载氧钴 (II.) 络合物的合成
Overview
资料来源: 迪皮卡·乌达加马, 塔玛拉, 德克萨斯大学化学系, #38; M 学院
无机化学是研究金属在生物学中所起的作用的领域。大约一半的蛋白质含有金属, 据估计, 所有蛋白质中有三分之一依赖于含有金属的活性部位来起作用。以金属为特征的蛋白质, 称为金属, 在各种细胞功能中起着至关重要的作用, 这是生命所必需的。几十年来, 金属一直对合成无机化学家产生了兴趣和启发, 许多研究小组都通过研究配位化合物, 致力于将含有金属的活性部位的化学模拟。
O2的传输是生物体的一个重要过程。O2-运输金属负责捆绑、运输和释放氧气, 然后可用于生命过程, 如呼吸。载氧钴配合物, [n,n' 双 (醛) ethylenediimino] 钴 (II) [Co (salen)]2已被广泛研究, 以了解金属络合物如何可逆地绑定 O2.1
在本实验中, 我们将合成 [Co (salen)]2 , 并研究其在砜 (亚砜) 存在时与 O2的可逆反应。首先, 我们将量化在 [Co (salen)]2与亚甲基亚砜接触时消耗的 O2的数量。然后, 我们将通过将实体公开为 CHCl3, 以可视方式观察从 [Co (salen)]2-2加合物中释放 o2 。
Procedure
1. 非活动 [Co (salen)] 的合成2
- 250毫升3颈圆底烧瓶, 120 毫升95% 乙醇和2.20 克 (0.192 毫升, 0.018 摩尔) 的醛。
- 使中心颈部与连接到 N2的冷凝器匹配。将其他两个颈部与橡胶隔膜配合, 并配有橡胶隔膜的附加漏斗。
- 在水浴中搅动反应并将溶液加热到回流 (80 ° c)。
- 通过圆底烧瓶隔膜, 通过注射器加入乙烯二胺 (0.52 克, 0.58 毫升, 0.0087 摩尔)。
- 在50毫升的圆底烧瓶中, 在0.0087 毫升蒸馏水中制备 Co (华侨)2· 4H2O (2.17 g, 15 摩尔) 的溶液。在含有3颈烧瓶的同一水浴中加热溶液, 以确保醋酸钴全部溶解。
- 添加醋酸钴溶液到加法漏斗。
- 通过冒泡 N2通过在加法漏斗中的液体在 10 min (参见 "用 Schlenk 线技术合成一个 Ti (III) 茂金属" 的视频, 用于清洗液体的更
Results
非活动 [Co (salen)]2的特性:
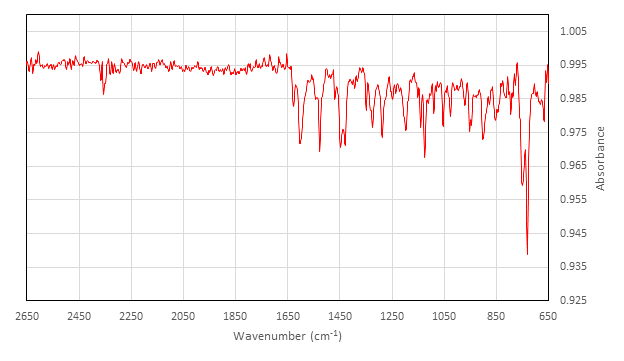
在 ATR 附件上收集的 IR (cm-1): 2357 (w)、1626 (w)、1602 (m)、1542 (w)、1528 (m)、1454 (w)、1448 (m)、1429 (m) 1348 (w)、1327 (w)、1323 (m)、1288 (m)、1248 (w)、1236 (w)、1197 (m)、1140 (m)、1124 (m)、1089 (w)、1053 (m)、1026 (w), 970 (w), 952 (w), 947 (w), 902 (m), 878 (w), 845 (w), 813 (w), 794 (w), 750 (s), 730 ...
Application and Summary
Tags
跳至...
此集合中的视频:

Now Playing
载氧钴 (II.) 络合物的合成
Inorganic Chemistry
51.7K Views

用 Schlenk 线技术合成钛 (III) 茂金属
Inorganic Chemistry
31.6K Views

手套和杂质传感器
Inorganic Chemistry
18.7K Views

二茂铁的升华纯化
Inorganic Chemistry
54.7K Views

埃文斯方法
Inorganic Chemistry
68.7K Views

单晶和粉末 X 射线衍射
Inorganic Chemistry
105.1K Views

电子顺磁共振 (EPR) 光谱学
Inorganic Chemistry
25.6K Views

尔光谱学
Inorganic Chemistry
22.0K Views

路易斯酸碱交互作用在 Ph 值3P BH3
Inorganic Chemistry
39.0K Views

二茂铁结构
Inorganic Chemistry
79.7K Views

群论在红外光谱分析中的应用
Inorganic Chemistry
45.8K Views

分子轨道 (MO) 理论
Inorganic Chemistry
35.5K Views

异体金属-金属粘结 Paddlewheels
Inorganic Chemistry
15.3K Views

染料敏化太阳能电池
Inorganic Chemistry
16.0K Views

自由基聚合反应的光化学引发
Inorganic Chemistry
17.1K Views
版权所属 © 2025 MyJoVE 公司版权所有,本公司不涉及任何医疗业务和医疗服务。
