A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
Preparation of Zinc Oxide Nanoparticles and the Evaluation of their Antibacterial Effects
* These authors contributed equally
In This Article
Summary
In this study, zinc oxide nanoparticles were synthesized using a precipitation method. The antibacterial effect of the synthesized particles was tested against multidrug-resistant methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa bacterial strains.
Abstract
Nosocomial bacterial infections have become increasingly challenging due to their inherent resistance to antibiotics. The emergence of multidrug-resistant bacterial strains in hospitals has been attributed to the extensive and varied use of antibiotics, further exacerbating the problem of antibiotic resistance. Metal nanomaterials have been widely studied as an alternative solution for eradicating antibiotic-resistant bacterial cells. Metallic nanoparticles attack bacterial cells through various mechanisms, such as the release of antibacterial ions, generation of reactive oxygen species, or physical disruption, against which bacteria cannot develop resistance. Among the actively researched antimicrobial metal nanoparticles, zinc oxide nanoparticles, which are FDA-approved, are known for their biocompatibility and antibacterial properties. In this study, we focused on successfully developing a precipitation method for synthesizing zinc oxide nanoparticles, analyzing the properties of these nanoparticles, and conducting antimicrobial tests. Zinc oxide nanoparticles were characterized using transmission electron microscopy (TEM), dynamic light scattering (DLS), ultraviolet/visible spectroscopy, and X-ray diffraction (XRD). Antibacterial tests were conducted using the broth microdilution test with the multidrug-resistant strains of methicillin-resistant Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa. This study demonstrated the potential of zinc oxide nanoparticles in inhibiting the proliferation of antibiotic-resistant bacteria.
Introduction
Multidrug-resistant (MDR) bacterial infections pose a significant global threat to human health1. As these infections can be fatal in patients with underlying conditions, active research is attempting to address this issue2. Bacteria have evolved to evade the action of various drugs. Penicillin, widely known and credited with saving millions of lives worldwide, is a β-lactam antibiotic that inhibits the synthesis of the bacterial cell wall3. However, bacteria have evolved to neutralize the efficacy of drugs through various mechanisms such as efflux pumps, transpeptidase alterations, or decreased permeability4. Additionally, bacterial cells can transmit these resistance genes to the next generation, increasing the survival rates of the subsequent generation and strengthening the problem of resistant strains5.
The increase in antibiotic-resistant bacteria has led to the emergence of MDR bacteria, which commonly exhibit resistance to multiple antibiotics. MDR strains are most frequently encountered in hospital settings, where multiple bacterial strains are exposed to and consequently develop resistance to different antibiotics6. Staphylococcus aureus, particularly methicillin-resistant S. aureus (MRSA), is a gram-positive commensal bacterium that forms clusters on the skin of approximately 30% of humans7,8. MRSA, which was first identified in the 1960s, exhibits reduced sensitivity to β-lactam antibiotics, resulting in a sharp increase in infection rates since the 1990s9. Among gram-negative bacteria, Pseudomonas aeruginosa (P. aeruginosa) is one of the most prevalent strains acquired in hospitals. This species, a facultative rod-shaped bacterium, causes opportunistic infections in humans10. Particularly, MDR strains that directly affect human health are responsible for over 50 % of healthcare-associated infections11. In this study, we utilized the most commonly encountered multidrug-resistant strains within hospitals, MRSA and P. aeruginosa.
The use of nanoparticles (NPs) for antimicrobial purposes has been extensively investigated to tackle the issue of antibiotic resistance. Metallic NPs, in particular, induce bacterial cell death through various mechanisms, offering a potential solution to the problem of drug resistance. Metallic NPs exert antimicrobial activity through multiple mechanisms, including the release of antimicrobial ions, generation of reactive oxygen species (ROS), and physical disruption of cells, among other means12. NPs composed of silver, copper, zinc oxide (ZnO), and titanium oxide possess high antimicrobial efficacy and are thus being actively researched13.
ZnO NPs have been approved by the U.S. Food and Drug Administration (FDA) for use in humans. Conversely, despite their high antimicrobial efficacy, the use of silver and copper NPs in humans is limited by their high cytotoxicity. However, ZnO NPs are commonly found in everyday life and are even present in widely used sunscreen formulations14. Of note, Zn2+ ions released from ZnO NPs are highly effective in bacterial treatment, inducing bacterial cell death through the generation of ROS and other physical damage mechanisms15.
This study outlines the protocol for synthesizing ZnO nanoparticles (NPs) using a precipitation method and introduces an antimicrobial testing approach using a microbroth dilution method with clinical samples of MRSA and P. aeruginosa. The precipitation method for ZnO NPs involves synthesizing insoluble solid ZnO NPs by adjusting pH and temperature using soluble precursors such as zinc acetate or zinc nitrate16. Along with relatively facile and rapid production, this method ensures repeatability in synthesis and facilitates control over particle size and morphology17. In this synthesis protocol, sodium hydroxide (NaOH), one of the most commonly used precipitation agents, was utilized to precipitate zinc acetate, and a small amount of hexadecyltrimethylammonium bromide (CTAB) was employed to inhibit the uncontrolled synthesis of the nanoparticles18. Among various antimicrobial tests, the antibacterial activity of ZnO nanoparticles was evaluated using the microbroth dilution method, which avoids optical interference from metal oxide nanoparticles and enables direct colony measurement for determining MIC19.
Protocol
The reagents and equipment used in this study are listed in the Table of Materials.
1. Preparation of zinc oxide nanoparticles
- Measure 200 mL of absolute ethyl alcohol and pour it into a glass round bottom flask.
- Place the round bottom flask on a heating mantle and maintain stirring at 25-40 °C.
- Measure 500 mg of CTAB in a 50 mL vial and add it to the ethyl alcohol in the flask. Stir until CTAB is completely dissolved.
- Add 1.4 g of zinc acetate into the solution and stir until it is completely dissolved.
- Raise the temperature of the solution by setting the heating mantle temperature to 70 °C.
- Add 25 mL of 0.5 M NaOH solution to the mixture and let it react for 1 h until the clear solution becomes white in color.
- Aliquot the solution to 50 mL conical tubes, centrifuge at 15000 × g for 15 min at room temperature, and then discard the supernatant.
- Add 10 mL of distilled water to one of the conical tubes and resuspend the nanoparticles by sonicating the solution. Transfer the suspended solution to a different conical tube containing the ZnO pellet and repeat until all ZnO solutions are collected in one conical tube.
- Wash the nanoparticles through centrifugation at 15000 × g for 15 min (room temperature), remove the supernatant, and resuspend in distilled water. Check the pH of the supernatant solution using pH test paper and repeat until the pH of the solution becomes neutral.
NOTE: When the pH of the supernatant solution becomes neutral (pH = 7), discard the supernatant solution and do not resuspend with distilled water. - Vacuum dry the sample pallet at 60 °C for 24 h and obtain the ZnO NP powder.
2. Antibacterial tests using MRSA and P. aeruginosa
- Bacterial culture
NOTE: Clinical MDR bacterial strains were obtained from Chung-Ang University Hospital, Seoul, South Korea.- Take out MRSA and P. aeruginosa bacterial strains stocked in Tryptic Soy Broth (TSB) from the deep freezer.
- After thawing the bacterial solutions, streak the solution onto a Tryptic Soy Agar (TSA) plate using a disposable inoculation loop. Place the streaked agar plates in the incubator and incubate for 24 h.
NOTE: A single colony picked from the TSA plate was added to 10 mL of TSB media in a 50 mL conical tube using a bacterial inoculation loop. Bacteria were cultured for 24 h. Bacteria were cultured under aerobic culture conditions at 37 °C. - To measure the concentration of the bacteria solution, dilute the cultured solution using 10-fold serial dilution to 10-6 using distilled water. Afterward, place 50 µL of the diluted solution onto TSA plates and spread the solution using a L-shaped spreader.
- Incubate the plates in the incubator for 24 h.
NOTE: Colonies were optically counted, and the concentration of the cultured solution was calculated by multiplying the dilution factors by the number of colonies counted.
- Bacterial sampling
- To test a wide range of ZnO concentrations, prepare 2 mg/mL ZnO NPs solution using Dulbecco's Phosphate-Buffered Saline (DPBS) and perform 2-fold serial dilution to make different concentrations.
NOTE: 1000 µg/mL, 500 µg/mL, 250 µg/mL, 125 µg/mL, and 62.5 µg/mL were tested. - Add 100 µL for each of the tested concentrations of ZnO NPs into a 96-well plate.
NOTE: Use double (2x) of the final desired concentration of ZnO NPs, as each sample in the well will be diluted with the addition of 100 µL of bacteria culture. Use a 2% Antibiotic-Antimycotic (A/A) solution as the positive control and DPBS as the negative control. A/A is a penicillin and streptomycin antibiotic complex, which is effective against gram-positive and gram-negative bacteria, respectively. - Dilute the bacterial culture to 1 × 106 CFU/mL using TSB media and add 100 µL to each well containing different concentrations of ZnO NPs.
NOTE: Initial MRSA and P. aeruginosa culture solution concentrations were 3 x 109 CFU/mL. Bacteria culture concentration 1 x 106 CFU/mL was made by 1/20 and 1/150 dilution of the culture solution. After mixing with 100 µL of ZnO NPs, the final concentration of bacteria will be 5 × 105 CFU/mL. - Place the 96-well plate in a 37 °C incubator and incubate for 24 h.
- To test a wide range of ZnO concentrations, prepare 2 mg/mL ZnO NPs solution using Dulbecco's Phosphate-Buffered Saline (DPBS) and perform 2-fold serial dilution to make different concentrations.
- Bacterial spreading
- Pipette 100 µL from each well and prepare various 10-fold serial dilutions until 10-6.
NOTE: Add 100 µL of ZnO NPs containing bacterial solution into 900 µL of sterilized distilled water and repeat 6 times. - Pipette 50 µL from four diluted solutions and add to TSA media plates. Use an L-shaped cell spreader to spread the bacterial suspension onto the agar plate.
NOTE: Use the 100, 10-2, 10-4, and 10-6 dilutions. Conduct all experiments in triplicate. Spread until all the bacterial solution is absorbed onto the agar plate. - Place the agar plates into a 37 °C incubator and incubate for 24 h.
- Pipette 100 µL from each well and prepare various 10-fold serial dilutions until 10-6.
- Bacterial CFU counting
- Select a dilution factor that is countable for each group. Mark all the colonies in the countable dilution plate and recalculate so that the concentration becomes # of CFU/mL.
NOTE: The countable dilution factor indicates plates that have 20-100 CFU on an individual plate. Initially counted concentration results # of Counted CFU/50 µL. Using the spread plate with the undiluted bacterial solution, determine the potential minimum bactericidal concentration. - Use the obtained data to represent the percentage of live bacteria relative to those in the negative control.
NOTE: Percentage of live bacteria (%) =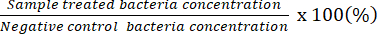
- Select a dilution factor that is countable for each group. Mark all the colonies in the countable dilution plate and recalculate so that the concentration becomes # of CFU/mL.
Results
The successful synthesis of ZnO NPs was confirmed using transmission electron microscopy (TEM), as shown in Figure 1A. The obtained ZnO NPs were observed to be round in shape, with an average particle size of 35.35 nm and a standard deviation of 6.81 nm. The precipitation of these nanoparticles was observed through a double-displacement reaction by adding NaOH solution to zinc acetate, where Zn2+ ions underwent hydrolysis.
Using dynamic light scattering...
Discussion
The synthesis of ZnO NPs via precipitation is relatively simple and straightforward. To successfully synthesize ZnO NPs using this method, stirring is crucial to ensure that the precursor (zinc acetate) is fully dissolved in the solvent. Moreover, increasing the temperature helps to induce a successful double-displacement reaction. In the synthesis of ZnO NPs, there are many factors that determine the size and shape, including the precipitation agent, the concentration of the precipitation agent, and the surfact...
Disclosures
Dr. Jonghoon Choi is the CEO/Founder, and Dr. Yonghyun Choi is the CTO of the Feynman Institute of Technology at the Nanomedicine Corporation.
Acknowledgements
This research was supported by the Chung-Ang University Graduate Research Scholarship in 2022 (Ms. Gahyun Lee). This work was also supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2020R1A5A1018052) and by the Technology development Program (RS202300261938) funded by the Ministry of SMEs and Startups (MSS, Korea).
Materials
| Name | Company | Catalog Number | Comments |
| DLS | Zetasizer Pro | ||
| Ethyl alcohol, absolute | DAEJUNG | 4023-2304 | |
| Microplate reader | BioTeck | ||
| Sodium Hydroxide | Sigma-Aldrich | 221465 | |
| TEM | JEOL JEM-F200 | ||
| TSA | DB difco | 236950 | |
| TSB | DB difco | 211825 | |
| XRD | NEW D8-Advance | ||
| Zinc acetate | Sigma-Aldrich | 383317 |
References
- Catalano, A., et al. Multidrug resistance (MDR): A widespread phenomenon in pharmacological therapies. Molecules. 27 (3), 616 (2022).
- Bazaid, A. S., et al. Bacterial infections among patients with chronic diseases at a tertiary care hospital in Saudi Arabia. Microorganisms. 10 (10), 1907 (2022).
- Miller, E. L. The penicillins: A review and update. J Midwifery Women's Health. 47 (6), 426-434 (2002).
- Martínez-Trejo, A., et al. Evasion of antimicrobial activity in acinetobacter baumannii by target site modifications: An effective resistance mechanism. Int J Mol Sci. 23 (12), 6582 (2022).
- Jiang, J. -. H., et al. Antibiotic resistance and host immune evasion in staphylococcus aureus mediated by a metabolic adaptation. Proc Natl Acad Sci U S A. 116 (9), 3722-3727 (2019).
- Lee, H. N., et al. A lateral flow assay for nucleic acid detection based on rolling circle amplification using capture ligand-modified oligonucleotides. BioChip J. 16 (4), 441-450 (2022).
- Craft, K. M., Nguyen, J. M., Berg, L. J., Townsend, S. D. Methicillin-resistant staphylococcus aureus (MRSA): Antibiotic-resistance and the biofilm phenotype. Med Chem Comm. 10 (8), 1231-1241 (2019).
- Tieu, M. -. V., Pham, D. T., Le, H. T. N., Hoang, T. X., Cho, S. Rapid and ultrasensitive detection of Staphylococcus aureus using a gold-interdigitated single-wave-shaped electrode (AU-ISWE) electrochemical biosensor. BioChip J. 17, 1-10 (2023).
- Turner, N. A., et al. Methicillin-resistant Staphylococcus aureus: An overview of basic and clinical research. Nat Rev Microbiol. 17 (4), 203-218 (2019).
- Tuon, F. F., Dantas, L. R., Suss, P. H., Tasca Ribeiro, V. S. Pathogenesis of the Pseudomonas aeruginosa biofilm: A review. Pathogens. 11 (3), 300 (2022).
- Sarabhai, S., Sharma, P., Capalash, N. Ellagic acid derivatives from terminalia chebula retz. Downregulate the expression of quorum sensing genes to attenuate pseudomonas aeruginosa pao1 virulence. PLoS One. 8 (1), e53441 (2013).
- Dizaj, S. M., Lotfipour, F., Barzegar-Jalali, M., Zarrintan, M. H., Adibkia, K. Antimicrobial activity of the metals and metal oxide nanoparticles. Mat Sci Eng: C. 44, 278-284 (2014).
- Ribeiro, A. I., Dias, A. M., Zille, A. Synergistic effects between metal nanoparticles and commercial antimicrobial agents: A review. ACS App Nano Mater. 5 (3), 3030-3064 (2022).
- Newman, M. D., Stotland, M., Ellis, J. I. The safety of nanosized particles in titanium dioxide-and zinc oxide-based sunscreens. J Am Acad Dermatol. 61 (4), 685-692 (2009).
- Sivakumar, P., Lee, M., Kim, Y. -. S., Shim, M. S. Photo-triggered antibacterial and anticancer activities of zinc oxide nanoparticles. J Mater Chem B. 6 (30), 4852-4871 (2018).
- Kołodziejczak-Radzimska, A., Jesionowski, T. Zinc oxide-from synthesis to application: A review. Materials. 7 (4), 2833-2881 (2014).
- Raoufi, D. Synthesis and microstructural properties of ZnO nanoparticles prepared by precipitation method. Renew Energy. 50, 932-937 (2013).
- Wang, Y. -. X., Sun, J., Fan, X., Yu, X. A CTAB-assisted hydrothermal and solvothermal synthesis of ZnO nanopowders. Ceram Int. 37 (8), 3431-3436 (2011).
- Balouiri, M., Sadiki, M., Ibnsouda, S. K. Methods for in vitro evaluating antimicrobial activity: A review. J Pharm Anal. 6 (2), 71-79 (2016).
- Clogston, J. D., Patri, A. K., McNeil, S. Zeta potential measurement. Characterization of nanoparticles intended for drug delivery. Methods Mol Biol. , 63-70 (2011).
- Abdelbaky, A. S., Mohamed, A. M., Sharaky, M., Mohamed, N. A., Diab, Y. M. Green approach for the synthesis of ZnO nanoparticles using Cymbopogon citratus aqueous leaf extract: Characterization and evaluation of their biological activities. Chem Biol Technol Agric. 10 (1), 63 (2023).
- Ankamwar, B. G., Kamble, V. B., Annsi, J. I., Sarma, L. S., Mahajan, C. M. Solar photocatalytic degradation of methylene blue by ZnO nanoparticles. J Nanosci Nanotechnol. 17 (2), 1185-1192 (2017).
- Babayevska, N., et al. Zno size and shape effect on antibacterial activity and cytotoxicity profile. Scientific Rep. 12 (1), 8148 (2022).
- Hamidian, K., Sarani, M., Behjati, S., Mahjoub, M., Zafarnia, N. Cytotoxic performance of synthesized mn-doped ZnO nanorods in mcf-7 cells. ChemistrySelect. 8 (19), e202300292 (2023).
- Gharpure, S., et al. Non-antibacterial and antibacterial ZnO nanoparticles composed of different surfactants. J Nanosci Nanotechnol. 21 (12), 5945-5959 (2021).
- Dhoke, S. K. Synthesis of nano-ZnO by chemical method and its characterization. Results Chem. 5, 100771 (2023).
- Choi, K. -. C., et al. Modifying hydrogen bonding interaction in solvent and dispersion of ZnO nanoparticles: Impact on the photovoltaic performance of inverted organic solar cells. RSC Adv. 4 (14), 7160-7166 (2014).
- Lee, W., et al. High colloidal stability ZnO nanoparticles independent on solvent polarity and their application in polymer solar cells. Scientific Rep. 10 (1), 18055 (2020).
- Cozzoli, P. D., Kornowski, A., Weller, H. Colloidal synthesis of organic-capped ZnO nanocrystals via a sequential reduction-oxidation reaction. J Phy Chem B. 109 (7), 2638-2644 (2005).
- Roh, S., Jang, Y., Yoo, J., Seong, H. Surface modification strategies for biomedical applications: Enhancing cell-biomaterial interfaces and biochip performances. BioChip J. 17, 1-18 (2023).
- Chen, D., Jiao, X., Cheng, G. Hydrothermal synthesis of zinc oxide powders with different morphologies. Solid State Commun. 113 (6), 363-366 (1999).
- Hu, X. -. L., Zhu, Y. -. J., Wang, S. -. W. Sonochemical and microwave-assisted synthesis of linked single-crystalline ZnO rods. Mater Chem Phys. 88 (2-3), 421-426 (2004).
- Ahamad Khan, M., et al. Phytogenically synthesized zinc oxide nanoparticles (ZnO-NPs) potentially inhibit the bacterial pathogens: In vitro studies. Toxics. 11 (5), 452 (2023).
- Xie, J., et al. Recent advances in ZnO nanomaterial-mediated biological applications and action mechanisms. Nanomaterials. 13 (9), 1500 (2023).
- Chawla, U., et al. A review on ZnO-based targeted drug delivery system. Lett Drug Des Discov. 21 (3), 397-420 (2024).
- Alavi, M., Nokhodchi, A. An overview on antimicrobial and wound healing properties of ZnO nanobiofilms, hydrogels, and bionanocomposites based on cellulose, chitosan, and alginate polymers. Carbohydr Polym. 227, 115349 (2020).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved