Method Article
Transconjunctival Approach for Injection into the Rat Optic Nerve
In This Article
Summary
This protocol describes a unique technique using a transconjunctival approach to access the rat optic nerve for injections.
Abstract
The rat serves as an important model for studying disorders of the optic nerve, including heritable, traumatic, neoplastic, and autoimmune conditions. Accessing the rat optic nerve for experimental manipulations and injections is challenging due to the small size of the orbit and the vascularity of the surrounding orbital tissues. Previous techniques have involved a cutaneous incision, which carries a higher risk of infection and requires wound closure. This study aims to describe a unique, less invasive, and potentially more efficient approach to accessing the rat optic nerve. A transconjunctival incision, along with a rat spinal cord hook, is used to isolate the optic nerve for experimental injections. In these experiments, an adenoviral vector is injected into the optic nerve with the goal of using this technique for direct gene therapy delivery in the future. This technique can also be applied by investigators requiring access to the rat optic nerve for other experimental studies, including drug delivery and optic nerve injury research.
Introduction
The rat is a key experimental model for the study of disorders of the optic nerve, including glaucoma, traumatic optic neuropathy, hereditary optic neuropathy, demyelinating disease, and tumors of the optic nerve,2,3,4,5,6,7. However, obtaining access to the optic nerve for experimental injection and manipulations can be difficult, given the small size of the orbit and the vascular orbital tissues.
The two prior techniques that have been described to access the rat optic nerve for experimental manipulations utilize either a superior orbital or lateral transcutaneous approach to access the orbit8,9. Raykova et al. describe the injection of dyed beads into the optic nerve using a superior orbital skin incision8. While their technique was effective, it requires hemostasis and the need for a traction suture for exposure, which can be more challenging with this approach8. Another group similarly described an approach using a lateral skin incision to access the orbit. They were successfully able to inject the optic nerve using this approach, but required hemostasis and wound closure9. While these techniques are effective approaches to accessing the optic nerve for experimental study, they both involve a cutaneous incision, need for hemostasis, and wound closure. These steps have associated risks for infection and potentially increased time under anesthesia due to the need for wound closure8,9,10.
Given the limitations of the techniques previously described, the goal of this study is to describe a more efficient and less traumatic approach to accessing the nerve for experimental study. A spinal cord hook is used to isolate the optic nerve, analogous to the surgical technique used in humans to isolate muscle and ocular adnexal structures using a muscle hook. This technique can be used by investigators who need access to the rat optic nerve for many indications, from drug delivery to gene therapy.
This article describes the novel use of a transconjunctival approach to access the rat orbit and optic nerve for experimental study with the aid of a spinal cord hook.
Protocol
All animal procedures were approved by the Office of the Institutional Animal Care and Use Committee at the University of Iowa. This study was funded by the Gilbert Family Foundation (grant number 913012). A combination of male and female Sprague Dawley rats, aged 4-12 weeks and weighing 100-400 g, were used for these experiments. The details of the reagents and equipment used are listed in the Table of Materials.
1. Anesthetization
- Anesthetize rats using inhalation of isoflurane gas. Place the animal into the induction chamber and turn the vaporizer to 4%-5% for induction of anesthesia and 2%-3% to maintain anesthesia.
- Verify the depth of anesthesia by assessing the absence of the pedal withdrawal reflex before proceeding.
- Apply artificial tear gel to the ocular surface.
2. Positioning
- Once anesthesia is induced, place the animal on a heating pad in a lateral recumbent position. Use a nosecone to maintain anesthesia.
- Position the orbit beneath the operating microscope.
3. Surgical exposure
- Grasp the conjunctiva with toothed forceps in the inferotemporal quadrant (Figure 1).
- Use Westcott scissors to create a radial conjunctival incision and then open Tenon's fascia in the inferonasal quadrant (Figure 1). Use blunt dissection to remove overlying orbital tissue.
4. Isolation of the optic nerve
- Rotate the eye nasally using toothed forceps and have the assistant pass a spinal cord hook into the inferotemporal quadrant to isolate the optic nerve (Figure 1). Take care to avoid the ventral ophthalmic vein and the extraocular muscles.
- Remove overlying orbital tissue and Tenon's fascia using blunt dissection with a cotton-tipped applicator or forceps to expose the optic nerve and sheath.
5. Injection
- Use a 33 G needle connected to a 10 uL or a 1 mL syringe to pierce the optic nerve sheath (Figure 1).
- Inject different concentrations of adeno-associated virus-eGFP or India Ink dye into the optic nerve under direct visualization to ensure that the material is injected directly into the nerve.
- To assess AAV-eGFP, inject a 10 μL of suspension containing AAV viral particles (2 x 1013 vector genomes (vg)/mL for a total dose of 2 x 1011 vg).
NOTE: Investigators may inject other dyes or stains depending on the experimental needs.
6. Post-procedural steps
- Reapproximate the conjunctiva to the limbus to cover the incision site and apply antibiotic ointment over the conjunctiva (Figure 1).
- Allow the animals to recover from anesthesia and monitor them for any signs of infection.
7. Staining
- Seven days after injection, sacrifice the animals via CO2 asphyxiation followed by cervical dislocation11 following ethical guidelines. Dissect the optic nerve.
- Fix the optic nerves in 4% paraformaldehyde and rinse in increased concentrations of sucrose. Embed cryomolds in a 2:1 solution of OCT compound to 20% sucrose. Flash freeze samples in liquid nitrogen, store at -80 °C and section at a thickness of 7 μm.
- Block cryosections of the optic nerve for 30 min at room temperature in immunocytochemical (IHC) buffer and 3% bovine serum albumin. Incubate sections with rat anti-GFP primary antibody diluted in IHC buffer for 2 h at room temperature.
- Incubate sections with goat anti-rat in IHC buffer for an additional 1 h at room temperature. During secondary antibody incubation, label sections with 4',6-Diamidino-2-phenylindole dihydrochloride (DAPI) to visualize cell nuclei. Rinse sections thoroughly with PBS and mount using a mounting medium.
8. Imaging
- Image the labeled sections using an upright confocal microscope system.
- Specifically, use a 405 nm violet laser to visualize DAPI labeling and a 488 nm cyan laser to visualize GFP.
NOTE: Settings for each laser (i.e., laser power, intensity/gain, background/noise filters) should be optimized by each user for different confocal microscope systems. Acquire images using a 20x objective lens.
Results
Representative experimental results using this technique for rat optic nerve injection are shown in Figure 2. In this example, adeno-associated virus-eGFP (AAV-eGFP) was injected into the rat optic nerve using the transconjunctival technique described above with a 33 G needle connected to a 10 µL syringe. These results were part of an experiment using this technique to study optic nerve injection as part of an ultimate aim to use AAV to deliver CRISPR correction to optic nerves in a rat model of neurofibromatosis 1 (NF1). A combination of dye and AAV vector was used to develop the injection technique. Animals were sacrificed seven days after injection into the optic nerve. Nerves were then dissected and labeled with anti-GFP antibodies to allow for imaging of the optic nerve with confocal microscopy to confirm that the material had successfully penetrated the optic nerve sheath and entered the optic nerve. The successful introduction of material into the optic nerve was confirmed by antibody staining, and it appeared green on confocal microscopy, as seen in Figure 2A. In cases where penetration through the optic nerve sheath was not complete, or there was efflux of material from the needle track, staining was seen along the edge of the optic nerve sheath rather than within the nerve tissue, as seen in Figure 2C.
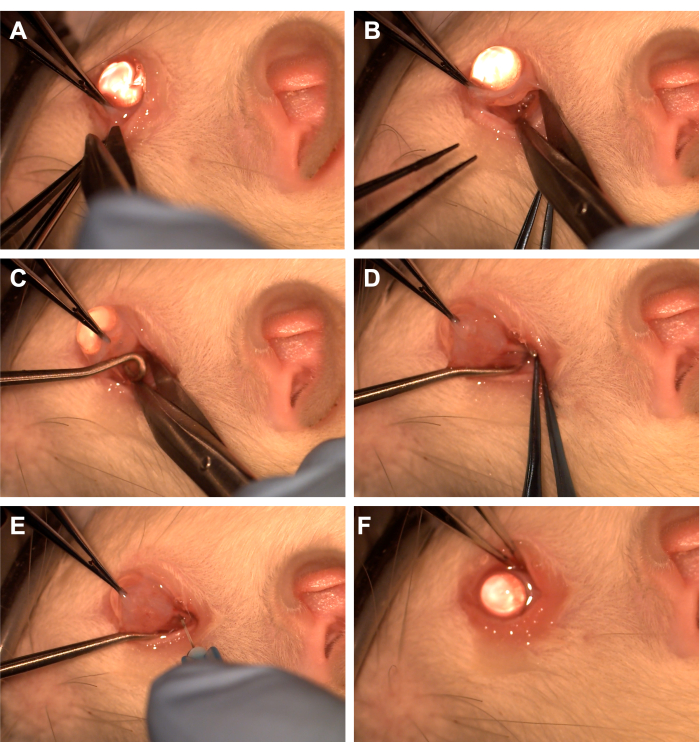
Figure 1: Sequence of the surgical steps. (A) The conjunctiva is grasped with toothed forceps, and Westcott scissors are used to create a limbal peritomy. (B) After the tenon's fascia is opened and bluntly dissected, the orbit is exposed with the assistance of Westcott scissors. (C) A spinal cord hook is inserted and used to isolate the optic nerve. (D) After blunt dissection of overlying orbital tissue, the optic nerve is exposed using the spinal cord hook. (E) A 33 G needle is used to pierce the optic nerve sheath and inject adenovirus or dye into the optic nerve. (F) The globe is reposited into the orbit, and the conjunctiva is re-positioned over the peritomy site. Please click here to view a larger version of this figure.
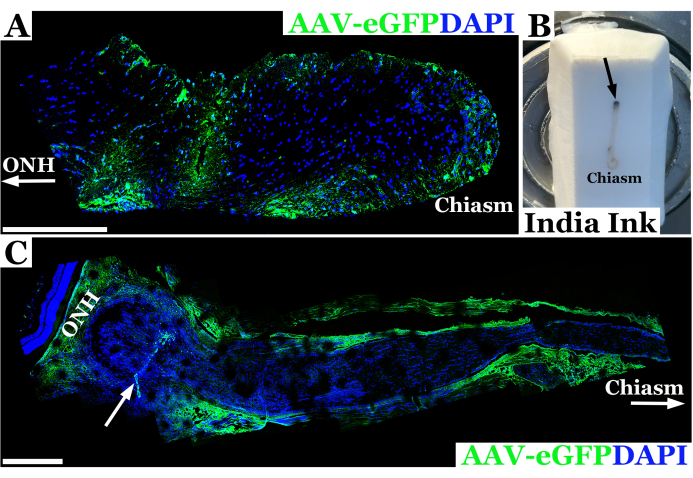
Figure 2: Representative fluorescent confocal panoramic image of rat optic nerve 7 days post successful injection with AAV-eGFP (green) in which vector transduced neurons near the optic chiasm. (A) Optic nerve nuclei are labeled with DAPI (blue). ONH with arrow denotes the optic nerve head or orbital end of the nerve, whereas the optic chiasm end is labeled as "chiasm". (B) Photograph of a rat optic nerve from orbit to chiasm embedded in frozen cryosection block. The nerve is injected with India Ink immediately posterior to the orbit (injection site denoted by black arrow) in order to orient the optic nerve. (C) Panoramic fluorescent image of rat optic nerve 7-days post unsuccessful injection with AAV-eGFP (green) in which most of the vector suspension likely effluxed out of nerve tract, transducing the optic sheath. Optic nerve nuclei are labeled with DAPI (blue). ONH denotes the Optic Nerve Head immediately posterior to the neural retina and injection site indicated by a white arrow. Chiasm and white arrow demarcate towards the optic chiasm. Scale bars = 400 μm in (A) and 200 μm in (C). Please click here to view a larger version of this figure.
| Variable | Approach | ||
| Transconjunctival | Superior | Lateral | |
| Skin incision | No | Yes | Yes |
| Conjunctival incision | Yes | No | No |
| Microscope | Yes | Yes | Yes |
| Transection of superior orbital muscle | No | Yes | No |
| Traction suture | No | Yes | Yes |
| Spinal cord hook | Yes | No | No |
| Skin suture | No | Yes | Yes |
Table 1: Comparison to previously published techniques for isolating the rat optic nerve for experimental study. Variables, including the need for skin incision, conjunctival incision, operating microscope, transection of the superior orbital muscle, traction suture, spinal cord hook, and skin suture, are compared using the transconjunctival, superior, and lateral approaches.
Discussion
This article describes a novel method for accessing the rat optic nerve for experimental study without the need for a skin incision. This approach provides the potential advantages of efficiency and decreased infection and bleeding risk, given the lack of a need for skin incision and fewer steps compared to other techniques8,9. This technique also uses the novel approach of isolating the optic nerve with a rat spinal cord hook to avoid trauma to the nerve and surrounding tissues.
Critical steps in the technique include making an adequate limbal incision for access to the sub-conjunctival space, use of the spinal cord hook to isolate the optic nerve, and use of a small-bore needle to pierce the optic nerve sheath and deliver material to the optic nerve. Care must be taken to rotate the spinal cord hook to isolate the nerve. When the nerve is not clearly isolated, the hook may need to be introduced several times in order to ensure that the nerve is isolated, and a cotton-tipped applicator may be used to remove connective tissue from the nerve sheath for better visualization. Exposure to this technique can be more challenging in larger-sized animals with a greater volume of orbital fat. The use of an assistant to retract the surrounding tissues for adequate exposure can be useful in these situations. Care must be taken to visualize the optic nerve directly during injection. Ensuring a perpendicular orientation of the needle through the nerve sheath during injection improves injection success.
This technique builds on prior work by Raykova et al. and Hallas and Wells8,9. Raykova et al. described access to the rat optic nerve using a superior approach with a skin incision created over the orbital ridge and transection of the superior orbital muscle8. While this approach allows for exposure of the nerve, it requires transection of an extraocular muscle with a higher risk for bleeding and the need for suturing of the skin incision. Similarly, Hallas and Wells describe a lateral approach to the rat orbit to reach the optic nerve9. They describe a lateral skin incision followed by a lateral conjunctival incision and a traction suture used to rotate the eye and expose the optic nerve9. Similar to the superior approach, this technique involves a skin incision with a resultant need for wound closure and a higher risk for infection and bleeding8,9. The transconjunctival technique presented here obviates the need for a skin and muscle incision, therefore reducing the risk of bleeding/infection and improving efficiency by avoiding the need to close a skin incision. The procedure causes minimal distress, is well tolerated, and does not require long-term use of analgesic or anti-inflammatory medications. The transconjunctival technique is compared and contrasted with the previously published techniques in Table 1.
Limitations of the technique presented in this study include a learning curve for isolating the optic nerve using the spinal cord hook, but once mastered, this technique allows for efficient and atraumatic isolation of the nerve. Familiarity with the use of an operating microscope is necessary for this technique. While there is a learning curve for this technique, the smaller number of steps and absence of the need for suturing of the wound will make this technique easier to master relative to the other published approaches8,9. Careful blunt dissection to expose the nerve and sheath is imperative, as there can be hemorrhage from the nearby vessels and muscle tissue. In larger rats, it can be more difficult to expose large sections of the optic nerve due to the increased orbital fat/tissue, but with care, this tissue can generally be removed with blunt dissection to improve visualization of the nerve. Further work could address the use of this technique in other model systems, such as the mouse, and could directly compare surgical efficiency to other techniques for optic nerve access.
This technique can be used by other groups who need to access the rat optic nerve for experimental study. The experiments presented here were part of a larger project aiming to develop ways to deliver CRISPR gene correction to the optic nerve in rat models of neurofibromatosis type 1 (NF1). The ultimate aim of this project is to find ways to deliver gene therapy directly to the optic nerve in patients with NF1 to prevent the development of optic nerve gliomas, which are vision-threatening12. In this study, India Ink dye or AAV-eGFP vector was injected into the optic nerve to ensure that material was being delivered into the optic nerve, and to demonstrate that the vector could penetrate the optic nerve.
This technique could be more broadly used by other investigators as the nerve may need to be accessed or novel therapeutics injected in an effort to treat a variety of vision-threatening conditions, including neoplasms of the optic nerve, degenerative processes such as Leber hereditary optic neuropathy, traumatic optic neuropathy, glaucoma, and autoimmune diseases such as optic neuritis13,14. For example, Singh et al. describe injection of C6 glioma cells into the rat optic nerve as a model of neurofibromatosis type 1 accessing the nerve using a cutaneous incision3, Kwon et al. accessed the optic nerve from a lateral cutaneous approach for studies involving optic nerve compression and regeneration with stem cells6, and Negishi et al. accessed the rat optic nerve for Schwann cell graft placement as a model of optic nerve regeneration15. The technique presented here could provide a more efficient and safer approach for studies such as these that require access to the rat optic nerve to answer a variety of experimental questions.
In summary, this work demonstrates efficacy in entering the optic nerve with a small-gauge needle approach and the ability to penetrate the nerve sheath without the need for a cutaneous incision. The representative images show that the adenoviral vector successfully penetrated the optic nerve. This technique has applications to a variety of optic nerve experiments in regenerative medicine, oncology, and inherited retinal diseases.
Disclosures
No relevant disclosures.
Acknowledgements
The authors would like to acknowledge the Gilbert Family Foundation for funding this project.
Materials
| Name | Company | Catalog Number | Comments |
| 4’,6-Diamidino-2-phenylindole dihydrochloride (DAPI) | MilliporeSigma | D9542 | 1:10,000 dilution, used for staining cryosections |
| AAV viral particles | Vectorbuilder | N/A | Vector used for optic nerve injection |
| Aqua-Mount Mounting Medium | ThermoFisher Scientific | 14-390-5 | Used for mounting stained cryosections |
| Bovine serum albumin | Research Products International Corp | 9048-46-8 | Used in process for staining cryosections |
| Goat anti-rat Alexa Fluor 488 | Thermo Fisher Scientific | A-11006 | 1:1000 dilution, used for staining cryosections |
| Isoflorane gas | Primal Healthcare | 66794-017-10 | Gas for anesthesia |
| Leica TCS SPE upright confocal microscope system | Leica Microsystems | N/A | Confocal microscope |
| Luer lock syringe | BD | 309628 | Syringe for injection |
| Microm HM505E cryostat | Microm | M-HM360 fom Marshall Scientific | Used for specimen sectioning |
| Rat anti-GFP | BioLegend | 338002 | 1:500 dilution, used for staining cryosections |
| Spinal cord hook | F.S.T | 10162-12 | Hook used to isolate optic nerve |
| Ten microliter syringe | Hamilton | CAL 803901 | Syringe for injection |
| Thirty-three-guage needle | JBP | JBP3308B | Needle for injection |
| Tissue-Tek OCT compound | VWR International | 25608-930 | Used for cryomold processing |
| Toothed forceps | Bausch and Lomb | E1809 | Forceps to grasp conjunctiva |
| Westcott curved tenotomy scissors | Bausch and Lomb | E3323-R | Scissors to create peritomy |
References
- Korn, P. et al. Evaluation of the neuroprotective effects of methylprednisolone and surgical decompression in a rodent model of traumatic optic neuropathy. Curr Eye Res. 47 (3), 461-467 (2022).
- Lim, J. Y., Utzschneider, D. A., Sakatani, K., Kocsis, J. D. The attenuation of GABA sensitivity in the maturing myelin-deficient rat optic nerve. Brain Res Dev Brain Res. 72 (1), 15-20 (1993).
- Singh, D., Dromel, P. C., Perepelkina, T., Baranov, P., Young, M. C6 cell injection into the optic nerve of long-evans rats: A short-term model of optic pathway gliomas. Cell Transplant. 29, 963689720964383 (2020).
- Schwaner, S. A., Feola, A. J., Ethier, C. R. Factors affecting optic nerve head biomechanics in a rat model of glaucoma. J R Soc Interface. 17 (165), 20190695 (2020).
- Ellouze, S. et al. Optimized allotopic expression of the human mitochondrial ND4 prevents blindness in a rat model of mitochondrial dysfunction. Am J Hum Genet. 83 (3), 373-387 (2008).
- Kwon, H., Park, M., Nepali, S., Lew, H. Hypoxia-preconditioned placenta-derived mesenchymal stem cells rescue optic nerve axons via differential roles of vascular endothelial growth factor in an optic nerve compression animal model. Mol Neurobiol. 57 (8), 3362-3375 (2020).
- Pang, I. H., Clark, A. F. Rodent models for glaucoma retinopathy and optic neuropathy. J Glaucoma. 16 (5), 483-505 (2007).
- Raykova, K., Jones, M. V., Huang, H., Hoffman, P. F., Levy, M. Minimally-invasive Technique for Injection into Rat Optic Nerve. J Vis Exp. 99, e52249 (2015).
- Hallas, B.H., Wells, M.R. A novel technique for mulitple injections into the mamallian optic nerve. Kopf Carrier. 54, 1-10 (2000).
- Yan, F. et al. Partial optic nerve transection in rats: A model established with a new operative approach to assess secondary degeneration of retinal ganglion cells. J Vis Exp. 128, e56272 (2017).
- Han I. H. et al. Biocompatibility of human induced pluripotent stem cell-derived retinal progenitor cell grafts in immunocompromised rats. Cell Transplantation. 31, 1-25 (2022).
- Tang, Y., Gutmann, D. H. Neurofibromatosis Type 1-associated optic pathway gliomas: Current challenges and future prospects. Cancer Manag Res. 15, 667-681 (2023).
- Pan, T. et al. Implantation of biomimetic polydopamine nanocomposite scaffold promotes optic nerve regeneration through modulating inhibitory microenvironment. J Nanobiotechnol. 22 (1), 683 (2024).
- Gokoffski, K. K., Peng, M., Alas, B., Lam, P. Neuro-protection and neuro-regeneration of the optic nerve: recent advances and future directions. Curr Opin Neurol. 33 (1), 93-105 (2020).
- Negishi, H., Dezawa, M., Oshitari, T., Adachi-Usami, E. Optic nerve regeneration within artificial Schwann cell graft in the adult rat. Brain Res Bull. 55 (3), 409-419 (2001).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved