A subscription to JoVE is required to view this content. Sign in or start your free trial.
Method Article
A Murine Model of Pressure Overload-Induced Right Ventricular Hypertrophy and Failure by Pulmonary Trunk Banding
In This Article
Summary
We describe a murine model of right ventricular pressure overload-induced by pulmonary trunk banding. Detailed protocols for intubation, surgery, and phenotyping by echocardiography are included in the paper. Custom-made instruments are used for intubation and surgery, allowing for fast and inexpensive reproduction of the model.
Abstract
Right ventricular (RV) failure caused by pressure overload is strongly associated with morbidity and mortality in a number of cardiovascular and pulmonary diseases. The pathogenesis of RV failure is complex and remains inadequately understood. To identify new therapeutic strategies for the treatment of RV failure, robust and reproducible animal models are essential. Models of pulmonary trunk banding (PTB) have gained popularity, as RV function can be assessed independently of changes in the pulmonary vasculature.
In this paper, we present a murine model of RV pressure overload induced by PTB in 5-week-old mice. The model can be used to induce different degrees of RV pathology, ranging from mild RV hypertrophy to decompensated RV failure. Detailed protocols for intubation, PTB surgery, and phenotyping by echocardiography are included in the paper. Furthermore, instructions for customizing instruments for intubation and PTB surgery are given, enabling fast and inexpensive reproduction of the PTB model.
Titanium ligating clips were used to constrict the pulmonary trunk, ensuring a highly reproducible and operator-independent degree of pulmonary trunk constriction. The severity of PTB was graded by using different inner ligating clip diameters (mild: 450 µm and severe: 250 µm). This resulted in RV pathology ranging from hypertrophy with preserved RV function to decompensated RV failure with reduced cardiac output and extracardiac manifestations. RV function was assessed by echocardiography at 1 week and 3 weeks after surgery. Examples of echocardiographic images and results are presented here. Furthermore, results from right heart catheterization and histological analyses of cardiac tissue are shown.
Introduction
Right ventricular (RV) failure is a clinical syndrome with symptoms of heart failure and signs of systemic congestion resulting from RV dysfunction1. RV dysfunction is strongly associated with morbidity and mortality in a number of cardiovascular and pulmonary diseases2. The etiology of RV dysfunction is complex, and its underlying signaling pathways and regulation remain inadequately elucidated.
Observations from current therapies show that improved RV function correlates closely to afterload reduction, suggesting pulmonary vasculature as the primary treatment target3. This indicates that current therapies only have a minimal direct effect on RV function, which can deteriorate even after the improvement of pulmonary vascular resistance3. Further research into improving RV function independently of afterload reduction is thus highly needed.
Robust and reproducible animal models are essential in the search for new therapeutic agents. In most models of chronic RV failure, the underlying cause is pulmonary hypertension induced by structural alteration of the pulmonary vasculature4,5,6. Well-characterized models include the chronic hypoxia model7,8, the Sugen-hypoxia model9,10,11, and the monocrotaline model12,13. Because the RV failure is secondary to pulmonary hypertension in these models, it is impossible to differentiate the effects of interventions on the pulmonary vasculature from the direct effects on the RV6.
To study the RV independently from the pulmonary vasculature, the pulmonary trunk banding (PTB) model has gained popularity and has been described in several animal species, including mice, rats, rabbits, dogs, sheep, and pigs6,14,15,16,17,18,19,20,21,22,23,24,25,26,27. In PTB models, constriction of the pulmonary trunk is achieved surgically, causing an increase in RV pressure6. Different approaches to the application of PTB exist, including constriction of the vessel with a ligature or with a metal ligating clip18,28. In models using ligatures, the pulmonary trunk is tied to a needle, and the needle is retracted, leaving the ligature in place. This results in a constriction of the vessel that depends on the needle size and the tension of the knot18,29. In models employing metal ligating clips, the degree of pulmonary trunk constriction may be more reproducible. Modified ligating clip appliers are used to close the ligating clips to a predefined and constant diameter. This makes the method operator-independent and reduces PTB-related variability in the disease phenotype15,27,28.
Murine PTB models have been shown to induce RV hypertrophy and failure18,28. One major challenge when using the PTB model is choosing the appropriate PTB diameter to achieve the desired degree of RV pathology. This is especially challenging when attempting to model decompensated RV failure. For this, the constriction needs to be tight enough to induce chronic RV failure without leading to acute RV failure and death shortly after surgery6. One approach to solving this challenge is to use weanlings or juvenile animals6,15. A PTB model has been used successfully to study different stages of RV failure using Wistar rat weanlings15,30. To achieve this, juvenile rats with remaining growth potential underwent PTB surgery with the application of titanium ligating clips. When the rats grew, the pulmonary stenosis gradually became more severe and resulted in RV hypertrophy or chronic RV failure, depending on the severity of PTB15,30. Inspired by this model, we hypothesized that different stages of RV pathology could be produced in a murine PTB model using juvenile mice. Studying a broad spectrum of RV pathology from mild to severe disease may help elucidate our understanding of disease progression and the transition from RV hypertrophy to RV failure.
Here, we present a murine model of RV pressure overload induced by PTB in juvenile mice. With this model, different degrees of RV pathology can be produced, ranging from RV hypertrophy to decompensated RV failure. This study includes detailed protocols for intubation, PTB surgery, and phenotyping by echocardiography.
Protocol
The study was approved by the Danish Animal Experiments Inspectorate (authorization number: 2021-15-0201-00928) and was performed in accordance with the national laboratory animal legislation. This study used 5-week-old male C57BL/6N mice.
1. Customization of instruments for intubation and surgery (Figure 1)
NOTE: This section details the most important steps in the preparation of custom-made instruments for intubation and PTB surgery from inexpensive and readily available materials.
- Prepare the endotracheal tube (Figure 1A).
Common intravenous (IV) catheters can be used for intubation and ventilation of mice.- Choose the appropriate size of intravascular catheter corresponding to the size of the mice. A 22G catheter is recommended for 5-week-old mice (17-20 g) and a 20G catheter for larger mice (>20 g).
- Pull out the needle and detach the locking mechanism. Insert the needle back into the catheter and cut the tip of the needle at a 45° angle, approximately 2 mm from the tip of the catheter.
- Use sandpaper to blunt the tip to prevent injury to the vocal cords of the mice.
- Cut off one wing of the catheter for a better view during intubation.
- Prepare the thoracic retractor (Figure 1B).
- Use a needle holder to bend an approximately 10 cm piece of flexible metal wire in the middle at a 30° angle.
- Use the needle holder to carefully create atraumatic hooks of 5 mm width on both ends of the wire.
- Prepare the intubation stand (Figure 1C).
NOTE: The use of an intubation stand enables continuous delivery of anesthesia via a nasal tube during intubation. This allows for controlled and safe intubation under visual guidance, which reduces the risk of injury to the vocal cords and trachea as well as the risk of misplacement of the tube in the esophagus. Any metal or plastic frame can be used as a frame for the intubation stand. A slightly bent kitchen role holder was used in this study.- Cut a 3 cm piece of rubber tubing that fits the snout of a mouse and connect it to an IV catheter valve. Inhalant anesthesia can be connected via the valve prior to intubation.
- Make a loop using a 1-0 braided suture approximately 5 mm from the opening of the tube. This will be used to secure the murine snout in the tube.
- Cut a hole near the top of a flexible plastic sheet and place the tube in the hole. The sheet is used to support the mouse in the intubation stand.
- Use tape to hold the individual parts together as shown in Figure 1C.
- Prepare the guidance cannula (Figure 1D, E).
- Pull a 6-0 monofilament suture through a blunt 27G cannula and tie a knot on the suture. Use this knot later to grasp the suture during PTB surgery.
- Use a needle holder to bend the tip of the cannula to an 80° angle.
- Prepare the ligating clip applier.
NOTE: A ligating clip applier with angled jaws is modified with an adjustable stop mechanism (Figure 1F), which stops the compression of the ligating clip when the jaws are at a precisely predetermined distance from each other. The modified ligating clip applier is used for the application of titanium ligating clips on the pulmonary trunk.- Fix a custom-cut piece of brass to the handles of the ligating clip applier with two screws. Mount an adjustable screw in the center (Figure 1F, white arrow) that determines the exact distance between the compressed handles of the ligating clip applier, which corresponds to a precise distance between the jaws.

Figure 1: Instruments for intubation and PTB surgery. (A) Endotracheal tube made from an IV catheter. (B) Thoracic retractor. (C) Intubation stand and mouse placed in intubation stand receiving anesthesia on a nasal tube. (D) Surgical instruments and modified ligating clip applier used for PTB surgery. (E) Guidance cannula. (F) Custom-made adjustable stop-mechanism. Please click here to view a larger version of this figure.
2. Adjustment of the ligating clip applier
- Choose the internal diameter of the ligating clip based on the desired severity of RV failure. For 5-week-old male C57BL/6N mice (17-20 g), use a clip diameter of 250 µm for severe and 450 µm for mild RV pressure overload. Use intermediate clip diameters to induce moderate pressure overload.
- Use metal wire or needles to adjust the ligating clip applier. Ensure that the diameter of the wire corresponds to the desired clip diameter.
- Load the clip applier with a ligating clip and place the adjustment wire in the center of the ligating clip. While compressing the clip applier, turn the screw until the jaws of the clip fit tightly around the wire. Ensure the clip stays in place on the adjustment wire once the clip applier is released.
- With the clip applier adjusted, place another ligating clip on the adjustment wire to validate the set diameter.
3. Preparations for surgery
- Induce anesthesia in an induction chamber using 7% sevoflurane in 0.6 L/min 100% oxygen. Confirm sufficient anesthesia by toe pinch prior to intubation.
NOTE: Isoflurane may also be used. Be aware that different concentrations should be used. - Intubate the mouse with a 22G IV catheter. Perform intubation under visual guidance, using a surgical microscope and an intubation stand, allowing for proper alignment for visualization of the vocal cords and continuous delivery of inhalant anesthetics on a nasal tube (Figure 1C).
- Ventilate the mouse at 175 strokes/min and a tidal volume of 300 µL/stroke.
NOTE: Tidal volumes of 8-10 µL/g are also recommended, and the ideal tidal volume depends on possible leakage and dead space in the ventilation system. - Place the intubated mouse on a covered heating pad (37 °C) and apply moisturizing ointment to the eyes of the mouse.
- Maintain anesthesia (3.5% sevoflurane in 0.6 L/min 100% oxygen) and administer 0.1 mg/kg buprenorphine and 5 mg/kg carprofen subcutaneously for perioperative analgesia. Use depilatory cream to remove all hair from the chest. Remove the cream after a maximum of 60 seconds to prevent skin irritation and disinfect the skin using disinfectant wipes.
4. PTB surgery
- Make a 10 mm incision in the skin above the second intercostal space from the sternal angle to the left anterior axillary line. Split the major and minor pectoral muscles by blunt dissection.
- Cut the intercostal muscles in the second intercostal space and bluntly dissect the thymus to expose the heart, pulmonary trunk, and aorta. Place a thoracic retractor in the intercostal space to keep the operating field accessible.
NOTE: Great caution must be used when cutting the intercostal muscles, as the left internal mammary artery runs just 1-2 mm laterally of the sternum. Injury to this artery may result in significant blood loss. - Separate the pulmonary trunk from the ascending aorta by bluntly removing the connective tissue between the vessels using microscopic forceps. Improve the exposure of the pulmonary trunk further by rotating the lower body of the mouse (left leg over right leg).
- Pass the guidance cannula through the transverse pericardial sinus posteriorly of the pulmonary trunk. Use forceps to grasp the knot on the tip of the guidance cannula and pull the suture through the guidance cannula. Carefully remove the guidance cannula while the suture stays in place around the pulmonary trunk.
- Load the ligating clip applier and use the suture to guide the pulmonary trunk into the jaws of the ligating clip and compress the clip. Release the suture immediately after placement of the clip and observe the filling of the pulmonary trunk.
NOTE: Bradycardia may be observed in the first few seconds after application of the ligating clip. - Place a 6-0 monofilament absorbable suture around the second and third costae and close the intercostal space. Evacuate as much air as possible from the thoracic cavity by applying gentle pressure on the chest while tightening the suture.
- Lastly, suture the skin with a 6-0 monofilament absorbable suture.
- Perform the same procedure, except for placing the ligating clip (step 4.5), during sham surgery.
- To ensure adequate postoperative analgesia, add buprenorphine (7.5 mg/L) to the drinking water and administer a subcutaneous injection of carprofen (5 mg/kg) daily for the first three postoperative days. Monitor water intake to ensure sufficient consumption.
5. Echocardiography
- After induction of anesthesia in an induction chamber (1-2 min, 6% sevoflurane in 100% oxygen), maintain anesthesia using a nasal tube (3.5% sevoflurane in 100% oxygen). Remove all hair from the chest and abdomen using depilatory cream and place the mouse on a heating pad. Apply moisturizing ointment to the eyes and ultrasound gel to the mouse's chest.
NOTE: Adjust 2D gain, focus depth, and image depth to improve image quality in all echocardiographic measurements. - Adjust the ultrasonic probe to find the parasternal long axis (PLAX) view (Figure 2). In PLAX, measure the pulmonary trunk inner diameter and velocity time integral (VTI) in the pulmonary trunk.
- Select B-mode and carefully move the heating pad in the x-, y- and z-axis to identify the pulmonary trunk in the center of the image. Use color to identify the largest pulmonary trunk diameter. Use cine store to capture a sequence for measurement of the pulmonary trunk diameter.
- Select color and pulse wave (PW) Doppler and place the cursor in the center of the pulmonary trunk. Adjust the PW angle until the dotted lines are parallel to the blood flow in the vessel.
- Press cine store to measure VTI. Place the cursor near both walls of the pulmonary trunk and press cine store again to acquire flow near the vessel wall.
NOTE: Right ventricular (RV) inner diameter and RV free wall thickness may also be assessed in the PLAX view.
- Find the parasternal short axis (PSAX) view (Figure 3) to measure left ventricular (LV) inner diameters, which can be used to assess septal bulging (D-configuration).
- Select B-mode and turn the probe 90° anti-clockwise. Angle the probe 20-30 degrees laterally to avoid shadowing of the RV by the sternum, and tilt the probe 20-30 degrees cranially until the left ventricle is as round as possible. Then, slide the probe in the craniocaudal direction to identify the level of the papillary muscles with the largest ventricular diameters and press cine store.
NOTE: PSAX at the midpapillary level may also be used to assess RV fractional area change. PSAX at the level of the aortic valve may be used to measure RV fractional shortening or RV free wall thickness.
- Select B-mode and turn the probe 90° anti-clockwise. Angle the probe 20-30 degrees laterally to avoid shadowing of the RV by the sternum, and tilt the probe 20-30 degrees cranially until the left ventricle is as round as possible. Then, slide the probe in the craniocaudal direction to identify the level of the papillary muscles with the largest ventricular diameters and press cine store.
- Use the apical 4-chamber (A4CH) view (Figure 4) to measure the tricuspid annular plane systolic excursion (TAPSE) and assess for tricuspid regurgitation.
- Position the probe as shown in Figure 4. Once the heart is identified, make small adjustments to the probe using only the wrist and fingers to identify all four chambers of the heart and the tricuspid valve.
- Identifying and maintaining a good A4CH view is challenging. Allow the probe-operating hand to rest on the heating pad for stability. Slide, tilt, and turn the probe slightly until the appropriate image is found.
- Only move the probe in one dimension at a time: E.g., slide craniocaudal to find the heart, then tilt the probe to identify all four chambers and finally turn the probe until all four chambers and the tricuspid valve are in the frame. If necessary, repeat all three steps several times before obtaining the ideal image.
- Once the tricuspid valve is identified, select M-mode and place the dotted line in the tricuspid annulus of the free wall. Press cine store to save the measurements.
- Select color to assess the tricuspid valve for regurgitation. If regurgitation is present, a jet of retrograde flow from the RV to the right atrium (RA) will be seen in systole (Figure 5).
NOTE: In the A4CH view, tissue Doppler in the RV free wall and RV inflow velocity may also be measured.
- Position the probe as shown in Figure 4. Once the heart is identified, make small adjustments to the probe using only the wrist and fingers to identify all four chambers of the heart and the tricuspid valve.
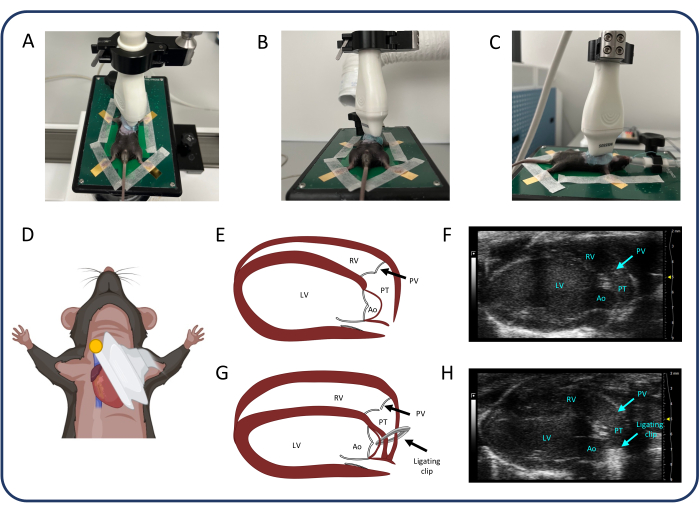
Figure 2: Parasternal long axis view (PLAX). (A-D) Positioning of the ultrasonic probe. (E, F) The normal murine heart in PLAX. (G, H) RV dilation and hypertrophy after PTB. Abbreviations: LV: left ventricle, RV: right ventricle, PV: pulmonary valve, PT: pulmonary trunk, Ao: aorta. Please click here to view a larger version of this figure.
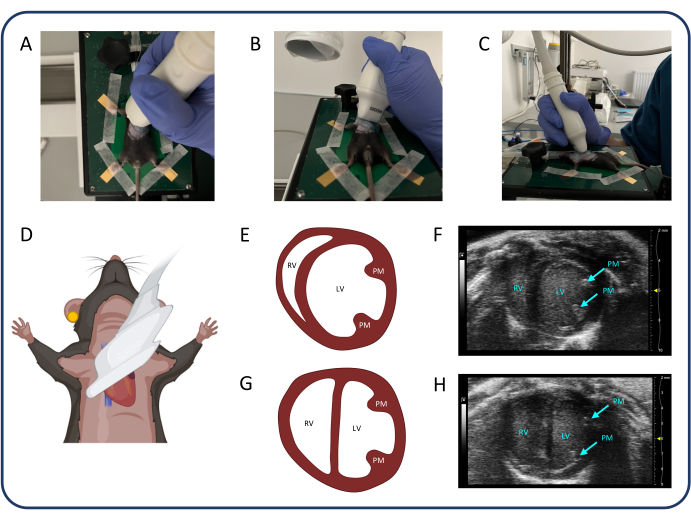
Figure 3: Parasternal short axis view (PSAX). (A-D) Positioning of the ultrasonic probe. (E, F) The normal murine heart in PSAX. (G, H) PSAX after PTB. Abbreviations: LV: left ventricle, RV: right ventricle, PM: papillary muscle. Please click here to view a larger version of this figure.
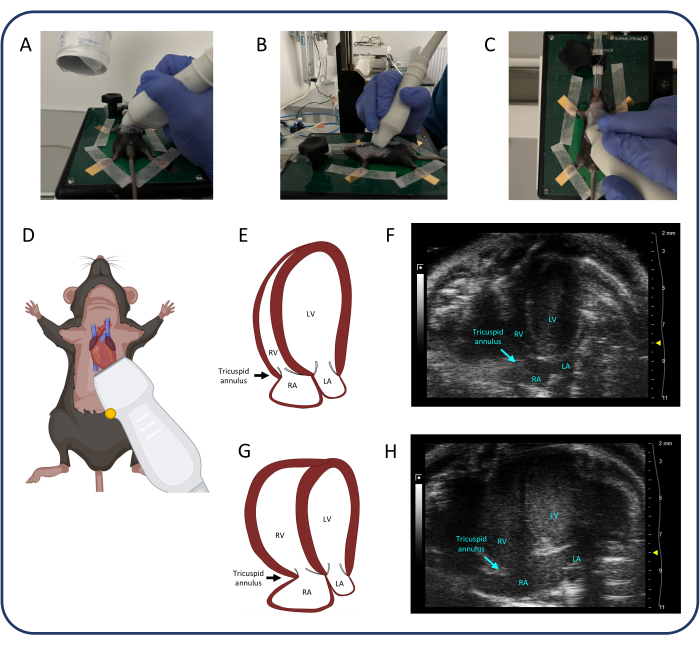
Figure 4: Apical 4-chamber view (A4CH). (A-D) Positioning of the ultrasonic probe. (E, F) The normal murine heart in the A4CH view. (G, H) RV and RA dilatation after PTB. Abbreviations: LV: left ventricle, RV: right ventricle, RA: right atrium, LA: left atrium. Please click here to view a larger version of this figure.
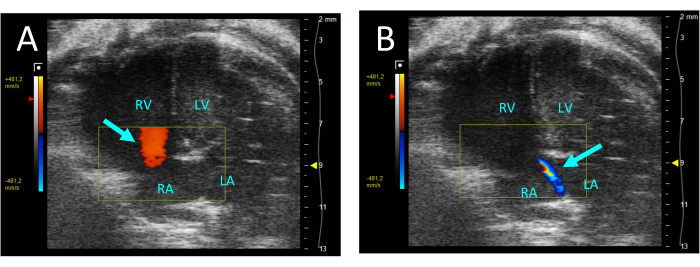
Figure 5: Tricuspid regurgitation visualized by color Doppler in the A4CH-view. (A) In diastole, flow from the RA to the RV is observed (arrow). (B) During systole, a thin jet of flow from the RV to the RA is visible (arrow). Abbreviations: LV: left ventricle, RV: right ventricle, RA: right atrium, LA: left atrium. Please click here to view a larger version of this figure.
6. Data analyses
- Measure the pulmonary trunk diameter in three cardiac cycles in PLAX and use the mean pulmonary trunk diameter for further data analyses. Measure VTI in three cardiac cycles in PLAX for each of the three stored cine loops (in the center of the pulmonary trunk and near the walls of the vessel). Use mean VTI of all VTI measurements for further analysis. Use the following formula to calculate CO:
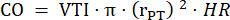
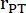 : radius of the pulmonary trunk, HR: heart rate
: radius of the pulmonary trunk, HR: heart rate - Measure LVEI in PSAX at midpapillary level. Use a measuring tool to measure the largest LV inner diameter (LVid1) from mid-septum to the free wall. Then, measure the LV inner diameter orthogonal to the first measurement (LVid2). Repeat these measurements in three cardiac cycles and calculate LVEI by using the mean LV inner diameters and the following formula:
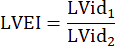
- Measure TAPSE in three cardiac cycles in the A4CH view and use mean TAPSE for further data analyses.
7. Right heart catheterization
- Measure the right ventricular (RV) pressure and volume by right heart catheterization with a 1.4F micro-tip catheter 3 weeks after PTB surgery.
- Anesthetize and intubate the mouse as described in steps 3.1-3.4. Place the mouse on a covered heating pad (37 °C) and maintain anesthesia (3.5% sevoflurane in 0.6 L/min 100% oxygen).
- Administer 2000 IU of Heparin (intramuscular [i.m.]) and 0.5 mL of NaCl (subcutaneous [s.c.]).
- Using surgical scissors, cut the abdominal wall just caudally to the xiphoid process and gain access to the thoracic cavity by carefully cutting the diaphragm along its incision in the thoracic wall. Cut the diaphragm and costae until sufficient access to the heart has been obtained.
- Place a ligature around the inferior vena cava. Use this for occluding the vessel to reduce preload for the recording of pressure-volume measurements later in the protocol.
- Use a 26G needle to carefully poke a small hole in the RV. Ensure this is as near to the apex as possible and the needle does not penetrate the ventricle entirely but merely acts as a guide for insertion of the conductance catheter. If bleeding occurs, apply gentle pressure with a small cotton swab to minimize blood loss.
- Identify the small hole in the ventricular wall and insert the catheter by penetrating the tissue.
NOTE: When inserting the catheter, be careful not to damage the inner ventricular wall. - Changes in right ventricular pressure (RVP) are often observed for several minutes after insertion of the catheter. Wait until RVP stabilizes to obtain representative steady-state measurements.
- To obtain pressure-volume loops, use the ligature previously placed around the inferior vena cava. Carefully pull the ligature to occlude the vessel, thereby gradually reducing preload.
- Once representative pressure-volume loops have been recorded, extract the catheter, and euthanize the mouse by excision of the heart. At this time, collect blood and tissue samples for further analysis.
Results
C57BL/6N mice (male, 5-week old, 17-20 g) were randomized to either severe PTB (sPTB, 250 µm, n = 12), mild PTB (mPTB, 450 µm, n = 9), or sham surgery (sham, n = 15). Evaluation of cardiac function was performed by echocardiography 1 week and 3 weeks after surgery. Right heart catheterization with subsequent euthanasia was performed 3 weeks post-surgery. Organs were weighed, and cardiac tissue was prepared for histological analyses.
Echocardiography 1 week after surgery revealed...
Discussion
In this paper, we present a murine model of pressure overload-induced RV hypertrophy and failure. We demonstrate that: (i) PTB in juvenile mice can induce varying degrees of RV pathology, ranging from mild RV hypertrophy to RV failure with extracardiac signs of decompensation and histologically confirmed RV fibrosis. (ii) Signs of RV dysfunction can be observed and quantified by echocardiography at 1 and 3 weeks after PTB surgery. (iii) The degree of RV hypertrophy is proportional to the severity of PTB and the...
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by Snedkermester Sophus Jacobsen og Hustru Astrid Jacobsens Fond, Helge Peetz og Verner Peetz og hustru Vilma Peetz Legat, Grosserer A.V. Lykfeldt og Hustrus Legat. Furthermore, the authors would like to acknowledge the staff of the animal facilities at the Department of Clinical Medicine, Aarhus University, for their support during the execution of the experimental work.
Materials
| Name | Company | Catalog Number | Comments |
| Biosyn 6-0, monofilament, absorbable suture | Covidien | UM-986 | |
| Blunt cannula, 27G 0.4x0.25, | Sterican | 292832 | |
| Bupaq Multidose vet 0,3 mg/ml (Buprenorphinum) | Salfarm Danmark | VNR 472318 | |
| C57BL/6NTac mice | Taconic Biosciences | C57BL/6NTac | |
| Dagrofil 1, braided, non-absorbable suture | B Braun | C0842273 | |
| Depilatory cream | Veet | 3132000 | |
| Disinfection Swabs (82% Ethanol + 0.5% Chlorhexidine) | Mediq | 3340122 | |
| Disposable scalpels, size 11 | Swann-Morton | 11708353 | |
| Dräger Vapor 2000 Sevoflurane | Dräger | M35054 | |
| Eye oinment neutral, "Ophta" | Actavis | MTnr.: 07586 Vnr: 53 96 68 | |
| Horizon ligating clips | Teleflex Medical | 5200 (IPN914931) | |
| Horizon Open Ligating Clips applier, curved, 6" (15 cm) | Teleflex Medical | 537061 | |
| Kitchen roll holder | n.a. | n.a. | |
| Metal wire of different thickness | n.a. | n.a. | |
| Microsurgical instruments set | Thompson | n.a. | |
| MiniVent Ventilator | Hugo Sachs | Type 845 | |
| MS505S transducer | Visual sonics | n.a. | |
| Rimadyl Bovis vet. 50 mg/ml (Carprofen) | Zoetis | MTnr: 34547, Vnr: 10 27 99, | |
| Sevoflurane Baxter 100 % | Baxter Medical | MTnr: 35015 | |
| Silicone tubing | n.a. | n.a. | |
| Soft plastic sheet | n.a. | n.a. | |
| Stereomicroscope, "Opmi Pico" | Carl Zeiss Surgicals GmbH | n.a. | |
| Ultrasonic probe holder/rail | Visual Sonics | 11277 | |
| Varming plate | Visual sonics | 11437 | |
| Venflon ProSafety, 22G, 0,9 x 25mm | Becton Dickinson | 393222 |
References
- Voelkel, N. F., et al. Right ventricular function and failure: Report of a national heart, lung, and blood institute working group on cellular and molecular mechanisms of right heart failure. Circulation. 114 (17), 1883-1891 (2006).
- Haddad, F., Doyle, R., Murphy, D. J., Hunt, S. A. Right ventricular function in cardiovascular disease, part ii: Pathophysiology, clinical importance, and management of right ventricular failure. Circulation. 117 (13), 1717-1731 (2008).
- Van De Veerdonk, M. C., et al. Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. J Am Coll Cardiol. 58 (24), 2511-2519 (2011).
- Gomez-Arroyo, J., et al. A brief overview of mouse models of pulmonary arterial hypertension: Problems and prospects. Am J Physiol Lung Cell Mol Physiol. 302 (10), L977-L991 (2012).
- Maarman, G., Lecour, S., Butrous, G., Thienemann, F., Sliwa, K. A comprehensive review: The evolution of animal models in pulmonary hypertension research; are we there yet. Pulm Circ. 3 (4), 739-756 (2013).
- Andersen, A., et al. Animal models of right heart failure. Cardiovasc Diagn Ther. 10 (5), 1561-1579 (2020).
- Voelkel, N. F., Tuder, R. M. Hypoxia-induced pulmonary vascular remodeling: A model for what human disease. J Clin Invest. 106 (6), 733-738 (2000).
- Rabinovitch, M., Gamble, W., Nadas, A. S., Miettinen, O. S., Reid, L. Rat pulmonary circulation after chronic hypoxia: Hemodynamic and structural features. Am J Physiol. 236 (6), H818-H827 (1979).
- Taraseviciene-Stewart, L., et al. Inhibition of the VEGF receptor 2 combined with chronic hypoxia causes cell death-dependent pulmonary endothelial cell proliferation and severe pulmonary hypertension. Faseb j. 15 (2), 427-438 (2001).
- Ciuclan, L., et al. A novel murine model of severe pulmonary arterial hypertension. Am J Respir Crit Care Med. 184 (10), 1171-1182 (2011).
- Nicolls, M. R., et al. New models of pulmonary hypertension based on VEGF receptor blockade-induced endothelial cell apoptosis. Pulm Circ. 2 (4), 434-442 (2012).
- Hessel, M. H., Steendijk, P., Den Adel, B., Schutte, C. I., Van Der Laarse, A. Characterization of right ventricular function after monocrotaline-induced pulmonary hypertension in the intact rat. Am J Physiol Heart Circ Physiol. 291 (5), H2424-H2430 (2006).
- Gomez-Arroyo, J. G., et al. The monocrotaline model of pulmonary hypertension in perspective. Am J Physiol Lung Cell Mol Physiol. 302 (4), L363-L369 (2012).
- Janssen, W., et al. 5-ht2b receptor antagonists inhibit fibrosis and protect from RV heart failure. Biomed Res Int. 2015, 438403 (2015).
- Andersen, S., et al. A pulmonary trunk banding model of pressure overload induced right ventricular hypertrophy and failure. J Vis Exp. (141), e58050 (2018).
- Axelsen, J. B., et al. Effects of 6-mercaptopurine in pressure overload induced right heart failure. PLoS One. 14 (11), e0225122 (2019).
- Egemnazarov, B., et al. Pressure overload creates right ventricular diastolic dysfunction in a mouse model: Assessment by echocardiography. J Am Soc Echocardiogr. 28 (7), 828-843 (2015).
- Wang, Q., et al. Induction of right ventricular failure by pulmonary artery constriction and evaluation of right ventricular function in mice. J Vis Exp. (147), e59431 (2019).
- Kojonazarov, B., et al. The peroxisome proliferator-activated receptor β/δ agonist gw0742 has direct protective effects on right heart hypertrophy. Pulm Circ. 3 (4), 926-935 (2013).
- Kojonazarov, B., et al. P38 MAPK inhibition improves heart function in pressure-loaded right ventricular hypertrophy. Am J Respir Cell Mol Biol. 57 (5), 603-614 (2017).
- Rai, N., et al. Effect of Riociguat and Sildenafil on right heart remodeling and function in pressure overload induced model of pulmonary arterial banding. Biomed Res Int. 2018, 3293584 (2018).
- Sydykov, A., et al. Genetic deficiency and pharmacological stabilization of mast cells ameliorate pressure overload-induced maladaptive right ventricular remodeling in mice. Int J Mol Sci. 21 (23), 9099 (2020).
- Andersen, S., et al. Effects of combined angiotensin ii receptor antagonism and neprilysin inhibition in experimental pulmonary hypertension and right ventricular failure. Int J Cardiol. 293, 203-210 (2019).
- Andersen, S., et al. Pressure overload induced right ventricular remodeling is not attenuated by the anti-fibrotic agent pirfenidone. Pulm Circ. 9 (2), 2045894019848659 (2019).
- Labazi, H., et al. Sex-dependent changes in right ventricular gene expression in response to pressure overload in a rat model of pulmonary trunk banding. Biomedicines. 8 (10), 430 (2020).
- Sun, X. Q., et al. Increased mao-a activity promotes progression of pulmonary arterial hypertension. Am J Respir Cell Mol Biol. 64 (3), 331-343 (2021).
- Axelsen, J. S., et al. Effects of Empagliflozin on right ventricular adaptation to pressure overload. Front Cardiovasc Med. 10, 1302265 (2023).
- Mamazhakypov, A., Veith, C., Schermuly, R. T., Sydykov, A. Surgical protocol for pulmonary artery banding in mice to generate a model of pressure-overload-induced right ventricular failure. STAR Protoc. 4 (4), 102660 (2023).
- Boehm, M., et al. Delineating the molecular and histological events that govern right ventricular recovery using a novel mouse model of pulmonary artery de-banding. Cardiovasc Res. 116 (10), 1700-1709 (2020).
- Andersen, S., et al. Effects of bisoprolol and losartan treatment in the hypertrophic and failing right heart. J Card Fail. 20 (11), 864-873 (2014).
- Hirata, M., et al. Novel model of pulmonary artery banding leading to right heart failure in rats. Biomed Res Int. 2015, 753210 (2015).
- Vildbrad, M. D., et al. Limitations and pitfalls in measurements of right ventricular stroke volume in an animal model of right heart failure. Physiol Meas. 36 (5), 925-937 (2015).
- Boehm, M., et al. Maintained right ventricular pressure overload induces ventricular-arterial decoupling in mice. Exp Physiol. 102 (2), 180-189 (2017).
- Cheng, H. W., et al. Assessment of right ventricular structure and function in mouse model of pulmonary artery constriction by transthoracic echocardiography. J Vis Exp. (84), e51041 (2014).
- Luitel, H., et al. Pressure overload leads to an increased accumulation and activity of mast cells in the right ventricle. Physiol Rep. 5 (6), e13146 (2017).
- Mamazhakypov, A., et al. Novel therapeutic targets for the treatment of right ventricular remodeling: Insights from the pulmonary artery banding model. Int J Environ Res Public Health. 18 (16), 8297 (2021).
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionThis article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved