A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Preparation of Contiguous Bisaziridines for Regioselective Ring-Opening Reactions
In This Article
Summary
Contiguous bisaziridines containing non-activated and activated aziridines were synthesized by asymmetric organocatalytic aziridinations and then subjected to chemoselective ring-opening reactions under acidic or basic conditions. The non-activated aziridine ring opens with less reactive nucleophiles under acidic conditions, whereas the activated aziridine ring opens with more reactive nucleophiles under basic conditions.
Abstract
Aziridines, a class of reactive organic molecules containing a three-membered ring, are important synthons for the synthesis of a large variety of functionalized nitrogen-containing target compounds through the regiocontrolled ring-opening of C-substituted aziridines. Despite the tremendous progress in aziridine synthesis over the past decade, accessing contiguous bisaziridines efficiently remains difficult. Therefore, we were interested in synthesizing contiguous bisaziridines bearing an electronically diverse set of N-substituents beyond the single aziridine backbone for regioselective ring-opening reactions with diverse nucleophiles. In this study, chiral contiguous bisaziridines were prepared by organocatalytic asymmetric aziridination of chiral (E)-3-((S)-1-((R)-1-phenylethyl)aziridin-2-yl)acrylaldehyde with N-Ts-O-tosyl or N-Boc-O-tosyl hydroxylamine as the nitrogen source in the presence of (2S)-[diphenyl(trimethylsilyloxy)methyl]pyrrolidine as a chiral organocatalyst. Also demonstrated here are representative examples of regioselective ring-opening reactions of contiguous bisaziridines with a variety of nucleophiles such as sulfur, nitrogen, carbon, and oxygen, and the application of contiguous bisaziridines to the synthesis of multi-substituted chiral pyrrolidines by Pd-catalyzed hydrogenation.
Introduction
Rational design of small organic molecules with diverse reactive sites that precisely control product selectivity is a key goal in modern organic synthesis and green chemistry1,2,3,4,5,6,7,8. To achieve this goal, we were interested in the modular synthesis of aziridines. Aziridines are of interest to most organic chemists, owing to their structurally important framework9 with an electronically diverse set of N-substituents that can lead to regioselective ring-opening reactions with multiple nucleophiles10,11,12,13,14,15,16,17,18,19, and varied pharmacological activities such as antitumor, antimicrobial, and antibacterial properties. Despite the advances in aziridine chemistry, non-activated aziridine and activated aziridine have independent syntheses and ring-opening reactions in the literature20.
Therefore, we aimed to synthesize contiguous bisaziridines comprising both the non-activated and activated aziridines. These contiguous bisaziridines can be used to systematically rationalize a chemoselective ring-opening pattern based on the following electronic properties of the two different aziridines and their reactivity to nucleophiles20,21,22,23,24: a) activated aziridines, in which the electron-withdrawing substituents conjugatively stabilize the negative charge on the nitrogen, readily react with multiple nucleophiles to allow ring-opened products; b) non-activated aziridines, in which the nitrogen is bound to the electron-donating substituents, are considerably inert toward nucleophiles; hence, a pre-activation step with a suitable activator (mainly Brønsted or Lewis acids) is required to afford the ring-opened products in high yields20,21,25,26.
The present study describes the rational design of contiguous bisaziridines as chiral building blocks via transition-metal-free organocatalysis and the synthesis of diverse nitrogen-rich molecules utilizing predictive modeling tools for ring-opening reactions of bisaziridines. This study aims to stimulate the advancement of practical methods for the construction of nitrogen-enriched bioactive compounds and natural products and the polymerization of aziridines.
Access restricted. Please log in or start a trial to view this content.
Protocol
The details of all the synthesized products (1-5), including the structure, full NMR spectra, optical purity, and HRMS-MALDI data, are provided in Supplementary File 1.
1. Synthesis of 3-(aziridin-2-yl)acryl aldehyde (1a)
- Flame dry a 50 mL round-bottomed flask equipped with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous toluene (19 mL) and (R)-1-((R)-1-phenylethyl)aziridine-2-carbaldehyde (1.00 g, 5.71 mmol) (see Table of Materials) to the flask. Then, stir the solution for 1 min.
- Add (triphenylphosphoranylidene)acetaldehyde (2.08 g, 6.85 mmol) (see Table of Materials) to the stirred solution.
- Heat the reaction mixture at 60 °C for 18 h. Then, cool the reaction mixture to room temperature and remove volatile solvent from the reaction mixture under reduced pressure.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (1:6 v/v, Rf = 0.25) as an eluent.
- Purify the crude product by silica gel flash chromatography with ethyl acetate:hexanes (1:6 v/v) as the eluent to isolate pure product 1a (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements (see steps 8 and 9 for measurement methods).
2. Synthesis of contiguous bisaziridine (2a)
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous ethyl acetate (3 mL) and 1a (step 1.6, 201 mg, 1.0 mmol) to the flask, and then stir the solution for 1 min.
- Add catalyst (S)-2-(diphenyl((trimethylsilyl)oxy)methyl)pyrrolidine (BS, see Table of Materials) (0.02 mL, 7 mol%) to the mixture and stir at room temperature for 30 min.
- Add 316 mg, 1.10 mmol of tert-butyl tosyloxycarbamate (BocNHOTs, see Table of Materials) and 123 mg, 1.50 mmol of sodium acetate to the reaction mixture and stir for 24 h.
- Monitor the reaction progress by TLC using diethyl ether:hexanes (1:4 v/v, Rf = 0.27) as an eluent.
- Extract the reaction mixture with ethyl acetate (3 x 50 mL) in a separatory funnel.
- Dry the combined organic layer over anhydrous Na2SO4, filter, and concentrate in vacuo.
- Purify the resulting crude product by flash chromatography on silica gel with diethyl ether:hexanes (1:4 v/v) as eluent to isolate pure product 2a (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements (see steps 8 and 9).
3. Synthesis of compound 3
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous methanol (11 mL) and aldehyde 2a (step 2.8, 1.00 g, 3.16 mmol) [or 2b (1.17 g, 3.16 mmol, see Supplementary File 1)] to the flask, and then stir the solution for a minute.
- Add NaBH4 (95 mg, 2.53 mmol) to the stirred solution.
- Stir the reaction mixture at 0 °C for 1 h.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (1:4 v/v, Rf = 0.27) as an eluent.
- After 1 h, quench the reaction mixture with distilled water and extract with ethyl acetate (3 x 50 mL) in a separatory funnel.
- Dry the combined organic layer over anhydrous Na2SO4, filter, and concentrate in vacuo.
- Purify the crude residue by silica gel flash chromatography with ethyl acetate:hexanes (1:4 v/v) as the eluent to isolate pure products 3a [or 3b] (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements (see steps 8 and 9).
4. Synthesis of compound 4
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous dichloromethane (11 mL) and alcohol 3a (step 3.8, 1.00 g, 3.14 mmol) [or 3b (1.17 g, 3.14 mmol)] to the flask, and then stir the solution for a minute.
- Add tert-butyldimethylsilyl chloride (TBSCl, 520 mg, 3.45 mmol) and imidazole (427 mg, 6.28 mmol) (see Table of Materials) to the stirred solution.
- Stir the reaction mixture at 0 °C for 18 h.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (1:4 v/v, Rf = 0.26) as an eluent.
- After 18 h, quench the reaction mixture with deionized water and extract with methylene chloride (3 x 50 mL) in a separatory funnel.
- Dry the combined organic layer over the anhydrous sodium sulfate, filter, and then concentrate under reduced pressure.
- Purify the crude residue by silica gel flash chromatography with ethyl acetate:hexanes (1:4 v/v) as the eluent to isolate pure products 4a [or 4b] (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements.
5. Selective ring-opening of non-activated aziridines: Synthesis of 5d
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add 3b (step 4.2, 100 mg, 0.27 mmol) and acetic acid (0.12 mL, 2.14 mmol) to the flask, and then stir the mixture at room temperature for 5 h.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (2:3 v/v, Rf = 0.28) as an eluent.
- After 5 h, remove the acetic acid in vacuo.
- Purify the crude residue by silica gel flash chromatography with ethyl acetate:hexanes (2:3 v/v) as the eluent to isolate pure product 5d (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements.
6. Selective ring-opening of activated aziridines: Synthesis of 5f
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous methanol (8 mL) and 4b (step 4.8, 100 mg, 0.21 mmol) to the flask, and then stir the solution for 1 min.
- Add NaN3 (39 mg, 0.6 mmol) and NH4Cl (21 mg, 0.41 mmol) in H2O (1 mL) to the above solution.
- Stir the reaction mixture at 0 °C for 4 h.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (1:4 v/v, Rf = 0.30) as an eluent.
- After 4 h, quench the reaction mixture with H2O and extract with ethyl acetate (3 x 50 mL) in a separatory funnel.
- Dry the combined organic layer over anhydrous Na2SO4, filter, and concentrate in vacuo.
- Purify the crude residue by silica gel flash chromatography with ethyl acetate:hexanes (1:4 v/v) as the eluent to isolate pure product 5f (see Supplementary File 1) as a yellow liquid.
- Confirm the product by NMR and polarimeter measurements.
7. Pd-catalyzed hydrogenation of contiguous aziridines: Synthesis of 5h
- Flame dry a 50 mL round-bottomed flask with a stirrer bar and a septum under vacuum conditions. Cool it to room temperature while filling it with argon gas.
- Add anhydrous methanol (5 mL), 2b (step 3.2, 100 mg, 0.27 mmol), Boc2O (70 mg, 0.32 mmol), and 20% Pd(OH)2/C (37 mg) to the flask.
- Stir the mixture under H2 atmosphere (balloon, 1 atm) at room temperature for 12 h.
- Monitor the reaction progress by TLC using ethyl acetate:hexanes (2:3 v/v, Rf = 0.29) as an eluent.
- Filter the reaction mixture through a commercially available celite pad (see Table of Materials) and wash with methanol.
- Evaporate in vacuo the filtrate and purify the residue by silica gel flash chromatography with ethyl acetate:hexanes (2:3 v/v) as the eluent to isolate pure product 5h (see Supplementary File 1) as a colorless liquid.
- Confirm the product by NMR and polarimeter measurements.
8. Polarimeter analysis
- Prepare an appropriate amount of the sample (~100 mg) to be measured.
- Dissolve the prepared sample in CHCl3 (c 0.05-1.00).
- Transfer the sample solution into the sample chamber, ensuring that there are no air bubbles
[ø = 1.8 mm, l = 10-1 m]. - Load the sample chamber into the polarimeter instrument (see Table of Materials) and check the orientation of the chamber.
- Set 0 as click zero clear in the 'control' section.
- Measure the specific rotation of CHCl3 for the blank.
NOTE: Light source: Na; λ = 589 nm; D.I.T: 5 s; cycle times: 5; cycle interval: 5 s; temperature: 20 °C. - Measure the specific rotation of the sample solution at a constant temperature.
NOTE: Light source: Na; λ = 589 nm; D.I.T: 5 s; cycle times: 5; cycle interval: 5 s; temperature: 20 °C. - Measure the specific rotation of the sample solution three times in the same way to obtain the average value.
- Calculate the specific rotation using the following equation27:
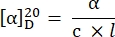
α = observed rotation (degrees), c = concentration (g/mL), l = path length (10-1 m).
9. 1H and 13C NMR analysis
- Prepare approximately 0.6-1.0 mL of the NMR solvent (CDCl3).
- Dissolve ~50 mg of sample in the solvent at a concentration of 0.02 M for 1H NMR and 0.05 M for 13C NMR measurements.
- Transfer the sample solution into an NMR tube using a Pasteur pipette.
- Load the tube into the NMR instrument (see Table of Materials).
NOTE: NMR measurements were performed using 400 MHz or 500 MHz spectrometers. Spin number: 16 (1H NMR), 256 or 512 (13C NMR); measurement time: 10 min (1H NMR), 20 or 30 min (13C NMR)]. - Record the NMR spectra and analyze the data.
NOTE: Reference the chemical shift of spectrum to CDCl3 signal [δ (1H NMR spectrum) = 7.26 ppm; δ (13C NMR spectrum) = 77.0 ppm)].
Access restricted. Please log in or start a trial to view this content.
Results
To investigate the achievability of preparing a contiguous bisaziridine, (E)-3-((S)-1-((R)-1-phenylethyl)aziridin-2-yl)acrylaldehyde (1a) was first synthesized as a model substrate according to the procedure mentioned in step 1 (Figure 1)28.
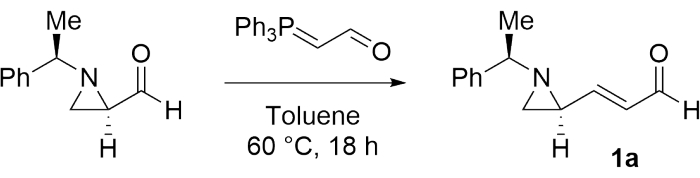
Figu...
Access restricted. Please log in or start a trial to view this content.
Discussion
The formation of an inseparable mixture of diastereomers has occasionally been observed during the course of organocatalytic aziridination of chiral 3-[1-(1-phenylethyl)aziridin-2-yl)]acrylaldehyde, when N-Boc-O-tosyl or N-Ts-O-tosyl hydroxylamine was used as the nitrogen source. Further, the yield of contiguous bisaziridine product decreased when the amount of diaryl silyl ether prolinol as catalyst was increased from 7 mol% to 20 mol%47,
Access restricted. Please log in or start a trial to view this content.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This research was supported by the Korea Basic Science Institute (National Research Facilities and Equipment Center) grant funded by the Ministry of Education (2022R1A6C101A751). This work was also supported by the National Research Foundation of Korea (NRF) grants (2020R1A2C1007102 and 2021R1A5A6002803).
Access restricted. Please log in or start a trial to view this content.
Materials
| Name | Company | Catalog Number | Comments |
| (R)-(+)-α,α-Diphenyl-2-pyrrolidinemethanol trimethylsilyl ether | Sigma-Aldrich | 677191 | reagent |
| (R)-1-((R)-1-phenylethyl)aziridine-2-carbaldehyde | Imagene Co.,Ltd. | reagent | |
| (S)-(–)-α,α-Diphenyl-2-pyrrolidinemethanol trimethylsilyl ether | Sigma-Aldrich | 677183 | reagent |
| (S)-2-(diphenyl((trim ethylsilyl)oxy)methyl)pyrrolidine | Sigma-Aldrich | 677183 | reagent |
| (Triphenylphosphoranylidene) acetaldehyde | Sigma-Aldrich | 280933 | reagent |
| 1,2-Dichloroethane | Sigma-Aldrich | 284505 | solvent |
| AB Sciex 4800 Plus MALDI TOFTM (2,5-dihydroxybenzoic acid (DHB) matrix | Sciex | High resolution mass spectra | |
| Acetic acid | Sigma-Aldrich | A6283 | reagent |
| Ammonium chloride | Sigma-Aldrich | 254134 | reagent |
| aniline | Sigma-Aldrich | 132934 | reagent |
| Autopol III digital polarimeter | Rudolph Research Analytical | polarimeter | |
| AVANCE III HD (400 MHz) spectrometer | Bruker | NMR spectrometer | |
| Bruker Ascend 500 (500 MHz) | Bruker | NMR spectrometer | |
| Celite 535 | Sigma-Aldrich | 22138 | For Celite pad |
| Dichloromethane | Sigma-Aldrich | 270997 | solvent |
| Di-tert-butyl dicarbonate | Sigma-Aldrich | 361941 | reagent |
| Ethyl Acetate | Sigma-Aldrich | 270989 | solvent |
| Ethyl nitroacetate | Sigma-Aldrich | 192333 | reagent |
| Imidazole | Sigma-Aldrich | I2399 | reagent |
| INOVA 400WB (400 MHz) | Varian | NMR spectrometer | |
| JMS-700 | JEOL | High resolution mass spectra | |
| Methanol | Sigma-Aldrich | 322415 | solvent |
| N-Boc-O-tosylhydroxylamine | Sigma-Aldrich | 775037 | reagent |
| P-2000 | JASCO | polarimeter | |
| Palladium hydroxide on carbon | Sigma-Aldrich | 212911 | reagent |
| Phenyl-1H-tetrazole-5-thiol | TCI | P0640 | reagent |
| Silica gel | Sigma-Aldrich | 227196 | For flash clromatography |
| Silica gel on TLC plates | Merck | 60768 | TLC plate |
| Sodium acetate | Sigma-Aldrich | S8750 | reagent |
| Sodium azide | Sigma-Aldrich | S2002 | reagent |
| Sodium borohydride | Sigma-Aldrich | 452882 | reagent |
| Sodium carbonate | Sigma-Aldrich | S2127 | reagent |
| tert-Butyldimethylsilyl chloride | Sigma-Aldrich | 190500 | reagent |
| Tetrahydrofuran | Sigma-Aldrich | 401757 | solvent |
| Toluene | Sigma-Aldrich | 244511 | solvent |
| Zinc bromide | Sigma-Aldrich | 230022 | reagent |
| Zinc chloride | Sigma-Aldrich | 429430 | reagent |
References
- Anastas, P. T., Warner, J. C. Principles of green chemistry. Green Chemistry: Theory and Practice. 29, Oxford University Press. (1998).
- Sheldon, R. A., Arends, I. W. C. E., Hanefeld, U. Green Chemistry and Catalysis. , Wiley. (2007).
- Trost, B. M. The atom economy-a search for synthetic efficiency. Science. 254 (5037), 1471-1477 (1991).
- Sheldon, R. A. The E factor: fifteen years on. Green Chemistry. 9 (12), 1273-1283 (2007).
- Li, C. J., Trost, B. M. Green chemistry for chemical synthesis. Proceedings of the National Academy of Sciences. 105 (36), 13197-13202 (2008).
- Sheldon, R. A. Fundamentals of green chemistry: efficiency in reaction design. Chemical Society Reviews. 41 (4), 1437-1451 (2012).
- Marion, P., et al. Sustainable chemistry: how to produce better and more from less. Green Chemistry. 19 (21), 4973-4989 (2017).
- Sheldon, R. A. The E factor 25 years on: the rise of green chemistry and sustainability. Green Chemistry. 19 (1), 18-43 (2017).
- Dembitsky, V. M., Terent'ev, A. O., Levitsky, D. O. Aziridine alkaloids: origin, chemistry and activity. Natural Products. , 977-1006 (2013).
- Ham, G. E. Activated aziridines. I. Reaction of anilines with O-ethyl-N,N-ethyleneurethane. mechanism and Hammett ρ-constant. Journal of Organic Chemistry. 29 (10), 3052-3055 (1964).
- Tanner, D. Chiral aziridine-their synthesis and use in stereoselective transformations. Angewandte Chemie International Edition. 33 (6), 599-619 (1994).
- Atkinson, R. S. 3-Acetoxyaminoquinazolinones (QNHOAc) as aziridinating agents: ring-opening of N-(Q)-substituted aziridines. Tetrahedron. 55 (6), 1519-1559 (1999).
- Sweeney, J. B. Aziridines: epoxides' ugly cousins. Chemical Society Reviews. 31 (5), 247-258 (2002).
- Lu, P. Recent developments in regioselective ring opening of aziridines. Tetrahedron. 14 (66), 2549-2560 (2010).
- Ohno, H. Synthesis and applications of vinylaziridines and ethynylaziridines. Chemical Reviews. 114 (16), 7784-7814 (2014).
- Callebaut, G., Meiresonne, T., De Kimpe, N., Mangelinckx, S. Synthesis and reactivity of 2-(carboxymethyl) aziridine derivatives. Chemical Reviews. 114 (16), 7954-8015 (2014).
- Ghosal, N. C., et al. Organocatalysis by an aprotic imidazolium zwitterion: Regioselective ring-opening of aziridines and applicable to gram scale synthesis. Green Chemistry. 18 (2), 565-574 (2016).
- Rai, V. K., Rai, P. K., Bajaj, S., Kumar, A. An unprecedented synthesis of γ-lactams via mercaptoacetylation of aziridines in water. Green Chemistry. 13 (5), 1217-1223 (2011).
- Srivastava, V. P., Yadav, L. D. S. The first example of ring expansion of N-tosylaziridines to 2-aroyl-N-tosylazetidines with nitrogen ylides in an aqueous medium. Green Chemistry. 12 (8), 1460-1465 (2010).
- Stanković, S., et al. Regioselectivity in the ring opening of non-activated aziridines. Chemical Society Reviews. 41 (2), 643-665 (2012).
- Ha, H. J., Jung, J. H., Lee, W. K. Application of regio-and stereoselective functional group transformations of chiral aziridine-2-carboxylates. Asian Journal of Organic Chemistry. 3 (10), 1020-1035 (2014).
- D'hooghe, M., Ha, H. -J. Synthesis of 4- to 7-membered Heterocycles by Ring Expansion: Aza-, Oxa- and Thiaheterocyclic Small-Ring Systems. 1st ed. , Springer International Publishing. Cham. (2016).
- Macha, L., Ha, H. -J. Total synthesis and absolute stereochemical assignment of microgrewiapine A and its stereoisomers. Journal of Organic Chemistry. 84 (1), 94-103 (2019).
- Srivastava, N., Macha, L., Ha, H. -J. Total synthesis and stereochemical revision of biemamides B and D. Organic Letters. 21 (22), 8992-8996 (2019).
- Lee, W. K., Ha, H. -J. Highlights of the chemistry of enantiomerically pure aziridine-2-carboxylates. Aldrichimica Acta. 36 (2), 57-63 (2003).
- Głowacka, I. E., Trocha, A., Wróblewski, A. E., Piotrowska, D. G. N-(1-Phenylethyl) aziridine-2-carboxylate esters in the synthesis of biologically relevant compounds. Beilstein Journal of Organic Chemistry. 15 (1), 1722-1757 (2019).
- JoVE. Organic Chemistry II. Polarimeter. JoVE Science Education Database. , JoVE. Cambridge, MA. (2022).
- Mao, H., et al. Preparation of chiral contiguous epoxyaziridines and their regioselective ring-opening for drug syntheses. Chemistry-A European Journal. 24 (10), 2370-2374 (2018).
- Vesely, J., Ibrahem, I., Zhao, G. L., Rios, R., Córdova, A. Organocatalytic enantioselective aziridination of α,β-unsaturated aldehydes. Angewandte Chemie International Edition. 11 (46), 778-781 (2007).
- Arai, H., et al. Enantioselective aziridination reaction of α,β-unsaturated aldehydes using an organocatalyst and tert-butyl N-arenesulfonyloxycarbamates. Tetrahedron Letters. 50 (26), 3329-3332 (2009).
- Desmarchelier, A., et al. Organocatalyzed aziridination of α-branched enals: enantioselective synthesis of aziridines with a quaternary stereocenter. European Journal of Organic Chemistry. 20 (2011), 4046-4052 (2011).
- Jiang, H., Halskov, K. S., Johansen, T. K., Jørgensen, K. A. Deracemization of axially chiral α,β-unsaturated aldehydes through an amino-catalyzed symmetry-making-symmetry-breaking cascade. Chemistry-A European Journal. 17 (14), 3842-3846 (2011).
- Deiana, L., et al. Catalytic asymmetric aziridination of α,β-unsaturated aldehydes. Chemistry-A European Journal. 17 (28), 7904-7917 (2011).
- Molnár, I. G., Tanzer, E. M., Daniliuc, C., Gilmour, R. Enantioselective aziridination of cyclic enals facilitated by the fluorine-iminium Ion gauche effect. Chemistry-A European Journal. 20 (3), 794-800 (2014).
- Nemoto, T., et al. Enantioselective synthesis of (R)-Sumanirole using organocatalytic asymmetric aziridination of an α,β-unsaturated aldehyde. Tetrahedron: Asymmetry. 25 (15), 1133-1137 (2014).
- Sim, T. B., et al. A novel synthesis of 5-functionalized oxazolidin-2-ones from enantiomerically pure 2-substituted N-[(R)-(+)-α-methylbenzyl] aziridines. Journal of Organic Chemistry. 68 (1), 104-108 (2003).
- Silva, M. A., Goodman, J. M. Aziridinium ring opening: a simple ionic reaction pathway with sequential transition states. Tetrahedron Letters. 46 (12), 2067-2069 (2005).
- Yun, S. Y., et al. Nucleophile-dependent regioselective ring opening of 2-substituted N,N-dibenzylaziridinium ions: bromide versus hydride. Chemical Communications. (18), 2508-2510 (2009).
- Dolfen, J., et al. Bicyclic aziridinium ions in azaheterocyclic chemistry-preparation and synthetic application of 1-azoniabicyclo [n. 1.0] alkanes. Advanced Synthesis & Catalysis. 358 (22), 3485-3511 (2016).
- D'hooghe, M., et al. Systematic study of halide-induced ring opening of 2-substituted aziridinium salts and theoretical rationalization of the reaction pathways. European Journal of Organic Chemistry. 2010 (25), 4920-4931 (2010).
- Boydas, E. B., et al. Theoretical insight into the regioselective ring-expansions of bicyclic aziridinium ions. Organic & Biomolecular Chemistry. 16 (5), 796-806 (2018).
- Lee, B. K., et al. An efficient synthesis of chiral terminal 1, 2-diamines using an enantiomerically pure [1-(1′ R)-methylbenzyl] aziridine-2-yl] methanol. Tetrahedron. 62 (35), 8393-8397 (2006).
- Ha, H. J., et al. Addition of 1-Boc-2-tert-butyldimethylsilyloxypyrrole to N-methyleneamine equivalents: synthesis of 1-Boc-5-aminomehtyl-2,5-dihydropyrrole-2-ones and 1-Boc-2-oxo-1,7,9-triazaspiro[4,5]-dec-3-ene. Heterocycles. 50 (1), 203-214 (1999).
- Laughlin, R. G. The basicity of aliphatic sulfonamides. Journal of the American Chemical Society. 89 (17), 4268-4271 (1967).
- Moreira, J. A., Rosa da Costa, A. M., García-Río, L., Pessêgo, M. Equilibrium constants and protonation site for N-methylbenzenesulfonamides. Beilstein Journal of Organic Chemistry. 7 (1), 1732-1738 (2011).
- Song, K., et al. Highly active ruthenium metathesis catalysts enabling ring-opening metathesis polymerization of cyclopentadiene at low temperatures. Nature Communications. 10, 3860(2019).
- Fukuta, Y., et al. De novo synthesis of Tamiflu via a catalytic asymmetric ring-opening of meso-aziridines with TMSN3. Journal of the American Chemical Society. 128 (19), 6312-6313 (2006).
- Jiang, H., et al. Intramolecular radical aziridination of allylic sulfamoyl azides by cobalt (II)-based metalloradical catalysis: effective construction of strained heterobicyclic structures. Angewandte Chemie International Edition. 55 (38), 11604-11608 (2016).
- Righi, G., Bovicelli, P., Barontini, M., Tirotta, I. Dimethyl carbonate in the regio-and stereocontrolled opening of three-membered heterocyclic rings. Green Chemistry. 14 (2), 495-502 (2012).
- Righi, P., et al. Solution- and solid-phase synthesis of 4-hydroxy-4,5-dihydroisoxazole derivatives from enantiomerically pure N-tosyl-2,3-aziridine alcohols. Organic Letters. 4 (4), 497-500 (2002).
- Yadav, N. N., Choi, J., Ha, H. -J. One-pot multiple reactions: asymmetric synthesis of 2, 6-cis-disubstituted piperidine alkaloids from chiral aziridine. Organic & Biomolecular Chemistry. 14 (27), 6426-6434 (2016).
- Rhee, H. J., et al. Preparation and utilization of contiguous bisaziridines as chiral building blocks. Advanced Synthesis & Catalysis. 363 (13), 3250-3257 (2021).
Access restricted. Please log in or start a trial to view this content.
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved