A subscription to JoVE is required to view this content. Sign in or start your free trial.
Methods Article
Randomized Controlled Trial to Study the Acute Effects of Strength Exercise on Insulin Sensitivity in Obese Adults
In This Article
Summary
This study describes a randomized controlled trial protocol aiming at assessing the acute effects of strength exercise volume on insulin sensitivity in obese individuals.
Abstract
An acute session of strength exercise (SE) ameliorates insulin sensitivity (IS) for several hours; however, the effects of SE volume (i.e., number of sets) have not been studied thoroughly. Although it is intuitive that some SE is better than none, and more is better than some for the improvement of IS, high-volume sessions might be challenging for diseased populations to complete, especially obese adults, for whom even a brisk walk can be challenging. This protocol details a randomized clinical trial to assess the acute effects of SE on IS in obese adults. The inclusion criteria are body mass index >30 kg/m2, central obesity (waist circumference >88 cm and >102 cm for women and men, respectively), and age >40 years. Participants will be familiarized with the SE (7 exercises targeting major muscle groups) and then will perform three sessions in a randomized order: session 1 - high-volume session (3 sets/exercise); session 2 - low-volume session (1 set/exercise); session 3 - control session (no exercise). Diet will be controlled the day before and on the day of the sessions. Sessions will be completed at night, and an oral glucose tolerance test will be performed the next morning, from which several indexes of IS will be derived, such as the area under the curve (AUC) of glucose and insulin, the Matsuda index, the Cederholm index, the muscle IS index, and the Gutt index. Based on pilot studies, we expect ~15% improvement in IS (insulin AUC, and Matsuda and Cederholm indexes) after the high-volume session, and ~8% improvement after the low-volume session compared to the control session. This study will benefit individuals who find high-volume SE sessions challenging but still aim to improve their IS by investing 1/3 of their time and effort.
Introduction
Although the chronic effects of strength training on insulin sensitivity have been repeatedly shown1,2,3, even an acute session of strength exercise can improve insulin action for up to 48 h4. This effect has been demonstrated in healthy5,6,7,8, obese 9, older10, insulin-resistant individuals4, and type 2 diabetes mellitus patients11. Others have not reported positive effects12,13,14,15,16,17, and it is unclear why these differences occur.
In a recent narrative review18, it was suggested that strength exercise volume (the number of sets per exercise) is essential to improve insulin sensitivity. For instance, a recent systematic review and meta-analysis demonstrated that sessions composed of 21 sets or more led to a greater improvement in insulin action compared to sessions with fewer than 21 sets19. However, only limited evidence from literature directly supports this notion. For example, higher strength exercise volume (30 sets) improved glucose metabolism more than lower volume (10 sets)20. But it is worth noting that this study implemented circuit-style strength exercises, which limits the comparison to traditional strength exercises. In another study, better insulin sensitivity was observed after a 32-set strength exercise protocol compared to an 8-set protocol21. However, the degree of effort after sets was not reported, and it was presumably greater after the high-volume protocol. This is important because the degree of effort (or proximity to concentric muscular failure, characterized as the inability to continue the set due to failure in the concentric movement of a given repetition) has also been considered an important variable to improve insulin and glucose metabolism18. Thus, the limited available studies on the topic, along with their methodological limitations, preclude further inferences regarding the effects of strength exercise volume on insulin sensitivity.
Another interesting point when discussing strength exercise volume is that it is inherently connected to time commitment. A lower exercise volume, by design, means less time spent in the gym. Among the reasons for not adhering to an exercise program, lack of time is at the top of the list22. Thus, a low-volume strength exercise session that effectively improves insulin sensitivity means less time commitment23 and might result in higher long-term adherence. Furthermore, subjective feelings, such as self-efficacy (self-perception of the ability to accomplish something) and the sensations of pleasure and fun (enjoyment), are also related to exercise adherence24,25,26. It is reasonable to speculate that people might feel more confident and enjoy their exercise experience more when they perform a low-volume strength exercise session that translates into health improvement.
To address the gaps in the literature summarized above, we describe a protocol for a randomized, controlled, crossover, clinical trial with the primary aim of assessing the effects of strength exercise volume on insulin sensitivity in obese individuals. As a secondary aim, we assess the effects of strength exercise volume on subjective feelings (self-efficacy, affection, and enjoyment).
The protocol here describes a randomized, controlled, 3-way, crossover, clinical trial. The chronological sequence of the protocol includes: a pre-participation assessment of health history and anthropometric measures (body mass, height, waist circumference, and body composition); an appointment with the team's certified nutritionist; a familiarization period with the strength exercises and subjective feelings questionnaires; strength assessment in each exercise; random allocation of the order of the sessions; performance of the 3 sessions (separated by 7-28 days), that is immediately followed by answering the subjective feelings questionnaires; an oral glucose tolerance test (OGTT) the next morning; and data analysis. Figure 1 delineates the protocol design.
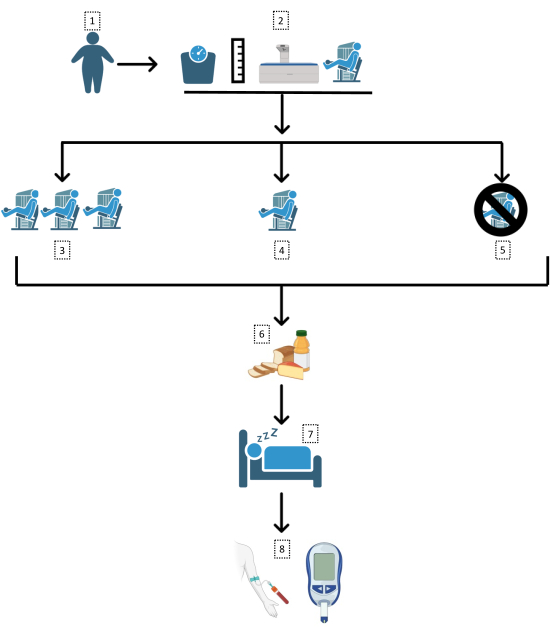
Figure 1: Study design. A flowchart of the trial methodology followed here. 1: Obese (body mass index [BMI] >30 kg/m2; waist circumference >102/88 cm) subjects; 2: Anthropometric assessments and familiarization; random assignment to 3: high-volume strength exercise session (21 sets), 4: low-volume strength exercise session (7 sets), or 5: control day; 6: standard meal following sessions; 7: sleep and overnight fast; 8: oral glucose tolerance test. Please click here to view a larger version of this figure.
Protocol
Obtain ethical clearance for the study by submitting the study protocol and informed consent document to the local Institutional Review Board (IRB) or local Research Ethics Committee (REC). The study can only begin after IRB or REC approval. The results presented below are from a pilot study, for which subjects signed a written informed consent before enrollment. After IRB or REC approval, prospectively register the protocol in a public repository, like Clinical Trials (https://clinicaltrials.gov). If your country has a national repository, register the protocol there (for example, in Brazil, there is the Brazilian Clinical Trials (https://ensaiosclinicos.gov.br). The present study was approved by the local IRB (certificate number CAAE 63190422.0.0000.5108) and was prospectively registered in a clinical trial registry (ReBEC #RBR-3vj5dc5 https://ensaiosclinicos.gov.br/rg/RBR-3vj5dc5).
1. Selection and preparation of volunteers
- Calculate the sample size required for the trials
- Select subjects that: are obese (body mass index >30 kg/m2); have central obesity (waist circumference > 102/88 cm in men/women); are aged >40 years; are capable of performing strength exercises.
- Exclude subjects that: show signs, symptoms, or presence of diabetes or any other metabolic disease, cardiovascular disease, cerebrovascular disease, kidney disease, respiratory disease, and osteoarticular disease27; report using any medication that may influence the expected results (including oral contraceptives28); report using of anabolic steroids; are pregnant or intend to get pregnant during the course of the study; describe dietary supplement intake known to affect exercise performance, such as caffeine, beta-alanine, creatine, and sodium bicarbonate29.
- Get the participant's signature in the informed consent and store it in a private locker.
2. Measure the participant's body composition
- For this study, evaluate body composition by means of dual-energy X-ray absorptiometry, following the manufacturer's instructions for analysis of fat mass, fat-free mass. However, any other established method (i.e., Bod Pod, under water weighing, skin fold) can be used.
3. Dietary control
- Schedule an appointment with the certified nutritionist of the research team to instruct participants on how to log their 3-day (2 weekdays, 1 weekend) diet.
- Develop a food plan with the nutritionist based on participant energy needs and food intake, to ensure an isocaloric diet the day before and on the day of the sessions30.
4. Familiarization
- Have participants perform 5 familiarization sessions, separated by at least 2 days.
- Familiarization session 1
- Instruct participant on lifting form: range of motion, duration of concentric and eccentric phases (~3 s total rep duration) in each of the 7 strength exercises (1 - hex bar squat; 2 - bench press; 3 - leg press; 4 - lat pulldown; 5 - leg extension; 6 - shoulder press; 7 - leg curl).
- Instruct participant on how to rate their effort based on the OMNI-RES scale30.
- Have participant perform a warm-up of 5 min by walking on a treadmill at ~3-4 km/h.
- Have participant perform 3 sets of 8 repetitions in each of the 7 exercises that they consider easy or between levels 3 and 4 according to the OMNI-RES scale.
- Allow 90 to 120 s recovery between sets and exercises.
- Familiarization session 2
- Instruct participant on how to answer the affection scale31, 32, the physical activity pleasure scale (PACES)32, 33, and the self-efficacy scale34.
- Have the participant answer the affection scale before the familiarization session.
- Have participant perform a warm-up of 5 min by walking on a treadmill at 3-4 km/h.
- Have the participant perform 3 sets of 8 repetitions in each of the 7 exercises, that they consider hard or between levels 5 and 7 according to the OMNI-RES scale.
- Allow 90 to 120 s recovery between sets and exercises.
- Have the participant answer the affection scale, the PACES, and the self-efficacy scale after the familiarization session.
- Familiarization session 3
- Have the participant answer the affection scale before the familiarization session.
- Have participant perform a warm-up of 5 min by walking on a treadmill at ~3-4 km/h.
- Have participant perform 3 sets of 8 repetitions in each of the 7 exercises, that they consider hard, or between levels 7 and 9 according to the OMNI-RES scale.
- Allow 90 to 120 s recovery between sets and exercises.
- Have the participant answer the affection scale, the PACES, and the self-efficacy scale after the familiarization session.
- Familiarization session 4
- Have the participant answer the affection scale before the familiarization session.
- Have participant perform a warm-up of 5 min by walking on a treadmill at 3-4 km/h.
- Have the participant perform 3 sets of 8 repetitions in each of the 7 exercises, that they consider hard and extremely hard or between levels 9 and 10 according to the OMNI-RES scale.
- Allow 90 to 120 s recovery between sets and exercises.
- Have the participant answer the affection scale, the PACES, and the self-efficacy scale after the familiarization session.
- Familiarization session 5
- Have participant mimic the strength tests (see details in step 5). The interval between familiarization sessions can be up to 7 days and can be scheduled based on the presence of delayed-onset muscle soreness.
NOTE: This last familiarization session is meant to familiarize participant with the strength tests. It is extremely important in this study to have a thorough familiarization period 1) to make sure participant can perform the sets at or very close to concentric failure18, and 2) to avoid the negative effect of muscle damage on insulin sensitivity35,36,37.
- Have participant mimic the strength tests (see details in step 5). The interval between familiarization sessions can be up to 7 days and can be scheduled based on the presence of delayed-onset muscle soreness.
- Familiarization session 1
5. Strength tests (8 repetition maximum)
- Perform the strength tests (8 repetition maximum [RM]) in each of the 7 exercises for a total of at least 72 h (but no longer than 7 days) after the last familiarization session.
- Have participant perform a warm-up for 5 min by walking on a treadmill at 3-4 km/h.
- Have participant perform 1 set with low-load (12 repetitions, 40%-50% of RM, 3-4 OMNI scale) before each exercise.
- Adjust weight to the anticipated load at which the participant will be able to perform 8, but not 9, repetitions with good form. Record this load.
- If participant is unable to perform 8 repetitions due to loss of good form, to concentric muscular failure, or if participant stops the set voluntarily due to fatigue, reduce the load by 5-10%, let them rest for 3-5 min, and have another attempt.
- If participant is able to perform 9 repetitions, increase the load by 5-10%, let them rest for 3-5 min, and have another attempt.
- Repeat steps 5.1.4 and 5.1.5 until participant is able to perform 8 but not 9 repetitions and record the load.
NOTE: To avoid inter-rater variability, the same research team member should conduct strength tests with all participants. - Perform the tests in the same order of the exercises described above 1 - hex bar squat; 2 - bench press; 3 - leg press; 4 - lat pulldown; 5 - leg extension; 6 - shoulder press; 7 - leg curl, allowing at least 3 min between exercises.
NOTE: Base the anticipated load on the performance of the fifth familiarization session, when the strength tests will be mimicked. To avoid excessive fatigue, it is optimal to find the 8 RM load in the first attempt of each exercise, but 2 attempts are acceptable.
6. Random session allocation
- Print random sequences with the numbers 1, 2, and 3 that represent high-volume, low-volume and control, respectively, and insert each one inside opaque, sequentially numbered envelope and seal the envelopes.
- Open the envelopes to reveal session order after the strength tests.
NOTE: Step 6.1 should be performed by a research collaborator not directly involved in data collection, or responsible for opening the envelopes.
7. Blind data collection
- Instruct participant not to disclose which session they performed the night before to the employees of the clinical lab where the OGTT will be performed.
- Have a research collaborator, not directly involved in data collection and analyzes, to double entry the data from the OGTT into separate spreadsheets using codes to hide participant's identity and session performed.
NOTE: Due to characteristics inherent to this study design (exercise), blinding both the participant and the therapist is not possible.
8. Exercise sessions
- Session 1 - high-volume
- Instruct participant not to engage in moderate-to-high intensity exercise for at least 2 days before the session.
- Instruct the participant to follow the diet prescribed by the nutritionist the day before and on the day of the session. The last meal is at 6:30 pm
- Instruct the participant to report to the gym at 7:30 pm.
- Have the participant answer the affection scale before the exercise session at 7:55 pm.
- Have participant perform a warm-up for 5 min by walking on a treadmill at 3-4 km/h.
- Have the participant perform 3 sets with as many repetitions as they can (until concentric muscular failure or until participant stops the set voluntarily) in each of the 7 exercises with the load from the 8 RM test and record the number of repetitions in each set.
- Ask the participant to rate their effort based on the OMNI-RES scale after each set and record. Allow 120 s recovery between sets and exercises.
- Have the participant answer the affection scale, the PACES, and the self-efficacy scale after they finish the session.
- Have the participant ingest the post-session meal between 9:00-9:30 pm and instruct participant not to ingest anything else (except for water) until the next morning when the OGTT will be performed and send them home.
- Session 2 - low-volume
- Repeat steps 8.1.1 to 8.1.3. Have participant answer the affection scale at 8:35 pm. Repeat step 8.1.5.
- Have participant perform 1 set with as many repetitions as they can (until concentric muscular failure or until participant stops the set voluntarily) in each of the 7 exercises with the load from the 8 RM test and record the number of repetitions in each set.
- Repeat steps 8.1.7 to 8.1.9
- Session 3 - control day
- Repeat procedures from 8.1.1 to 8.1.4.
- Position participant in each exercise/equipment for 30 s (mimicking the active set period) but instruct them not to perform any muscle contraction (or simply to remain still in the exercise/equipment).
- Repeat step 8.3.2 for 3 fake sets for all 7 exercises (mimicking session 1, but without any muscle contraction).
- Repeat steps 8.1.7 to 8.1.9
9. OGTT and data analysis
- Instruct participants to report to the lab between 7:00 and 7:30 am.
- Collect a fasting blood sample as described in38, 39.
- Have participants ingest 75 g of glucose in 300 mL solution. Collect blood samples at 30 min, 60 min, 90 min, and 120 min post glucose ingestion.
- Measure plasma glucose and serum insulin concentration in all blood samples as described in38, 39. Plot data from OGTT.
- Calculate a) the AUC for glucose and insulin following the trapezoidal rule40; b) the oral glucose insulin sensitivity index41; c) the Matsuda insulin sensitivity index42; d) the Cederholm's index43; e) the muscle insulin sensitivity index44, 45; f) the Gutt index46; g) the Avignon et al. index47; and h) the Stumvoll et al. index48.
10. Statistical analysis
- Analyze data blinded to allocation using coded spreadsheets from step 7.2.
- Express data as mean and standard deviation. Assess data normality with the Shapiro-Wilk test. For normally distributed data, analyze data with one-way analyzes of variance, if a significant main effect is observed, use post hoc Tukey test.
- For non-parametric data, use the Kruskal-Wallis's test or Friedman's test. Consider significance when p values are lower than 0.05.
- Reveal allocation after all statistics are run.
Results
Figure 2 shows representative (from a pilot study) responses for glucose (Figure 2A) and insulin (Figure 2B) during the OGTT. Usually, peaks for glucose and insulin values are observed at 30 min measurement, which is followed by a constant decrease until 120 min measurement. The lower the glucose peak, the better the result, which is indicative of inhibition of hepatic glucose production. The faster the decrease in glucose afte...
Discussion
This paper detailed the steps for a randomized controlled trial that aimed to assess the effects of strength exercise volume on insulin sensitivity in obese adults. Randomized controlled trials are the best research protocols to establish cause-and-effect of a treatment in an unbiased manner49,50. Specifically, in this study, we will employ a crossover design, which means every subject recruited will perform each condition in a randomized order5...
Disclosures
The authors have no conflicts of interest to declare.
Acknowledgements
This study is supported by the National Council for Scientific and Technological Development (CNPQ: Grant#407975/2018-7 and # 402091/2021-3) and by the Minas Gerais State Agency for Research and Development (FAPEMIG: Grant# APQ-00008-22). The funders played no role in this study's design, and do not play any roles in study conduct, interpretation of data, or reporting of results. This study is based at the Federal University of the Jequitinhonha and Mucuri Valleys (Diamantina-MG, Brazil) which provide the equipment and space (DXA, strength training room, strength training equipment, etc.) necessary for conducting the research.
Materials
| Name | Company | Catalog Number | Comments |
| dual-energy X-ray absorptiometry | GE | DXA, Lunar, iDXA Advanced | for assessing body composition |
| G*Power program | Heinrich-Heine-Universität Düsseldorf, Germany | version 3.1.9.6 | for calculating sample size |
References
- Ismail, A. D., et al. The effect of short-duration resistance training on insulin sensitivity and muscle adaptations in overweight men. Experimental physiology. 104 (4), 540-545 (2019).
- Jiahao, L., Jiajin, L., Yifan, L. Effects of resistance training on insulin sensitivity in the elderly: A meta-analysis of randomized controlled trials. Journal of Exercise Science and Fitness. 19 (4), 241-251 (2021).
- Liu, Y., et al. Resistance exercise intensity is correlated with attenuation of HbA1c and insulin in patients with type 2 diabetes: A systematic review and meta-analysis. International Journal of Environmental Research and Public Health. 16 (1), (2019).
- van Dijk, J. W., et al. Both resistance- and endurance-type exercise reduce the prevalence of hyperglycaemia in individuals with impaired glucose tolerance and in insulin-treated and non-insulin-treated type 2 diabetic patients. Diabetologia. 55 (5), 1273-1282 (2012).
- Koopman, R., et al. A single session of resistance exercise enhances insulin sensitivity for at least 24 in healthy men. European Journal of Applied Physiology. 94 (1-2), 180-187 (2005).
- Andersen, E., Høstmark, A. T. Effect of a Single Bout of Resistance Exercise on Postprandial Glucose and Insulin Response the Next Day in Healthy, Strength-Trained Men. The Journal of Strength and Conditioning Research. 21 (2), 487 (2007).
- Tong, T. K., Kong, Z., Shi, X., Shi, Q. Comparable Effects of Brief Resistance Exercise and Isotime Sprint Interval Exercise on Glucose Homeostasis in Men. Journal of Diabetes Research. 2017, (2017).
- Monroe, J. C., Naugle, K. M., Naugle, K. E. Effect of Acute Bouts of Volume-Matched High-Intensity Resistance Training Protocols on Blood Glucose Levels. Journal of Strength and Conditioning Research. 34 (2), 445-450 (2020).
- Bittel, A. J., et al. A Single Bout of Premeal Resistance Exercise Improves Postprandial Glucose Metabolism in Obese Men with Prediabetes. Medicine and science in sports and exercise. 53 (4), (2021).
- Fluckey, J. D., et al. Effects of resistance exercise on glucose tolerance in normal and glucose-intolerant subjects. Journal of Applied Physiology. 77 (3), 1087-1092 (1994).
- Fenicchia, L. M., et al. Influence of resistance exercise training on glucose control in women with type 2 diabetes. Metabolism: Clinical and Experimental. 53 (3), 284-289 (2004).
- Gordon, B. A., Fraser, S. F., Bird, S. R., Benson, A. C. Insulin sensitivity not modulated 24 to 78h after acute resistance exercise in type 2 diabetes patients. Diabetes, Obesity and Metabolism. 15 (5), 478-480 (2013).
- Gordon, B. A., Fraser, S. F., Bird, S. R., Benson, A. C. Insulin sensitivity in response to a single resistance exercise session in apparently healthy individuals. Journal of Endocrinological Investigation. 35 (7), 665-669 (2012).
- Malin, S. K., Hinnerichs, K. R., Echtenkamp, B. G., Evetovich, T. K., Engebretsen, B. J. Effect of adiposity on insulin action after acute and chronic resistance exercise in non-diabetic women. European Journal of Applied Physiology. 113 (12), 2933-2941 (2013).
- Moreno-Cabañas, A., et al. One Bout of Resistance Training Does Not Enhance Metformin Actions in Prediabetic and Diabetic Individuals. Medicine and Science in Sports and Exercise. 54 (7), (2022).
- Luebbers, P. E., et al. Glucose Uptake After Resistance Training of Different Intensities but of Equal Work Volume. Journal of Strength and Conditioning Research. 22 (4), 1094-1100 (2008).
- Chapman, J., Garvin, A. W., Ward, A., Cartee, G. D. Unaltered insulin sensitivity after resistance exercise bout by postmenopausal women. Medicine & Science in Sports & Exercise. 34 (6), 936-941 (2002).
- Brown, E. C., Franklin, B. A., Regensteiner, J. G., Stewart, K. J. Effects of single bout resistance exercise on glucose levels, insulin action, and cardiovascular risk in type 2 diabetes: A narrative review. Journal of Diabetes and its Complications. 34 (8), (2020).
- Ishiguro, H., et al. In Search of the Ideal Resistance Training Program to Improve Glycemic Control and its Indication for Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Sports Medicine. 46 (1), 67-77 (2016).
- Reed, M. E., Ben-Ezra, V., Biggerstaff, K. D., Nichols, D. L. The Effects of Two Bouts of High- and Low-Volume Resistance Exercise on Glucose Tolerance in Normoglycemic Women. Journal of Strength and Conditioning Research. 26 (1), 251-260 (2012).
- Black, L. E., Swan, P. D., Alvar, B. A. Effects of Intensity and Volume on Insulin Sensitivity During Acute Bouts of Resistance Training. Journal of Strength and Conditioning Research. 24 (4), 1109-1116 (2010).
- Carballo-Fazanes, A., et al. Physical Activity Habits and Determinants, Sedentary Behaviour and Lifestyle in University Students. International Journal of Environmental Research and Public Health. 17 (9), 3272 (2020).
- SCHOENFELD, B. J., et al. Resistance Training Volume Enhances Muscle Hypertrophy but Not Strength in Trained Men. Medicine & Science in Sports & Exercise. 51 (1), 94-103 (2019).
- Neupert, S. D., Lachman, M. E., Whitbourne, S. B. Exercise Self-Efficacy and Control Beliefs: Effects on Exercise Behavior after an Exercise Intervention for Older Adults. Journal of Aging and Physical Activity. 17 (1), 1-16 (2009).
- Gjestvang, C., Abrahamsen, F., Stensrud, T., Haakstad, L. A. H. Motives and barriers to initiation and sustained exercise adherence in a fitness club setting—A one-year follow-up study. Scandinavian Journal of Medicine and Science in Sports. 30 (9), (2020).
- Collado-Mateo, D., et al. Key factors associated with adherence to physical exercise in patients with chronic diseases and older adults: An umbrella review. International Journal of Environmental Research and Public Health. 18 (4), (2021).
- Riebe, D., et al. Updating ACSM’s Recommendations for Exercise Preparticipation Health Screening. Medicine & Science in Sports & Exercise. 47 (11), 2473-2479 (2015).
- Cortés, M. E., Alfaro, A. A. The effects of hormonal contraceptives on glycemic regulation. Linacre Quarterly. 81 (3), (2014).
- Kerksick, C. M., et al. ISSN exercise & sports nutrition review update: research & recommendations. , 1-57 (2018).
- ROBERTSON, R. J., et al. Concurrent Validation of the OMNI Perceived Exertion Scale for Resistance Exercise. Medicine & Science in Sports & Exercise. 35 (2), 333-341 (2003).
- Hardy, C. J., Rejeski, W. J. Not What, but How One Feels: The Measurement of Affect during Exercise. Journal of Sport and Exercise Psychology. 11 (3), (2016).
- Alves, E. D., Panissa, V. L. G., Barros, B. J., Franchini, E., Takito, M. Y. Translation, adaptation, and reproducibility of the Physical Activity Enjoyment Scale (PACES) and Feeling Scale to Brazilian Portuguese. Sport Sciences for Health. 15 (2), (2019).
- Kendzierski, D., DeCarlo, K. J. Physical Activity Enjoyment Scale: Two Validation Studies. Journal of Sport and Exercise Psychology. 13 (1), (2016).
- McAuley, E., Lox, C., Duncan, T. E. Long-term maintenance of exercise, self-efficacy, and physiological change in older adults. Journals of Gerontology. 48 (4), (1993).
- Chen, T. C., et al. Changes in Insulin Sensitivity and Lipid Profile Markers Following Initial and Secondary Bouts of Multiple Eccentric Exercises. Frontiers in Physiology. 13, (2022).
- Jimenez, C., Santiago, M., Sitler, M., Boden, G., Homko, C. Insulin-Sensitivity Response to a Single Bout of Resistive Exercise in Type 1 Diabetes Mellitus. Journal of Sport Rehabilitation. 18 (4), 564-571 (2009).
- Gonzalez, J. T., Barwood, M. J., Goodall, S., Thomas, K., Howatson, G. Alterations in whole-body insulin sensitivity resulting from repeated eccentric exercise of a single muscle group: A pilot investigation. International Journal of Sport Nutrition and Exercise Metabolism. 25 (4), 405-410 (2015).
- Ismail, A. D., et al. The effect of short-duration resistance training on insulin sensitivity and muscle adaptations in overweight men. Experimental Physiology. 104 (4), 540-545 (2019).
- de Matos, M. A., et al. High-Intensity Interval Training Improves Markers of Oxidative Metabolism in Skeletal Muscle of Individuals With Obesity and Insulin Resistance. Frontiers in Physiology. 9 (OCT), (2018).
- . Carbohydrate Homeostasis. New England Journal of Medicine. 283 (5), 237-246 (1970).
- Mari, A., Pacini, G., Murphy, E., Ludvik, B., Nolan, J. J. A Model-Based Method for Assessing Insulin Sensitivity From the Oral Glucose Tolerance Test. Diabetes Care. 24 (3), 539-548 (2001).
- Matsuda, M., DeFronzo, R. A. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 22 (9), 1462-1470 (1999).
- Cederholm, J., Wibell, L. Insulin release and peripheral sensitivity at the oral glucose tolerance test. Diabetes Research and Clinical Practice. 10 (2), 167-175 (1990).
- Abdul-Ghani, M. A., Matsuda, M., Balas, B., DeFronzo, R. A. Muscle and Liver Insulin Resistance Indexes Derived From the Oral Glucose Tolerance Test. Diabetes Care. 30 (1), 89-94 (2007).
- de Matos, M. A., et al. High-Intensity Interval Training Improves Markers of Oxidative Metabolism in Skeletal Muscle of Individuals With Obesity and Insulin Resistance. Frontiers in Physiology. 9, (2018).
- Gutt, M., et al. Validation of the insulin sensitivity index (ISI0,120): comparison with other measures. Diabetes Research and Clinical Practice. 47 (3), 177-184 (2000).
- Avignon, A., Bœgner, C., Mariano-Goulart, D., Colette, C., Monnier, L. Assessment of insulin sensitivity from plasma insulin and glucose in the fasting or post oral glucose-load state. International Journal of Obesity. 23 (5), (1999).
- Stumvoll, M., van Haeften, T., Fritsche, A., Gerich, J. Oral Glucose Tolerance Test Indexes for Insulin Sensitivity and Secretion Based on Various Availabilities of Sampling Times. Diabetes Care. 24 (4), 796-797 (2001).
- Kendall, J. M. Designing a research project: Randomised controlled trials and their principles. Emergency Medicine Journal. 20 (2), (2003).
- Moher, D., et al. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ (Clinical research ed.). 340, (2010).
- Sibbald, B., Roberts, C. Understanding controlled trials Crossover trials. BMJ. 316 (7146), (1998).
- Lim, C. Y., In, J. Considerations for crossover design in clinical study. Korean Journal of Anesthesiology. 74 (4), (2021).
- Taylor, H. L., et al. Post-exercise carbohydrate-energy replacement attenuates insulin sensitivity and glucose tolerance the following morning in healthy adults. Nutrients. 10 (2), (2018).
- Johnson-Bonson, D. A., et al. Interactive effects of acute exercise and carbohydrate-energy replacement on insulin sensitivity in healthy adults. Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme. 46 (10), (2021).
- Patarrão, R. S., Wayne Lautt, W., Paula Macedo, M. Assessment of methods and indexes of insulin sensitivity. Revista Portuguesa de Endocrinologia, Diabetes e Metabolismo. 9 (1), 65-73 (2014).
- DeFronzo, R. A., Tobin, J. D., Andres, R. Glucose clamp technique: A method for quantifying insulin secretion and resistance. American Journal of Physiology Endocrinology Metabolism and Gastrointestinal Physiology. 6 (3), (1979).
- Monzillo, L. U., Hamdy, O. Evaluation of Insulin Sensitivity in Clinical Practice and in Research Settings. Nutrition Reviews. 61 (12), 397-412 (2003).
- Radziuk, J. Insulin Sensitivity and Its Measurement: Structural Commonalities among the Methods 1. The Journal of Clinical Endocrinology & Metabolism. 85 (12), (2000).
- Gordon, B. A., Fraser, S. F., Bird, S. R., Benson, A. C. Reproducibility of multiple repeated oral glucose tolerance tests. Diabetes Research and Clinical Practice. 94 (3), (2011).
- Beaudry, K. M., Surdi, J. C., Mari, A., Devries, M. C. Exercise mode influences post-exercise glucose sensitivity and insulin clearance in young, healthy males and females in a sex-dependent manner: A randomized control trial. Physiological Reports. 10 (13), (2022).
- Aguiar, S. d. a. S., et al. Acute metabolic responses following different resistance exercise protocols. Applied Physiology, Nutrition and Metabolism. 43 (8), 838-843 (2018).
- Venables, M. C., Shaw, C. S., Jeukendrup, A. E., Wagenmakers, A. J. M. Effect of acute exercise on glucose tolerance following post-exercise feeding. European Journal of Applied Physiology. 100 (6), 711-717 (2007).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved