC-like cells (P2-MSCs in Figure 2 A). These exponentially growing cells can readily differentiate into osteoblasts or adipocytes when transferred into selective media for at least 2 weeks, thus confirming their MSC nature. In osteogenic induced cultures, calcium deposits can be detected by either colorimetric Alizarin-S stain or specific fluorescent dyes (green in Figure 2 B). After adipogenic induction cells show lipid droplet accumulation as revealed by either colorimetric Oil Red or fluorescent Nile Red stain (red in Figure 2 B).
MPC typing was confirmed by sprouting angiogenesis assay. MPCs showed their ability to invade (over 50 µm) murine ECM protein gel from 3D spheroids after 24 hr VEGF-stimulus (Figure 2 C). After one week invading cells were detected at 300 - 600 µm distance. Conversely, invasion capacity was lost in P2-MSCs after mesengenic differentiation (Figure 2 D).
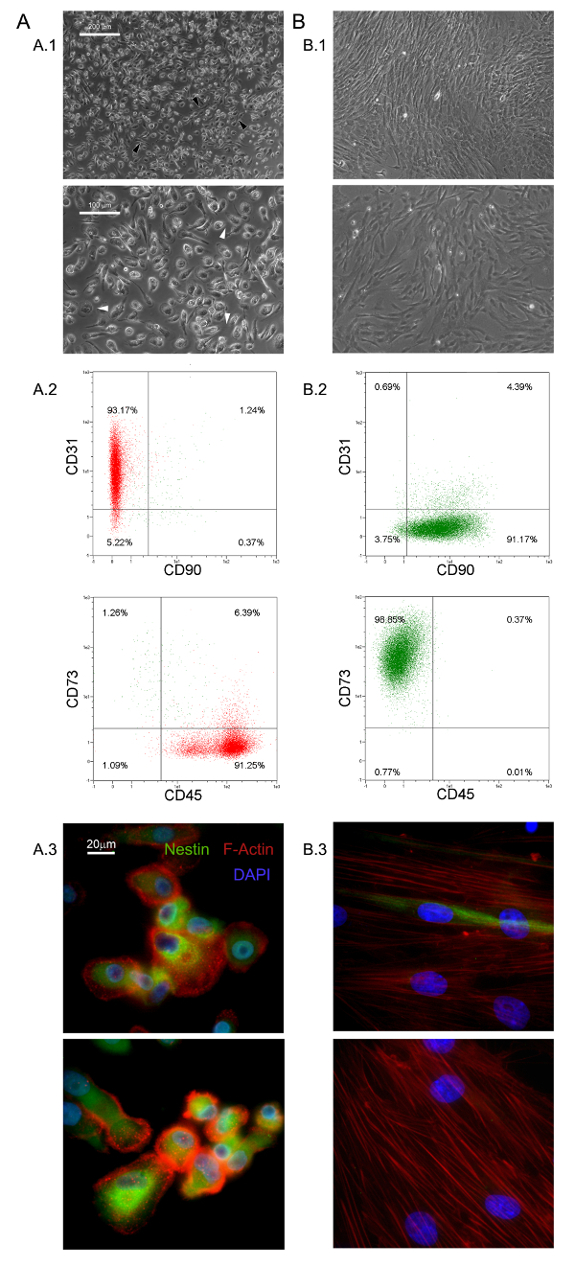
Figure 1. Freshly Isolated MPCs have Distinctive Features. Culturing hBM-MNCs in DMEM/10% PhABS for seven days gives rise to a population of quiescent MPCs (A) easily distinguishable from MSCs (B) in terms of morphology (A.1, B.1, scale bars = 100 µm), phenotype (A.2, B.2), F-actin distribution (red in A.3, B.3), and nestin expression (green in A.3, B.3, scale bars = 20 µm). Please click here to view a larger version of this figure.
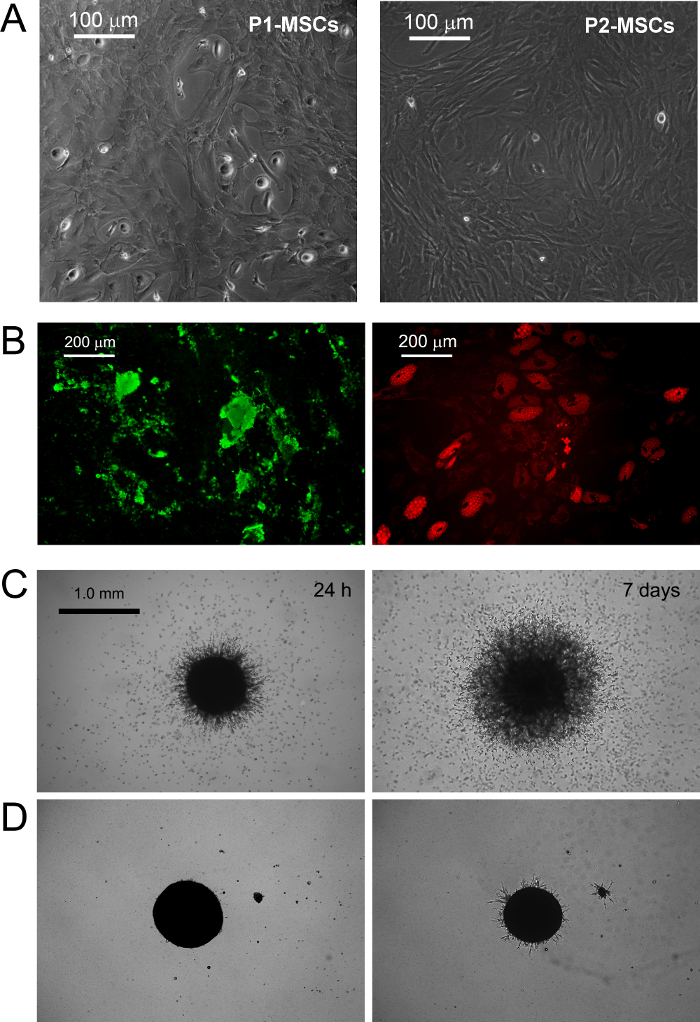
Figure 2. MPCs Differentiate into Standard MSCs and Show Sprouting Angiogenesis in vitro. Replacement of DMEM/PhABS with commercially available RS medium designed for MSC expansion triggers mesengenic induction of MPCs. After one week in culture few residual MPCs are still detectable (P1-MSCs) while a further passage in MSC-RS medium leads to a population of confluent MSC-like cells (P2-MSCs, A scale bars = 100 µm). P2-MSCs terminally differentiate into osteocytes or adipocytes under proper stimuli as revealed by calcium deposition (green in B) and lipid droplet accumulation (red in B, scale bars = 200 µm), respectively. MPCs show consistent sprouting from spheroids in murine ECM protein gel (C) at a difference with P2-MSCs (D, scale bars = 1.0 mm). Please click here to view a larger version of this figure.
Discussion
In the last decades, MSCs have been extensively researched and pre-clinically evaluated for possible application in the treatment of various bone/articular, immunological, neurological, cardiovascular, gastrointestinal and hematological disorders14,15. The easy and inexpensive isolation of multipotent MSCs, from many different tissues, together with their lack of significant immunogenicity16, contribute to make these cells one of the most interesting cell population to be applied in cell based therapies. Nonetheless, the very low frequency in the tissue of origin represents a great limitation to the MSCs application in clinics, forcing the expansion of these cells, in vitro, before the infusion or transplantation.
Expanded MSC cultures have revealed high grades of heterogeneity and variability17-19 making it difficult to reach a consensus about MSC production and characterization protocols. Moreover, recent investigations suggested the presence of multiple in vivo MSC ancestors in a wide range of tissues, which contribute to culture heterogeneity10,20. In fact, it has been proposed that particular culture conditions possibly select or simply promote specific sub-populations of MSCs progenitors present, in various percentages, in "crude" and unprocessed samples like bone marrow (hBM-MNCs) or adipose tissues (stromal vascular fraction)2. Thus, the variability in MSC-initiating cell populations together with the great number of different enrichment/isolation and culture protocols applied, represent a great obstacle to the definition of feasible MSC-based therapies.
A crucial factor affecting heterogeneity of MSC cultures is serum supplementation21. In our hands replacement of FBS with PhABS in primary cultures from hBM-MNCs, combined with high density seeding on hydrophobic plastics, led to the isolation of a novel highly adherent cell population with distinct biological features named MPCs5,6. We observed that the addition of small percentages of PhABS to FBS primary cultures also allowed MPC isolation, suggesting the presence of MPC inducing agents in the human serum6. At the moment, the MPC isolation/characterization protocol is a unique method available to obtain almost pure MPCs. The protocol has been carefully adjusted and it is highly reproducible for quality screening of MPC preparations before further applications.
MPCs could be used as a source for MSC production, thus limiting the variability introduced by use of unfractionated starting material. The precise definition of the multiple steps characterizing MPC mesengenic differentiation reported9 would allow synchronized mesenchymal cell expansion. Nonetheless, this latest condition could be realized exclusively applying highly purified MPC population, as a consequence the characterization of the cell products obtained by the protocol described here, results of crucial importance. This isolating method has been reported allowing MPC recovery with purity generally around 95%. However, donor/patient variability together with the variability related to the different batches of human pooled serum applied, could lead to a significant percentage of MSC-like cells co-isolated together with MPCs, under selective conditions.
It is not clear if these "contaminating" MSC-like cells could arise from the other different in vivo progenitors described in bone marrow22 or from uncontrolled and spontaneous MPC differentiation. In any case, a consistent percentage of MSC-like cells in the MPC products nullify the possibility to applying these cells as homogeneous starting material for the MSC expansion. Thus, here it has been suggested a simple and inexpensive method, based on the MPC resistance to trypsin digestion, increasing the purity of the MPC products. Similar or even better results in purifying MPC cultures could be achieved by fluorescent or magnetic cell sorting performing CD73 and/or CD90 depletion, but significantly prolonging the process time and increasing the costs.
Moreover, MPCs showed expression of pluripotency-associated markers and Nestin, all rapidly lost during mesengenic differentiation7. Sprouting assay revealed MPC ability to invade murine ECM protein gel. Taken together these results indicate that MPCs have to be considered a more immature progenitor, retaining angiogenic potential. Nonetheless, the initial enthusiasm about mesodermal differentiation potential of MPCs is actually waning. In fact, after more than 7 years of studies on MPCs, mesengenic and angiogenic potential have been extensively described5-9, but differentiation toward any other cells of mesodermal origin is still lacking. Thus, here we propose a new, and more rigorous, definition of these cells as "Mesangiogenic Progenitor Cells", maintaining the acronym MPCs.
We also believe that most controversies about MSC angiogenic potential could be related to the heterogeneous composition of expanded cultures consisting of sub-populations of MPCs and MSCs in variable percentages23.
Finally, MPCs could also play a crucial role for the implementation of CBMPs applicable for tissue reconstruction, as these cells could also support the neo-vascularization. In fact, future studies on regeneration should take in consideration that the newly formed tissue growth should be supported by concomitant neo-angiogenesis. The co-existence of mesengenic and angiogenic potential in MPCs could significantly improve the regeneration potential of new therapeutic approaches that involve these interesting cells.
Disclosures
The authors have no competing financial interests or other conflicts of interest.
Acknowledgements
The Authors would especially like to thank Dr. Paolo Parchi, department of Surgical, Medical and Molecular Pathology and Critical Care Medicine, University of Pisa, for providing bone marrow samples and his expertise in human osteo-progenitors
Materials
| Name | Company | Catalog Number | Comments |
| Matrigel Basement Membrane Matrix | BD Bioscience (San Jose, CA-USA) | 354230 | Murine ECM proteins Stock Concentration: 100% (9 - 12 mg/ml) Final Concentration: 100% |
| Dulbecco's Phosphate-Buffered Saline (D-PBS) | Sigma (St. Louis, MO, USA) | D8537 | |
| 70 μm Filters | Miltenyi Biotec (BergischGladbach, Germany) | 130-095-823 | |
| Ficoll-Paque PREMIUM | GE Healthcare (Uppsala, Sweden) | 17-5442-03 | medium for discontinuos density gradient centrifugation |
| Pooled human AB type serum (PhABS) | LONZA (Walkersville MD-USA) | 14-490E | Final Concentration: 10% |
| Glutamax-I | ThermoFisher (Waltham, MA USA) | 35050-038 | Stabilized L-Glutamine Stock Concentration: 100x Final Concentration: 2 mM |
| Bovine Serum Albumin (BSA) | Sigma (St. Louis, MO, USA) | A8412 | Stock Concentration: 7.5% Final Concentration: 0.5% |
| Sodium Azide | Sigma (St. Louis, MO, USA) | S8032 | Final Concentration: 0.02% |
| Penicillin/Streptomycin (Pen Strep) | Gibco (Grand Island, NY, USA) | 15070-063 | Antibiotics Stock Concentration: 5,000 UI/ml penicillin, 5,000 μg/ml Streptomycin Final Concentration: 50 UI/ml penicillin, 50 μg/ml Streptomycin |
| T-75 culture flask for suspension cultures | Greiner Bio-one (Frickenhausen, Germany) | 658 190 | |
| T-75 culture flask TC treated | Greiner Bio-one (Frickenhausen, Germany) | 658170 | |
| TrypLE Select | ThermoFisher (Waltham, MA USA) | 12563-011 | Animal-free proteases detaching solution Stock Concentration: 1x Final Concentration: 1x |
| Trypsin/EDTA | ThermoFisher (Waltham, MA USA) | 15400-054 | Phenol red free Stock Concentration: 0.5% Final Concentration: 0.25% |
| anti-CD90 APC antibody (CD90) | MiltenyiBiotec (BergischGladbach, Germany) | 130-095-402 | Final Concentration: 1:40 |
| anti-CD45 APC-Vio770 antibody (CD45) | MiltenyiBiotec (BergischGladbach, Germany) | 130-096-609 | Final Concentration: 1:40 |
| anti-CD73 PE antibody (CD73) | MiltenyiBiotec (BergischGladbach, Germany) | 130-095-182 | Final Concentration: 1:40 |
| anti-CD31 PE Vio-770 antibody (CD31) | MiltenyiBiotec (BergischGladbach, Germany) | 130-105-260 | Final Concentration: 1:40 |
| Mouse IgG1 APC antibody | MiltenyiBiotec (BergischGladbach, Germany) | 130-098-846 | Final Concentration: 1:40 |
| Mouse IgG2a APC Vio770 antibody | MiltenyiBiotec (BergischGladbach, Germany) | 130-096-637 | Final Concentration: 1:40 |
| Mouse IgG1 PE antibody | MiltenyiBiotec (BergischGladbach, Germany) | 130-098-845 | Final Concentration: 1:40 |
| Mouse IgG1 PE Vio-770 antibody | MiltenyiBiotec (BergischGladbach, Germany) | 130-098-563 | Final Concentration: 1:40 |
| Low Glucose Dulbecco's Modified Eagle Medium (DMEM) | ThermoFisher (Waltham, MA USA) | 13-1331-82 | Phenol red-free minimal essential medium Stock Concentration: 1,000 mg/L glucose |
| Fetal Bovine Serum (FBS) | ThermoFisher (Waltham, MA USA) | 10500 | Stock Concentration:0.2 mg/ml Final Concentration: 2 μg/ml |
| Prolong Gold antifade reagent with 4’,6-diamidino-2-phenylindole | Invitrogen (Waltham, MA, USA) | P-36931 | Aqueous mounting medium + DAPI Final Concentration: 1x |
| Paraformaldehyde | Sigma (St. Louis, MO, USA) | P6148 | Fixative Final Concentration: 4% |
| LAB-TEK two-well chamber slides | Sigma (St. Louis, MO, USA) | C6682 | |
| Anti-Nestin antibody [clone 10C2] | Abcam (Cambridge, UK) | ab2035 | Stock Concentration: 1 mg/ml Final Concentration: 7 μg/ml |
| Alexa Fluor 555 Phalloidin | ThermoFisher (Waltham, MA USA) | A34055 | Stock Concentration: 200 UI/ml Final Concentration: 5 UI/ml |
| Triton X-100 | Euroclone (Milan, Italy) | EMR237500 | Final Concentration: 0.05% |
| MesenPRO RS Medium (MSC-RS medium) | ThermoFisher (Waltham, MA USA) | 12746-012 | |
| Alexa Fluor 488 anti-mouse SFX kit | ThermoFisher (Waltham, MA USA) | A31619 | Goat anti-mouse secondary antibody + Signal enhancer Stock Concentration: 2 mg/ml Final Concentration: 2 μg/ml |
| Pasteur Pipette | Kartell Labware (Noviglio (MI), ITALY ) | 329 | |
| StemMACS AdipoDiff Media | MiltenyiBiotec (BergischGladbach, Germany) | 130-091-679 | |
| StemMACS OsteoDiff Media | MiltenyiBiotec (BergischGladbach, Germany) | 130-091-678 | |
| Osteoimage Bone mineralization Assay | LONZA (Walkersville MD-USA) | PA-1503 | Hydroxyapatite specific fluorescent staining solution |
| 50 ml Polystyrene conical tube | Greiner bio-one (Kremsmünster Austria) | 227261 | |
| Nile Red | ThermoFisher (Waltham, MA USA) | N1142 | Fluorescent staining solution for lipids Stock Concentration: 100 mM Final Concentration: 200 Nm |
| Glycerin | Sigma (St. Louis, MO, USA) | G2289 | Final Concentration: 50% |
| Polistirene Petri dishes | Sigma (St. Louis, MO, USA) | P5606 | |
| 24-well plates TC-treated | Greiner Bio-one GmbH (Frickenhausen, Germany) | 662160 | |
| Endothelial Growth Medium, EGM-2 BulletKit (EGM-2) | LONZA (Walkersville MD-USA) | CC-3162 | VEGF-rich endothelial cell growth medium |
| Leica Qwin Image Analisys Software | Leica (Wetzlar, Germany) | Image analysis software |
References
- Stoltz, J. F., et al. Stem Cells and Regenerative Medicine: Myth or Reality of the 21th Century. Stem Cells Int. 2015, 734731 (2015).
- Pacini, S. Deterministic and stochastic approaches in the clinical application of mesenchymal stromal cells (MSCs). Front Cell Dev Biol. 2, 50 (2014).
- Galvez, P., Clares, B., Hmadcha, A., Ruiz, A., Soria, B. Development of a cell-based medicinal product: regulatory structures in the European Union. Br Med Bull. 105, 85-105 (2013).
- Herberts, C. A., Kwa, M. S., Hermsen, H. P. Risk factors in the development of stem cell therapy. J Transl Med. 9, 29 (2011).
- Petrini, M., et al. Identification and purification of mesodermal progenitor cells from human adult bone marrow. Stem Cells Dev. 18 (6), 857-866 (2009).
- Trombi, L., et al. Selective culture of mesodermal progenitor cells. Stem Cells Dev. 18 (8), 1227-1234 (2009).
- Pacini, S., et al. Constitutive expression of pluripotency-associated genes in mesodermal progenitor cells (MPCs). PLoS One. 5 (3), 9861 (2010).
- Pacini, S., et al. Specific integrin expression is associated with podosome-like structures on mesodermal progenitor cells. Stem Cells Dev. 22 (12), 1830-1838 (2013).
- Fazzi, R., et al. Mesodermal progenitor cells (MPCs) differentiate into mesenchymal stromal cells (MSCs) by activation of Wnt5/calmodulin signalling pathway. PLoS One. 6 (9), 25600 (2011).
- Tormin, A., et al. CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood. 117 (19), 5067-5077 (2011).
- Greenspan, P., Mayer, E. P., Fowler, S. D. Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol. 100 (3), 965-973 (1985).
- Wang, Y. H., Liu, Y., Maye, P., Rowe, D. W. Examination of mineralized nodule formation in living osteoblastic cultures using fluorescent dyes. Biotechnol Prog. 22 (6), 1697-1701 (2006).
- Horwitz, E. M., et al. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy. 7 (5), 393-395 (2005).
- Wang, S., Qu, X., Zhao, R. C. Clinical applications of mesenchymal stem cells. J Hematol Oncol. 5, 19 (2012).
- Si, Y. L., Zhao, Y. L., Hao, H. J., Fu, X. B., Han, W. D. MSCs: Biological characteristics, clinical applications and their outstanding concerns. Ageing Res Rev. 10 (1), 93-103 (2011).
- Le Blanc, K., Tammik, C., Rosendahl, K., Zetterberg, E., Ringden, O. HLA expression and immunologic properties of differentiated and undifferentiated mesenchymal stem cells. Exp Hematol. 31 (10), 890-896 (2003).
- Phinney, D. G. Biochemical heterogeneity of mesenchymal stem cell populations: clues to their therapeutic efficacy. Cell Cycle. 6 (23), 2884-2889 (2007).
- Phinney, D. G. Functional heterogeneity of mesenchymal stem cells: implications for cell therapy. J Cell Biochem. 113 (9), 2806-2812 (2012).
- Tolar, J., Le Blanc, K., Keating, A., Blazar, B. R. Concise review: hitting the right spot with mesenchymal stromal cells. Stem Cells. 28 (8), 1446-1455 (2010).
- Corselli, M., et al. The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 21 (8), 1299-1308 (2012).
- Bieback, K., et al. Human alternatives to fetal bovine serum for the expansion of mesenchymal stromal cells from bone marrow. Stem Cells. 27 (9), 2331-2341 (2009).
- Watson, L., Elliman, S. J., Coleman, C. M. From isolation to implantation: a concise review of mesenchymal stem cell therapy in bone fracture repair. Stem Cell Res Ther. 5 (2), 51 (2014).
- Pacini, S., Petrini, I. Are MSCs angiogenic cells? New insights on human nestin-positive bone marrow-derived multipotent cells. Front Cell Dev Biol. 2, 20 (2014).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved